Abstract
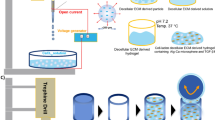
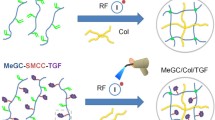
Decellularized tissues represent promising biomaterials, which harness the innate capacity of the tissue-specific extracellular matrix (ECM) to direct cell functions including stem cell proliferation and lineage-specific differentiation. However, bioscaffolds derived exclusively from decellularized ECM offer limited versatility in terms of tuning biomechanical properties, as well as cell–cell and cell–ECM interactions that are important mediators of the cellular response. As an alternative approach, in the current chapter we describe methods for incorporating cryo-milled decellularized tissues as a cell-instructive component within a hydrogel carrier designed to crosslink under mild conditions. This composite strategy can enable in situ cell encapsulation with high cell viability, allowing efficient seeding with a homogeneous distribution of cells and ECM. Detailed protocols are provided for the effective decellularization of human adipose tissue and porcine auricular cartilage, as well as the cryo-milling process used to generate the ECM particles. Further, we describe methods for synthesizing methacrylated chondroitin sulphate (MCS) and for performing UV-initiated and thermally induced crosslinking to form hydrogel carriers for adipose and cartilage regeneration. The hydrogel composites offer great flexibility, and the hydrogel phase, ECM source, particle size, cell type(s) and seeding density can be tuned to promote the desired cellular response. Overall, these systems represent promising platforms for the development of tissue-specific 3-D in vitro cell culture models and in vivo cell delivery systems.
Similar content being viewed by others
References
Frantz C, Stewart KM, Weaver VM (2010) The extracellular matrix at a glance. J Cell Sci 123:4195–4200. doi:10.1242/jcs.023820
Jabbari E, Leijten J, Xu Q, Khademhosseini A (2015) The matrix reloaded: the evolution of regenerative hydrogels. Mater Today 19:190–196. doi:10.1016/j.mattod.2015.10.005
Knight E, Przyborski S (2015) Advances in 3D cell culture technologies enabling tissue-like structures to be created in vitro. J Anat 227:746–756. doi:10.1111/joa.12257
Hinderer S, Layland SL, Schenke-Layland K (2016) ECM and ECM-like materials—biomaterials for applications in regenerative medicine and cancer therapy. Adv Drug Deliv Rev 97:260–269. doi:10.1016/j.addr.2015.11.019
Sart S, Tsai A-C, Li Y, Ma T (2014) Three-dimensional aggregates of mesenchymal stem cells: cellular mechanisms, biological properties, and applications. Tissue Eng Part B Rev 20:365–380. doi:10.1089/ten.TEB.2013.0537
Geckil H, Xu F, Zhang X et al (2010) Engineering hydrogels as extracellular matrix mimics. Nanomedicine 5:469–484. doi:10.2217/nnm.10.12
Benders KEM, van Weeren PR, Badylak SF et al (2013) Extracellular matrix scaffolds for cartilage and bone regeneration. Trends Biotechnol 31:169–176. doi:10.1016/j.tibtech.2012.12.004
Hoshiba T, Lu H, Kawazoe N, Chen G (2010) Decellularized matrices for tissue engineering. Expert Opin Biol Ther 10:1717–1728. doi:10.1517/14712598.2010.534079
Crapo PM, Gilbert TW, Badylak SF (2011) An overview of tissue and whole organ decellularization processes. Biomaterials 32:3233–3243. doi:10.1016/j.biomaterials.2011.01.057
Flynn LE (2010) The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials 31:4715–4724. doi:10.1016/j.biomaterials.2010.02.046
Huang S-P, Hsu C-C, Chang S-C et al (2012) Adipose-derived stem cells seeded on acellular dermal matrix grafts enhance wound healing in a murine model of a full-thickness defect. Ann Plast Surg 69:656–662. doi:10.1097/SAP.0b013e318273f909
Dainese L, Guarino A, Burba I et al (2012) Heart valve engineering: decellularized aortic homograft seeded with human cardiac stromal cells. J Heart Valve Dis 21:125–134
Woods T, Gratzer PF (2005) Effectiveness of three extraction techniques in the development of a decellularized bone–anterior cruciate ligament–bone graft. Biomaterials 26:7339–7349. doi:10.1016/j.biomaterials.2005.05.066
Han TTY, Toutounji S, Amsden BG, Flynn LE (2015) Adipose-derived stromal cells mediate in vivo adipogenesis, angiogenesis and inflammation in decellularized adipose tissue bioscaffolds. Biomaterials 72:125–137. doi:10.1016/j.biomaterials.2015.08.053
Turner AEB, Yu C, Bianco J et al (2012) The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells. Biomaterials 33:4490–4499. doi:10.1016/j.biomaterials.2012.03.026
Yu C, Bianco J, Brown C et al (2013) Porous decellularized adipose tissue foams for soft tissue regeneration. Biomaterials 34:3290–3302. doi:10.1016/j.biomaterials.2013.01.056
Ki H, Tian T, Han Y et al (2014) Biomaterials composite hydrogel scaffolds incorporating decellularized adipose tissue for soft tissue engineering with adipose-derived stem cells. Biomaterials 35:1914–1923. doi:10.1016/j.biomaterials.2013.11.067
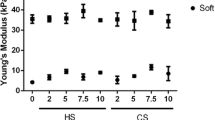
Brown CFC, Yan J, Han TTY et al (2015) Effect of decellularized adipose tissue particle size and cell density on adipose-derived stem cell proliferation and adipogenic differentiation in composite methacrylated chondroitin sulphate hydrogels. Biomed Mater 10:45010. doi:10.1088/1748-6041/10/4/045010
Heinbuch D (2016) Adipose-derived stem cell differentiation in cell aggregates supplemented with micronized, decellularized extracellular matrix. University of Western Ontario, London
Agmon G, Christman KL (2016) Controlling stem cell behavior with decellularized extracellular matrix scaffolds. Curr Opin Solid State Mater Sci 20:193–201. doi:10.1016/j.cossms.2016.02.001
Kang H, Peng J, Lu S et al (2014) In vivo cartilage repair using adipose-derived stem cell-loaded decellularized cartilage ECM scaffolds. J Tissue Eng Regen Med 8:442–453. doi:10.1002/term.1538
Quint C, Kondo Y, Manson RJ et al (2011) Decellularized tissue-engineered blood vessel as an arterial conduit. Proc Natl Acad Sci U S A 108:9214–9219. doi:10.1073/pnas.1019506108
Badylak SF, Freytes DO, Gilbert TW (2009) Extracellular matrix as a biological scaffold material: structure and function. Acta Biomater 5:1–13. doi:10.1016/j.actbio.2008.09.013
Tsai C-H, Chou M-Y, Jonas M et al (2002) A composite graft material containing bone particles and collagen in osteoinduction in mouse. J Biomed Mater Res 63:65–70
Mazzitelli S, Luca G, Mancuso F et al (2011) Production and characterization of engineered alginate-based microparticles containing ECM powder for cell/tissue engineering applications. Acta Biomater 7:1050–1062. doi:10.1016/j.actbio.2010.10.005
Chang C-H, Chen C-C, Liao C-H et al (2014) Human acellular cartilage matrix powders as a biological scaffold for cartilage tissue engineering with synovium-derived mesenchymal stem cells. J Biomed Mater Res Part A 102:2248–2257. doi:10.1002/jbm.a.34897
Beck EC, Barragan M, Libeer TB et al (2016) Chondroinduction from naturally derived cartilage matrix: a comparison between devitalized and decellularized cartilage encapsulated in hydrogel pastes. Tissue Eng Part A 22(7–8):665–679. doi:10.1089/ten.tea.2015.0546
Drury JL, Mooney DJ (2003) Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials 24:4337–4351. doi:10.1016/S0142-9612(03)00340-5
Cai S, Liu Y, Zheng Shu X, Prestwich GD (2005) Injectable glycosaminoglycan hydrogels for controlled release of human basic fibroblast growth factor. Biomaterials 26:6054–6067. doi:10.1016/j.biomaterials.2005.03.012
Lim HL, Hwang Y, Kar M, Varghese S (2014) Smart hydrogels as functional biomimetic systems. Biomater Sci 2:603. doi:10.1039/c3bm60288e
Spiller KL, Maher SA, Lowman AM (2011) Hydrogels for the repair of articular cartilage defects. Tissue Eng Part B Rev 17:281–299. doi:10.1089/ten.teb.2011.0077
Hayami JWS, Waldman SD, Amsden BG (2015) Photo-cross-linked methacrylated polysaccharide solution blends with high chondrocyte viability, minimal swelling, and moduli similar to load bearing soft tissues. Eur Polym J 72:1–11. doi:10.1016/j.eurpolymj.2015.01.038
Flynn LE, Prestwich GD, Semple JL, Woodhouse KA (2008) Proliferation and differentiation of adipose-derived stem cells on naturally derived scaffolds. Biomaterials 29:1862–1871. doi:10.1016/j.biomaterials.2007.12.028
Kong HJ, Smith MK, Mooney DJ (2003) Designing alginate hydrogels to maintain viability of immobilized cells. Biomaterials 24:4023–4029. doi:10.1016/S0142-9612(03)00295-3
Rouillard AD, Berglund CM, Lee JY et al (2011) Methods for photocrosslinking alginate hydrogel scaffolds with high cell viability. Tissue Eng Part C Methods 17:173–179. doi:10.1089/ten.tec.2009.0582
Koh W, Revzin A, Pishko MV (2002) Poly (ethylene glycol) hydrogel microstructures encapsulating living cells. Langmuir 18:2459–2462. doi:10.1021/la0115740
Thanh T, Thi H, Lee JS et al (2016) Enhanced cellular activity in gelatin-poly (ethylene glycol) hydrogels without compromising gel stiffness 16(3): 334–340
Wang D-A, Varghese S, Sharma B et al (2007) Multifunctional chondroitin sulphate for cartilage tissue-biomaterial integration. Nat Mater 6:385–392. doi:10.1038/nmat1890
Li Q, Williams CG, Sun DDN et al (2004) Photocrosslinkable polysaccharides based on chondroitin sulfate. J Biomed Mater Res 68A:28–33. doi:10.1002/jbm.a.20007
Goude MC, McDevitt TC, Temenoff JS (2014) Chondroitin sulfate microparticles modulate transforming growth factor-β1-induced chondrogenesis of human mesenchymal stem cell spheroids. Cells Tissues Organs 199:117–130. doi:10.1159/000365966
Varghese S, Hwang NS, Canver AC et al (2008) Chondroitin sulfate based niches for chondrogenic differentiation of mesenchymal stem cells. Matrix Biol 27:12–21. doi:10.1016/j.matbio.2007.07.002
Ravanat J-L, Douki T, Cadet J (2001) Direct and indirect effects of UV radiation on DNA and its components. J Photochem Photobiol B 63:88–102. doi:10.1016/S1011-1344(01)00206-8
Park H, Temenoff JS, Holland TA et al (2005) Delivery of TGF-β1 and chondrocytes via injectable, biodegradable hydrogels for cartilage tissue engineering applications. Biomaterials 26:7095–7103. doi:10.1016/j.biomaterials.2005.05.083
Park H, Temenoff JS, Tabata Y et al (2007) Injectable biodegradable hydrogel composites for rabbit marrow mesenchymal stem cell and growth factor delivery for cartilage tissue engineering. Biomaterials 28:3217–3227. doi:10.1016/j.biomaterials.2007.03.030
Temenoff JS, Park H, Jabbari E et al (2004) Thermally cross-linked oligo(poly(ethylene glycol) fumarate) hydrogels support osteogenic differentiation of encapsulated marrow stromal cells in vitro. Biomacromolecules 5:5–10. doi:10.1021/bm030067p
Gilbert TW, Sellaro TL, Badylak SF (2006) Decellularization of tissues and organs. Biomaterials 27:3675–3683. doi:10.1016/j.biomaterials.2006.02.014
Acknowledgements
Scholarship funding to support this work was provided by the NSERC CREATE CONNECT! Program and Western’s Collaborative Graduate Training Program in Musculoskeletal Health Research (CMHR). Operational grant funding support was provided by NSERC. The authors would like to thank Cody Brown for technical assistance with the SEM imaging and the Western Nanofabrication facility for access to their imaging equipment. Finally, we would like to acknowledge Drs. Aaron Grant, Brian Evans and Robert Richards for their clinical collaborations in providing human adipose tissue samples, as well as the Mount Brydges abattoir for providing the porcine auricular cartilage tissue.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media New York
About this protocol
Cite this protocol
Shridhar, A., Gillies, E., Amsden, B.G., Flynn, L.E. (2017). Composite Bioscaffolds Incorporating Decellularized ECM as a Cell-Instructive Component Within Hydrogels as In Vitro Models and Cell Delivery Systems. In: Turksen, K. (eds) Decellularized Scaffolds and Organogenesis. Methods in Molecular Biology, vol 1577. Humana Press, New York, NY. https://doi.org/10.1007/7651_2017_36
Download citation
DOI: https://doi.org/10.1007/7651_2017_36
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7655-3
Online ISBN: 978-1-4939-7656-0
eBook Packages: Springer Protocols