Abstract
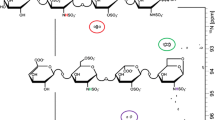
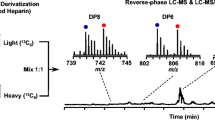
Studies on the structure-function relationships of the complex linear polysaccha-rides, known as glycosaminoglycans (GAGs), are becoming increasingly important as biological functions are established for them. However, structural analysis of GAGs presents a difficult technical problem, particularly in the case of the N-sulfated GAGs heparan sulfate (HS) and heparin, which display remarkable structural diversity (1). A widely used and effective approach is to degrade the chains into smaller saccharide units that can then be separated and analyzed. In this regard, strong anion-exchange (SAX) HPLC techniques have proved particularly useful for both the analysis of disac-charide composition (2,3) and the separation of complex mixtures of larger saccharides (3–6). However, in many methods the columns used have been silica-based and suffer from drawbacks related to poor stability of the support (e.g., inconsistency of run times, peak broadening, and short column life). There is clearly a need for improvements in column performance, especially for the purification of larger saccharides to homogeneity for sequencing and bioactivity testing. This chapter describes how a single type of polymer-based SAX column, ProPac PA1, can be used to provide high-resolution separations of both disaccharides and larger oligosaccharides derived from HS and heparin, with consistent elution times and excellent column performance characteristics (see Notes 1 and 2). Disaccharides from chondroitin sulfate and dermatan sulfate can also be separated (see Note 3). The improved resolution of saccharides compared to other SAX-HPLC methods, combined with the versatility and longevity of the columns, makes them a valuable tool for purification and structural analysis of HS/heparin and other GAG saccharides.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Gallagher, J. T., Turnbull, J. E., and Lyon, M. (1992) Patterns of sulphation in heparan sulphate: polymorphism based on a common structural theme. Int. J. Biochem. 24, 553–556.
Yoshida, K., Miyauchi, S., Kikuchi, H., Tawada, A., and Tokuyasu, K. (1989) Analysis of unsaturated disaccharides from glycosaminoglycuronan by HPLC. Anal. Biochem. 177, 327–332.
Bienkowski, M. J. and Conrad, H. E. (1984) Structural characterisation of the oligosac-charides formed by depolymerisation of heparin with nitrous acid. J. Biol. Chem. 260, 356–365.
Guo, Y. and Conrad, H. E. (1988) Analysis of oligosaccharides from heparin by reversed-phase ion pairing HPLC. Anal. Biochem. 168, 54–62.
Linhardt, R. J., Gu, K., Loganathan, D., and Carter, S. R. (1989). Analysis of glycosami-noglycan-derived oligosaccharides using reverse-phase ion-pairing and ion-exchange chro-matography with suppressed conductivity detection. Anal. Biochem. 181, 288–296.
Rice, K. G., Kim, Y., Grant, A., Merchant, Z., and Linhardt, R. J. (1985) HPLC separation of heparin-derived oligosaccharides. Anal. Biochem. 150,325–331.
Turnbull, J. E. and Gallagher, J. T. (1991) Distribution of iduronate-2-sulphate residues in heparan sulphate: evidence for an ordered polymeric structure. Biochem. J. 273, 553–559.
Turnbull, J. E., Fernig, D., Ke, Y., Wilkinson, M. C., and Gallagher, J. T. (1992) Identification of the basic FGF binding sequence in fibroblast HS.J. Biol. Chem. 267, 10,337–10,341.
Walker, A., Turnbull, J. E., and Gallagher, J. T. (1994) Specific HS saccharides mediate the activity of basic FGF. J. Biol. Chem. 269, 931–935.
Midura, R. J., Salustri, A., Calabro, A., Yanagashita, M., and Hascall, V. C. (1994) High-resolution separation of disaccharide and oligosaccharide alditols from chondroitin sulphate, dermatan sulphate and hyaluronan using CarboPac PA1 chromatography. Glycobiology 4, 333–342.
Sanderson, R. D., Turnbull, J. E., Gallagher, J. T., and Lander, A.D. (1994) Fine structure of HS regulates cell syndecan-1 function and cell behaviour. J. Biol. Chem. 269, 13,100–13,106.
Turnbull, J. E., Hopwood, J., and Gallagher, J. T. (1999) A strategy for rapid sequencing of heparan sulphate/heparin saccharides. Proc. Nat. Acad. Sci. USA 96, 2698–2703
Guimond, S. E. and Turnbull, J. E. (1999) Fibroblast growth factor receptor signalling is dictated by specific heparan sulphate saccharides. Current Biology 9, 1343–1346.
Linhardt R. J., Turnbull J. E., Wang H., Loganathan D., and Gallagher J. T. (1990) Examination of the substrate specificity of heparin and heparan sulphate lyases. Biochemistry 29,2611–2617.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2001 Humana Press Inc., Totowa, NJ
About this protocol
Cite this protocol
Turnbull, J.E. (2001). Analytical and Preparative Strong Anion-Exchange HPLC of Heparan Sulfate and Heparin Saccharides. In: Iozzo, R.V. (eds) Proteoglycan Protocols. Methods in Molecular Biology™, vol 171. Humana Press. https://doi.org/10.1385/1-59259-209-0:141
Download citation
DOI: https://doi.org/10.1385/1-59259-209-0:141
Publisher Name: Humana Press
Print ISBN: 978-0-89603-759-5
Online ISBN: 978-1-59259-209-8
eBook Packages: Springer Protocols