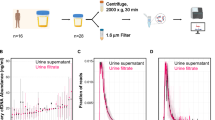
Abstract
Urine cell-free DNA is an important source of diagnostic markers for different diseases (e.g., cancer and prenatal diagnosis). It is important to achieve a simple and fast protocol to maximize the recovery of DNA from urine supernatant and to assess its quality. Here we describe a simple approach from urine collection to DNA quality assessment for downstream analyses.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ponti G, Maccaferri M, Manfredini M et al (2018) The value of fluorimetry (Qubit) and spectrophotometry (NanoDrop) in the quantification of cell-free DNA (cfDNA) in malignant melanoma and prostate cancer patients. Clin Chim Acta 479:14–19
Bartels S, Persing S, Hasemeier B et al (2017) Molecular analysis of circulating cell-free DNA from lung cancer patients in routine laboratory practice: a cross-platform comparison of three different molecular methods for mutation detection. J Mol Diagn 19(5):722–732
Lu T, Li J (2017) Clinical applications of urinary cell-free DNA in cancer: current insights and promising future. Am J Cancer Res 7(11):2318–2332
Salvi S, Martignano F, Molinari C et al (2016) The potential use of urine cell free DNA as a marker for cancer. Expert Rev Mol Diagn 16(12):1283–1290
Salvi S, Gurioli G, Martignano F et al (2015) Urine cell-free DNA integrity analysis for early detection of prostate cancer patients. Dis Markers 2015:574120
Casadio V, Calistri D, Tebaldi M et al (2013) Urine cell-free DNA integrity as a marker for early bladder cancer diagnosis: preliminary data. Urol Oncol 31(8):1744–1750
Sands J, Li Q, Hornberger J (2017) Urine circulating-tumor DNA (ctDNA) detection of acquired EGFR T790M mutation in non-small-cell lung cancer: an outcomes and total cost-of-care analysis. Lung Cancer 110:19–25
Botezatu I, Serdyuk O, Potapova G et al (2000) Genetic analysis of DNA excreted in urine: a new approach for detecting specific genomic DNA sequences from cells dying in an organism. Clin Chem 46(8 Pt 1):1078–1084
Majer S, Bauer M, Magnet E, Strele A et al (2007) Maternal urine for prenatal diagnosis—an analysis of cell-free fetal DNA in maternal urine and plasma in the third trimester. Prenat Diagn 27(13):1219–1223
Tsui NB, Jiang P, Chow KC et al (2012) High resolution size analysis of fetal DNA in the urine of pregnant women by paired-end massively parallel sequencing. PLoS One 7(10):e48319
Lin SY, Linehan JA, Wilson TG et al (2017) Emerging utility of urinary cell-free nucleic acid biomarkers for prostate, bladder, and renal cancers. Eur Urol Focus 3(2–3):265–272
Casadio V, Salvi S, Martignano F et al (2017) Cell-free DNA integrity analysis in urine samples. J Vis Exp (119)
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Casadio, V., Salvi, S. (2019). Urinary Cell-Free DNA: Isolation, Quantification, and Quality Assessment. In: Casadio, V., Salvi, S. (eds) Cell-free DNA as Diagnostic Markers. Methods in Molecular Biology, vol 1909. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8973-7_16
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8973-7_16
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8972-0
Online ISBN: 978-1-4939-8973-7
eBook Packages: Springer Protocols