Abstract
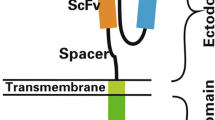
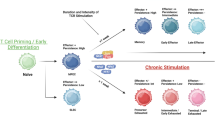
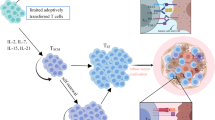
T cells engineered to express chimeric antigen receptor (CAR) against the B cell antigen CD19 are achieving remarkable clinical effects on hematological malignancies. Allogeneic transplantation approach is promising for broaden application of CART therapy. iPSCs are one of the ideal cell sources for this approach. CAR-engineered iPSCs are demonstrated to give rise to CAR-engineered T cell and exert their effector function. In this section, we describe the method to generate CAR-engineered iPSCs and differentiate them into T cells.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lee DW et al (2015) T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385:517–528
Maude SL et al (2014) Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med 371:1507–1517
Davila ML et al (2014) Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med 6:224ra25–224ra25
Themeli M, Rivière I, Sadelain M (2015) New cell sources for T cell engineering and adoptive immunotherapy. Cell Stem Cell 16:357–366
Qasim W et al (2017) Molecular remission of infant B-ALL after infusion of universal TALEN gene-edited CAR T cells. Sci Transl Med 9:1–9
Nishimura T et al (2013) Generation of rejuvenated antigen-specific T cells by reprogramming to pluripotency and redifferentiation. Cell Stem Cell 12:114–126
Vizcardo R et al (2013) Regeneration of human tumor antigen-specific T cells from iPSCs derived from mature CD8+ T cells. Cell Stem Cell 12:31–36
Kitayama S et al (2016) Cellular adjuvant properties, direct cytotoxicity of re-differentiated Vα24 invariant NKT-like cells from human induced pluripotent stem cells. Stem Cell Reports 6:213–227
Themeli M et al (2013) Generation of tumor-targeted human T lymphocytes from induced pluripotent stem cells for cancer therapy. Nat Biotechnol 31:928–933
Zou J et al (2011) Plenary paper oxidase-deficient neutrophils from X-linked chronic granulomatous disease iPS cells: functional correction by zinc finger nuclease – mediated safe harbor targeting. Blood 117:5561–5572
Hong S et al (2007) Functional analysis of various promoters in lentiviral vectors at different stages of in vitro differentiation of mouse embryonic stem cells. Mol Ther 15:1630–1639
Jiang H et al (2010) Hemato-endothelial differentiation from lentiviral-transduced human embryonic stem cells retains durable reporter gene expression under the control of ubiquitin promoter. Cytotechnology 62:31–42
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Ueda, T., Kaneko, S. (2019). In Vitro Differentiation of T Cell: From CAR-Modified T-iPSC. In: Kaneko, S. (eds) In Vitro Differentiation of T-Cells. Methods in Molecular Biology, vol 2048. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9728-2_10
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9728-2_10
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9727-5
Online ISBN: 978-1-4939-9728-2
eBook Packages: Springer Protocols