Summary
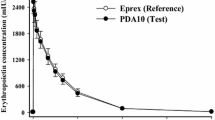
Epoetin (recombinant human erythropoietin, EPO) is of proven benefit in the treatment of renal anaemia, and preliminary reports suggest that it may have a role in the management of other anaemic conditions. Pharmacokinetic and therapeutic studies have examined the use of epoetin administered intravenously, intraperitoneally and subcutaneously, and there is accumulating evidence that the last route has several advantages. After intravenous administration, epoetin is distributed in a volume comparable to the plasma volume, and plasma concentrations decay monoexponentially with a half-life of between 4 and 12 hours. Administration of epoetin in peritoneal dialysis fluid results in detectable concentrations in the bloodstream after 1 to 2 hours, and peak concentrations of the order of 2 to 4% of those obtained with the same intravenous dose are found after approximately 12 hours. The bioavailability of epoetin administered intraperitoneally in dialysis fluid is about 3 to 8%, but this may be increased by injecting the drug into a dry peritoneal cavity. Subcutaneous administration results in peak concentrations at about 18 hours which are 5 to 10% of those found after the same intravenous dose. The bioavailability of subcutaneous epoetin is about 20 to 30%, and detectable serum concentrations are still present 4 days after administration, in contrast to intravenous administration after which concentrations have returned to baseline within 2 to 3 days.
Remarkably little is known about the metabolic fate of either erythropoietin or epoetin. In addition, there is much controversy surrounding the relative roles of the kidney and liver in the catabolism of epoetin. About 3 to 10% of epoetin is excreted unchanged in the urine. In common with other glycoproteins, the carbohydrate residues which constitute 40% of its molecular size are essential for maintaining the stability of epoetin in circulation. Desialated epoetin, although biologically active in vitro, is cleared very rapidly from plasma with resultant loss of activity. Further work is required, however, in identifying the pathways of metabolism and elimination of this glycoprotein hormone.
Similar content being viewed by others
References
Abbrecht PH, Litteil JK. Plasma erythropoietin in men and mice during acclimatisation to different altitudes. Journal of Applied Physiology 32: 54–58, 1972
Al-khatti A, Veith RW, Papayannopoulou T, Fritsch EF, Gold-wasser E. et al. Stimulation of fetal hemoglobin synthesis by erythropoietin in baboons. New England Journal of Medicine 317: 415–420, 1987
Anagnostou A. Erythropoietin: a hematopoietic hormone produced by the kidney. Seminars in Nephrology 5: 104–114, 1985
Annable L, Cotes PM, Müssen MV. The second international reference preparation of erythropoietin, human, urinary, for bioassay. Bulletin Organization Monde Santé 47: 99–112, 1972
Bargman JM, Breborowicz A, Rodela H, Sombolos K, Oreopoulos DG. Intraperitoneal administration of recombinant human erythropoietin in uremic animals. Peritoneal Dialysis International 8: 249–252, 1988
Birgegard G, Hallgren R, Caro J. Serum erythropoietin in rheumatoid arthritis and other inflammatory arthritides: relationship to anaemia and the effect of anti-inflammatory treatment. British Journal of Haematology 65: 479–483, 1987
Birgegard G, Miller O, Caro J, Erslev AJ. Serum erythropoietin levels by radioimmunoassay in polycythaemia. Scandinavian Journal of Haematology 29: 161–167, 1982
Boelaert JR, Schurgers ML, Matthys EG, Belpaire FM, Daneels RF, et al. Comparative pharmacokinetics of recombinant erythropoietin administered by the intravenous, subcutaneous and intraperitoneal routes in continuous ambulatory peritoneal dialysis patients. Peritoneal Dialysis International 9: 95–98, 1989
Bommer J, Alexiou U, Muller-Buhl E, Eifert J, Ritz E. Recombinant human erythropoietin therapy in haemodialysis patients — dose determination and clinical experience. Nephrology, Dialysis, Transplantation 2: 238–242, 1987
Bommer J, Ritz E, Weinreich T, Bommer G, Ziegler T. Subcutaneous erythropoietin. Lancet 2: 406, 1988
Caro J, Brown S, Miller O, Murray T, Erslev AJ, Erythropoietin levels in uremic nephric and anephric patients. Journal of Laboratory and Clinical Medicine 93: 449–458, 1979
Caro J, Erslev AJ, Erythropoietin assays and their use in the study of anemias. Contributions to Nephrology 66: 54–62, 1988
Casati S, Passerini P, Campise MR, Graziani G, Cesana B, et al. Benefits and risks of protracted treatment with human recombinant erythropoietin in patients having haemodialysis. British Medical Journal 295: 1017–1020, 1987
Cotes PM, Pippard MJ, Reid CDL, Winearls CG, Oliver DO, et al. Characterization of the anaemia of chronic renal failure and the mode of ils correction by a preparation of human erythropoietin (r-Hu EPO): an investigation of the pharmacokinetics of intravenous erythropoietin and its effects on erythro-kinetics. Quarterly Journal of Medicine 262: 113–137, 1989
Danielson BG. Pharmacokinetics of intravenous and subcutaneous human recombinant erythropoietin in healthy volunteers Abstract. 2nd Investigators’ Meeting on R-hu EPO, Vienna, p. 3. 1988
Davis JM, Arakawa T, Strickland TW, Yphantis DA. Characterization of recombinant human erythropoietin produced in Chinese hamster ovary cells. Biochemistry 26: 2633–2638, 1987
Dinkelaar RB, Engels EY, Hart AAM, Schoemaker LP, Bosch E. et al. Metabolic studies on erythropoietin (Ep): II. The role of liver and kidney in the metabolism of Ep. Experimental Haematology 9: 796–803, 1981
Dunn CDR, Lange RD. Erythropoietin: assay and characterization: In Roath (Ed.) Topical reviews in hematology, pp. 1–32. Wright, Bristol. 1980
Egrie JC, Browne JK, Lai P, Lin FK. Characterization and biological effects of recombinant human erythropoietin. Immu-nobiology 172: 213–224, 1986
Egrie JC, Eschbach JW, McGuire T, Adamson JW. Pharmacokinetics of recombinant human erythropoietin administered to hemodialysis patients. Kidney International 33: 262. 1988
Elliott S, Delorme E, Talbot C, Chang D, Hesterberg L. Characterization of anti-erythropoietin monoclonal antibodies. Blood 74(Suppl): 1228. 1989
Emanucle RM, Fareed J, The effect of molecular weight on the bioavailability of heparin. Thrombosis Research 48: 591–596, 1987
Emmanouel DS, Fang VS, Katz AI. Prolactin metabolism in the rat: role of the kidney in degradation of the hormone. American Journal of Physiology 240: F437–F445. 1981
Emmanouel DS, Goldwasser E, Katz AI. Metabolism of pure human erythropoietin in the rat. American Journal of Physiology 247: F168–FI76. 1984
Emmanouel DS, Jaspan JB, Rubenstein AH, Huen AH-J, Fink E, et al. Glucagon metabolism in the rat Contribution of the kidney to the metabolic clearance rate of the hormone. Journal of Clinical Investigation 62: 6–13, 1978
Eschbach JW, Egrie JC, Downing MR, Browne JK, Adamson JW. Correction of the anemia of end-stage renal disease with recombinant human erythropoietin. New England Journal of Medicine 316: 73–78, 1987
Eschbach JW, Kelly MR, Haley NR, Abels RI, Adamson JW. Treatment of the anemia of progressive renal failure with recombinant human erythropoietin. New England Journal of Medicine 321: 158–163, 1989
Eschbach JW, Mladenovic J, Garcia JF, Wahl PW, Adamson JW. The anemia of chronic renal failure in sheep. Response to erythropoietin-rich plasma in vivo Journal of Clinical Investigation 74: 434–441, 1984
Faulds D, Sorkm EM. Epoetin (Recombinant Human Erythropoietin): A review of us pharmacodynamic and pharmacokinetic properties and therapeutic potential in anaemia and the stimulation of erythropoiesis. Drugs 38: 863–899, 1989
Fischer S. Roheim PS Role of liver in the inactivation of erythropoietin. Nature 200: 899–900, 1963
Fisher JW. Extrarenal erythropoietin production. Journal of Laboratory and Clinical Medicine 93: 695–699, 1979
Frenken LAM, Coppens PJW, Tiggeler RGWL. Koene RAP In-traperitoneal erythropoietin. Lancet 2: 1495. 1988
Fried W. The liver as a source of extrarenal erythropoietin pro-duction. Blood 40: 671–677, 1972
Fu J-S, Lertora JJL, Brookins J, Rice JC, Fisher JW. Pharmacokinetics of erythropoietin in intact and anephric dogs. Journal of Laboratory and Clinical Medicine 111: 669–676, 1988
Fukuda MN, Sasaki H, Lopez L, Fukuda M. Survival of recombinant erythropoietin in the circulation: the role of carbohydrates. Blood 73: 84–89, 1989
Gahl GM, Passlick J, Pustelnik A, Kampf D, Grabensee B, et al. Intraperitoneal versus intravenous recombinant human erythropoietin in stable CAPD patients. 26th Congress of the European Dialysis and Transplant Association — European Renal Association. Gothenburg. Abstract, p. 199, 1989
Galloway JA, Spradlin CT, Nelson RL, Wentworth SM, Davidson JA. el al. Factors influencing ihe absorption, serum insulin concentration, and blood glucose responses after injeclions of regular insulin and various insulin mixtures. Diabetes Care 4: 366–376, 1981
Galpin J, Thompkins J, Abels R, Turpen TJ. A study of the safely and efficacy of recombinant human erythropoietin in AIDS patients with anaemia induced by their disease and zidovudine therapy. Vth International Conference on AIDS, Montreal. MBP 328. 1989
Garcia J, Sherwood J, Goldwasser E. Radioimmunoassay of erythropoietin. Blood Cells 5: 405–419, 1979
George WJ, Briggs DW, Rodgers GM, Fisher JW. Metabolism of erythropoietin. In Fisher (Ed.) Kidney Hormones. Vol. II: Erythropoietin, p. 73. Academic Press Inc., London/New York. 1977
Goldwasser E, Kung CK-H. Progress in the purification of erythropoietin. Annals of the New York Academy of Sciences 149: 49–53, 1968
Goodnough LT, Rudnick S, Price TH. Increased preoperative collection of autologous blood with recombinant human erythropoietin therapy. New England Journal of Medicine 321: 1163–1168, 1989
Granolleras C, Branger B, Beau MC, Deschodt G, Alsabadani B. et al. Experience with daily self-administered subcutaneous erythropoietin. Contributions to Nephrology 76: 143–148, 1989
Hughes RT, Cotes PM, Oliver DO, Pippard MJ, Royston P. et al. Correction of the anaemia of chronic renal failure with erythropoietin: pharmacokinetic studies in patients on haemo-dialysis and CAPD. Contributions to Nephrology 76: 122–130, 1989
Jacobs K, Shoemaker C, Rudcrsdorf R, Neill SD, Kaufman RJ. et al. Isolation and characterization of genomic and cDNA clones of human erythropoietin. Nature 313: 806–810, 1985
Jacobson LO, Goldwasser E, Fried W, Plzak LF. The role of the kidney in erythropoiesis. Nature 179: 633–634, 1957
Jelkmann W. Temporal pattern of erythropoietin titres in kidney tissue during hypoxic hypoxia. Pflugers Archiv 393: 88–91, 1982
Kampf D, Kahl A, Passlick J, Pustelnik A, Eckardt K-U. et al. Single-dose kinetics of recombinant human erythropoietin after intravenous, subcutaneous and intraperitoneal administration. Contributions to Nephrology 76: 106–111, 1989
Katz AI, Rubenstein AH, Metabolism of proinsulin, insulin and C-peptide in the rat. Journal of Clinical Investigation 52: 11 13–1 121. 1973
Kazal LA, Erslev AJ. The measurement of erythropoietin. Annals of Clinical and Laboratory Science 5: 91–97, 1975
Kindlcr J, Eckardt K-U. Ehmer B, Jandeleit K, Kurtz A. et al. Single-dose pharmacokinetics of recombinant human erythropoietin in patients with various degrees of renal failure. Nephrology. Dialysis, Transplantation 4: 345–349, 1989
Koury ST, Bondurant MC, Koury MJ. Localisation of erythropoietin synthesizing cells in murine kidneys by in situ hybridisation. Blood 71: 524–527, 1988
Kromer G, Solf A, Ehmer B, Kaufmann B, Quellhorst E. Single dose pharmacokinetics of recombinant human erythropoietin comparing intravenous, subcutaneous and intraperitoneal administration in 1PD patients. Kidney International 37: 331, 1990
KukraI J, Carney AI, Ebroon E. The role of the liver and surgical stress. Surgical Forum 19: 348–349, 1968
Kurtz A, Eckardt K-U. Tannahill L, Bauer C. Regulation of erythropoietin production. Contributions to Nephrology 66: 1–16, 1988
Lacombe C, DaSilva JL, Bruneval P, Fournier JG, Wendung F, et al. Peritubular cells are the site of erythropoietin synthesis in the murine hypoxic kidney. Journal of Clinical Investigation 81: 620–623, 1988
Levine EA, Rosen AL, Gould SA. Recombinant human erythropoietin and autologous blood donation. Surgery 104: 365–369, 1988
Lim VS, DeGowin RL, Zavala D, Kirchner PT, Abels R, et al. Recombinant human erythropoietin treatment in pre-dialysis patients: a double-blind placebo-controlled trial. Annals of Internal Medicine 110: 108–114, 1989
Lin FK, Suggs S, Lin CH, Browne JK, Smalling R, et al. Cloning and expression of the human erythropoietin gene. Proceedings of the National Academy of Sciences of the United Stales of America 82: 7580–7585, 1985
Macdougall IC, Cavill I, Davies ME, Hutton RD, Coles GA, et al. Subcutaneous recombinant erythropoietin in the treatment of renal anaemia in CAPD patients. Contributions to Nephrology 76: 219–226, 1989a
Macdougall IC, Roberts DE, Coles GA, Williams JD. Intraperitoneal erythropoietin. Lancet 1: 1389, 1989b
Macdougall IC, Roberts DE, Neubert P, Dharmasena AD, Coles GA, et al. Pharmacokinetics of intravenous, intraperitoneal, and subcutaneous recombinant erythropoietin in patients on CAPD. Contributions to Nephrology 76: 112–121, 1989c
Macdougall IC, Roberts DE, Neubert P, Dharmasena AD, Coles GA, et al. Pharmacokinetics of recombinant human erythropoietin in patients on continuous ambulatory peritoneal dialysis. Lancet 2: 425–427, 1989d
Maxwell AP, Douglas JF, Afrasiabi M, Lappin TRJ, Bridges JM. Erythropoietin pharmacokinetics and red cell metabolism in haemodialysis patients. Nephrology, Dialysis. Transplantation 4: 476, 1989a
Maxwell AP, Lappin TRJ, Bridges JM, Johnston CF, McGeown MG. Renal tubular cell production of erythropoietin co-localised by immunohistochemistry and in situ hybridisation. Nephrology. Dialysis, Transplantation 4: 420, 1989b
Means RT, Olsen NJ, Krantz SB, Dessypris EN, Graber SE, et al. Treatment of the anemia of rheumatoid arthritis with recombinant human erythropoietin: clinical and in vitro studies. Arthritis and Rheumatism 32: 638–642, 1989
Milledge JS, Cotes PM. Serum erythropoietin in humans at high altitude and its relation to plasma renin. Journal of Applied Physiology 59: 360–364, 1985
Miyake T, Kung CKH, Goldwasser E. Purification of human erythropoietin. Journal of Biological Chemistry 252: 5558–5564. 1977
Mladenovic J, Eschbach JW, Koup JR, Garcia JF, Adamson JW. Erythropoietin kinetics in normal and uremic sheep. Journal of Laboratory and Clinical Medicine 105: 659–663, 1985
Naets JP, Wittek M. A role of the kidney in the catabolism of erythropoietin in the rat. Journal of Laboratory and Clinical Medicine 84: 99–106, 1974
Neumayer H-H, Brockmoller J, Fritschka E, Roots I, Scigalla P. et al. Pharmacokinetics of recombinant human erythropoietin after SC administration and in long-term IV treatment in patients on maintenance hemodialysis. Contributions to Nephrology 76: 131–142, 1989
Nielsen OJ, Egfjord M, Hirth P. Erythropoietin metabolism in the isolated perfused rat liver. Contributions to Nephrology 76: 90–97, 1989
Paulsen EP, Courtney JW, Duckworth WC. Insulin resistance caused by massive degradation of subcutaneous insulin. Diabetes 28: 640–645, 1979
Recny MA, Scoble HA, Kim Y. Structural characterisation of natural human urinary and recombinant DNA-derivcd erythropoietin (identification of des-Arginine 166 erythropoietin). Journal of Biological Chemistry 262: 17156–17163, 1987
Reissmann KR, Diederich KA, Kenjiro I, Schmaus JW. Influence of disappearance rate and distribution space on plasma concentration of erythropoietin in normal rats. Journal of Laboratory and Clinical Medicine 65: 967–975, 1965
Rhondeau SM, Christensen RD, Ross MP, Rothstein G, Simmons MA. Responsiveness to recombinant human erythropoietin of marrow erythroid progenitors from infants with the “anemia of prematurity”. Journal of Pediatrics 112: 935–940, 1988
Roh BL, Paulo G, Fisher JW. Metabolism of erythropoietin by isolated perfused livers of dogs treated with SKF 525-A. American Journal of Physiology 223: 1345–1348, 1972a
Roh BL, Paulo LG, Thompson J, Fisher JW. Plasma disappearance of 125I-labelled erythropoietin in anesthetised rabbits. Proceedings of the Society for Experimental Biology and Medicine 141: 268–270, 1972b
Rosse WF, Waldmann TA. The metabolism of erythropoietin in patients with anemia due to deficient erythropoiesis. Journal of Clinical Investigation 43: 1348–1354, 1964
Salmonson T, Danielson BG, Wikstrom B. Pharmacokinetics and pharmacodynamics of recombinant erythropoietin after SC and IV administration. 26th Congress of the European Dialysis and Transplant Association — European Renal Association. Gothenburg. Abstract, p. 210, 1989
Sasaki H, Bothner B, Dell A, Fukuda M. Carbohydrate structure of erythropoietin expressed in Chinese hamster ovary cells by a human erythropoietin cDNA. Journal of Biological Chemistry 262; 12059–12076. 1987
Sawyer ST, Krantz SB, Goldwasser E. Binding and receptor-mediated endocytosis of erythropoietin in Friend virus-infected erythroid cells. Journal of Biological Chemistry 262: 5554–5562, 1987
Scigalla P, Hoelk G, Pahlke W. Pharmacokinetics of recombinant erythropoietin in normal and uraemic rats. Nephrology, Dialysis. Transplantation 2: 389, 1987
Shannon KM, Naylor GS, Torkildson JC, Circulating erythroid progenitors in the anemia of prematurity. New England Journal of Medicine 317: 728–733, 1987
Sherwood JB, Carmichael LD, Goldwasser E. The heterogeneity of circulating human serum erythropoietin. Endocrinology 122: 1472–1477, 1988
Sinai-Trieman L, Salusky IB, Fine RN. Use of subcutaneous recombinant human erythropoietin in children undergoing continuous cycling peritoneal dialysis. Journal of Pediatrics 114: 550–554, 1989
Spivak JL, Hogans BB. The in vivo metabolism of recombinant human erythropoietin in the rat. Blood 73: 90–99, 1989
Steinhauer HB, Lubrich-Birkner I, Dreyling KW, Horl WH, Schollmeyer P. Increased ultrafiltration after erythropoietin-induced correction of renal anemia in patients on continuous ambulatory peritoneal dialysis. Nephron 53: 87–88, 1989
Stevens JM, Strong CA, Oliver DO, Winearls CG, Cotes PM. Subcutaneous erythropoietin and peritoneal dialysis. Lancet 1: 1388–1389, 1989
Walker RE, Parker RI, Kovacs JA. Anemia and erythropoiesis in patients with the acquired immunodeficiency syndrome and Kaposi’s sarcoma treated with zidovudine. Annals of Internal Medicine 108: 372–376, 1988
Weintraub AH, Gordon AS, Becker EL, Camiscoli JF, Contrera JF. Plasma and renal clearance of exogenous erythropoietin in the dog. American Journal of Physiology 207: 523–529, 1964
Wikstrom B, Salmonson T, Grahnen A, Danielson BG. Phar-macokineiics of recombinant human erythropoietin in haemo dialysis patients. Nephrology, Dialysis, Transplantation 3: 503 1988
Wilton P, Widlund L, Guilbaud. Bioequivalence of genotropit and somatonorm. Acta Paediatrica Scandinavica 337 (Suppl.) 118–121, 1987
Winearls CG, Oliver DO, Pippard MJ, Reid C, Downing MR, et al. Effect of human erythropoietin derived from recombinan DNA on the anaemia of patients maintained by chronii haemodialysis. Lancet 2: 1175–1178, 1986
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Macdougall, I.C., Roberts, D.E., Coles, G.A. et al. Clinical Pharmacokinetics of Epoetin (Recombinant Human Erythropoietin). Clin Pharmacokinet 20, 99–113 (1991). https://doi.org/10.2165/00003088-199120020-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199120020-00002