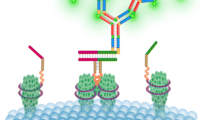
Abstract
Activation of receptor tyrosine kinases (RTK), such as those belonging to the human epidermal growth factor receptor (HER) family, occurs only after receptor dimerization, which is a crucial step for cellular signal transduction and diversification. The HER family includes four members (EGFR/HER1, HER2, HER3, and HER4) that can homodimerize or heterodimerize. Here, we describe immunoassays based on time-resolved Förster resonance energy transfer (TR-FRET) to profile EGFR-EGFR, HER2-HER2, and EGFR-HER2 dimers directly in tumor samples.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V et al (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344:783–792
Johannessen LE, Haugen KE, østvold AC, Stang E, Madshus IH (2001) Heterodimerization of the epidermal-growth-factor (EGF) receptor and ErbB2 and the affinity of EGF binding are regulated by different mechanisms. Biochem J 356:87–96
Söderberg O, Gullberg M, Jarvius M, Ridderstråle K, Leuchowius KJ et al (2006) Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat Methods 12:995–1000
Spears M, Taylor KJ, Munro AF, Cunningham CA, Mallon EA et al (2012) In situ detection of HER2:HER2 and HER2:HER3 protein-protein interactions demonstrates prognostic significance in early breast cancer. Breast Cancer Res 132:463–470
Shi Y, Huang W, Tan Y, Jin X, Dua R et al (2009) A novel proximity assay for the detection of proteins and protein complexes: quantitation of HER1 and HER2 total protein expression and homodimerization in formalin-fixed, paraffin-embedded cell lines and breast cancer tissue. Diagn Mol Pathol 18:11–21
Bazin H, Trinquet E, Mathis G (2002) Time resolved amplification of cryptate emission: a versatile technology to trace biomolecular interactions. J Biotechnol 82:233–250
Mathis G (1993) Rare earth cryptates and homogeneous fluoroimmunoassays with human sera. Clin Chem 39:1953–1959
Gaborit N, Larbouret C, Vallaghe J, Peyrusson F, Bascoul-Mollevi C et al (2011) Time-resolved fluorescence resonance energy transfer (TR-FRET) to analyze the disruption of EGFR/HER2 dimers: a new method to evaluate the efficiency of targeted therapy using monoclonal antibodies. J Biol Chem 286:11337–11345
Ho-Pun-Cheung A, Bazin H, Gaborit N, Larbouret C, Garnero P et al (2012) Quantification of HER expression and dimerization in patients' tumor samples using time-resolved Förster resonance energy transfer. PLoS One 7:e37065
Acknowledgments
This work was supported by ANR grant 07-RIB-003/Dim-HER.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Lopez-Crapez, E., Ho-Pun-Cheung, A., Garnero, P., Bazin, H. (2015). Evaluation of the Dimerization Profiles of HER Tyrosine Kinases by Time-Resolved Förster Resonance Energy Transfer (TR-FRET). In: Germano, S. (eds) Receptor Tyrosine Kinases. Methods in Molecular Biology, vol 1233. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1789-1_5
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1789-1_5
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1788-4
Online ISBN: 978-1-4939-1789-1
eBook Packages: Springer Protocols