Summary
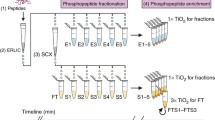
Mass spectrometry-based protein phosphorylation analysis on a proteome-wide scale remains a formida ble challenge, hampered by the complexity and dynamic range of protein expression on the global level and multi-site phosphorylation at substoichiometric ratios at the individual protein level. It is recognized that reduction of sample complexity or enrichment of the phosphopeptide pool is a necessary prereq uisite for global phospho-proteomics. Immobilized metal affinity chromatography (IMAC) and strong cation exchange chromatography, either alone or in tandem, have emerged as the most widely used chromatographic-based enrichment strategies. However, each is not without shortcomings. Both tech niques provide little fractionation of phosphorylated species and are compromised by competition and co-elution of highly acidic peptides. Here, we describe a phosphopeptide prefractionation scheme using hydrophilic interaction chromatography, which both enriches the phosphopeptide pool and efficiently fractionates the remaining peptides. When used in front of IMAC, the selectivity of the metal affinity resin is improved to greater than 95%. The lack of significant numbers of nonphosphorylated peptides also allows for more efficient use of the mass spectrometer duty cycle in that the instrument spends nearly all of its time in sequencing the phosphopeptides.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
de Godoy LM, Olsen JV, de Souza GA, Li G, Mortensen P, Mann M, (2006) Status of com plete proteome analysis by mass spectrom-etry: SILAC labeled yeast as a model system. Genome Biol 6, R50.
Beausoleil SA, Jedrychowski M, Schwartz D, Elias JE, Villen J, Li J, Cohn MA, Cantley LC, Gygi SP, (2004) Large-scale characterization of HeLa cell nuclear phosphoproteins. Proc Natl Acad Sci USA 101, 12130–12135.
Beausoleil SA, Villen J, Gerber SA, Rush J, Gygi SP, (2006) A probability-based approach for high-throughput protein phosphorylation analysis and site localization. Nat Biotechnol 24, 1285–1292.
Olsen JV, Blagoev B, Gnad F, Macek B, Kumar C, Mortensen P, Mann M, (2006) Global, in vivo, and site-specific phosphoryla-tion dynamics in signaling networks. Cell 127, 635–648.
Nuwaysir LM, Stults JT, (1993) Electrospray ionization mass spectrometry of phosphopep-tides isolated by on-line immobilized metal-ion affinity chromatography. J Am Soc Mass Spectrom 4, 662–667.
Posewitz MC, Tempst P (1999) Immobilized gallium(III) affinity chromatography of phos-phopeptides. Anal Chem 71, 2883–2892.
Pinkse MW, Uitto PM, Hilhorst MJ, Ooms B, Heck AJ, (2006) Selective isolation at the fem-tomole level of phosphopeptides from proteo-lytic digests using 2D-NanoLC-ESI-MS/MS and titanium oxide precolumns. Anal Chem 76, 3935–3943.
Larsen MR, Thingholm TE, Jensen ON, Roepstorff P, Jorgensen TJ, (2005) Highly selective enrichment of phosphorylated peptides from peptide mixtures using tita nium dioxide microcolumns. Mol Cell Pro-teomics 4, 873–886.
Ficarro SB, McCleland ML, Stukenberg PT, Burke DJ, Ross MM, Shabanowitz J, Hunt DF, White FM, (2002) Phosphoproteome analysis by mass spectrometry and its application to Saccharomyces cerevisiae. Nat Biotechnol 20, 301–305.
Alpert AJ. (1990) Hydrophilic-interaction chromatography for the separation of pep-tides, nucleic acids and other polar com pounds. J Chromatogr 499, 177–196.
Yoshida T, Okada T, (1999) Peptide separation in normal-phase liquid chromatography: Study of selectivity and mobile phase effects on various columns. J Chromatogr A 840, 1–9.
Gilar M, Olivova P, Daly AE, Gebler JC, (2005) Orthogonality of separation in two-dimensional liquid chromatography. Anal Chem 77, 6426–6434.
McNulty DE and Annan RS, (2008) Hydrophilic-interaction chromatography reduces the complexity of the phosphopro-teome and improves global phosphopeptide isolation and detection. Mol Cell Proteomics 7, 971–980.
Thingholm TE, Jensen ON, Robinson PJ, Larsen MR. (2008) SIMAC – A phospho-proteomics strategy for the rapid separation of monophosphorylated from multiply phos-phorylated peptides. Mol Cell Proteomics 7, 661–671.
Elias JE, Gygi SP. (2007) Target-decoy search strategy for increased confidence in large-scale protein identifications by mass spectrometry. Nat Methods 4, 207–214.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
McNulty, D.E., Annan, R.S. (2009). Hydrophilic Interaction Chromatography for Fractionation and Enrichment of the Phosphoproteome. In: Graauw, M.d. (eds) Phospho-Proteomics. Methods in Molecular Biology™, vol 527. Humana Press. https://doi.org/10.1007/978-1-60327-834-8_8
Download citation
DOI: https://doi.org/10.1007/978-1-60327-834-8_8
Publisher Name: Humana Press
Print ISBN: 978-1-60327-833-1
Online ISBN: 978-1-60327-834-8
eBook Packages: Springer Protocols