Abstract
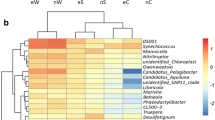
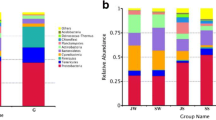
Increasing evidence has revealed a close association between intestinal bacterial communities and human health. However, given that host phylogeny shapes the composition of intestinal microbiota, it is unclear whether changes in intestinal microbiota structure in relation to shrimp health status. In this study, we collected shrimp and seawater samples from ponds with healthy and diseased shrimps to understand variations in bacterial communities among habitats (water and intestine) and/or health status. The bacterial communities were clustered according to the original habitat and health status. Habitat and health status constrained 14.6 and 7.7 % of the variation in bacterial communities, respectively. Changes in shrimp intestinal bacterial communities occurred in parallel with changes in disease severity, reflecting the transition from a healthy to a diseased state. This pattern was further evidenced by 38 bacterial families that were significantly different in abundance between healthy and diseased shrimps; moderate changes were observed in shrimps with sub-optimal health. In addition, within a given bacterial family, the patterns of enrichment or decrease were consistent with the known functions of those bacteria. Furthermore, the identified 119 indicator taxa exhibited a discriminative pattern similar to the variation in the community as a whole. Overall, this study suggests that changes in intestinal bacterial communities are closely associated with the severity of shrimp disease and that indicator taxa can be used to evaluate shrimp health status.




Similar content being viewed by others
References
Anderson MJ (2001) A new method for non‐parametric multivariate analysis of variance. Aust Ecol 26(1):32–46
Austin B (2006) The bacterial microflora of fish, revised. Sci World J 6:931–945
Balcázar JL, Blas ID, Ruiz-Zarzuela I, Cunningham D, Vendrell D, Múzquiz JL (2006) The role of probiotics in aquaculture. Vet Microbiol 114(3):173–186
Bates ST, Cropsey GW, Caporaso JG, Knight R, Fierer N (2011) Bacterial communities associated with the lichen symbiosis. Appl Environ Microbiol 77(4):1309–1314
Beardsley C, Moss S, Malfatti F, Azam F (2011) Quantitative role of shrimp fecal bacteria in organic matter fluxes in a recirculating shrimp aquaculture system. FEMS Microbiol Ecol 77(1):134–145
Berry D, Reinisch W (2013) Intestinal microbiota: a source of novel biomarkers in inflammatory bowel diseases? Best Pract Res Clin Gastroenterol 27(1):47–58
Berry D, Schwab C, Milinovich G, Reichert J, Mahfoudh KB, Decker T, Engel M, Hai B, Hainzl E, Heider S (2012) Phylotype-level 16S rRNA analysis reveals new bacterial indicators of health state in acute murine colitis. ISME J 6(11):2091–2106
Boutin S, Bernatchez L, Audet C, Derôme N (2013) Network analysis highlights complex interactions between pathogen, host and commensal microbiota. PLoS One 8(12):e84772
Caporaso JG, Bittinger K, Bushman FD, DeSantis TZ, Andersen GL, Knight R (2010a) PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatics 26(2):266–267
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI (2010b) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336
Chaiyapechara S, Rungrassamee W, Suriyachay I, Kuncharin Y, Klanchui A, Karoonuthaisiri N, Jiravanichpaisal P (2012) Bacterial community associated with the intestinal tract of P. monodon in commercial farms. Microb Ecol 63(4):938–953
Churchill GA (2004) Using ANOVA to analyze microarray data. Biotechniques 37:173–175
Clarke KR (1993) Non‐parametric multivariate analyses of changes in community structure. Aust J Ecol 18(1):117–143
Clemente JC, Ursell LK, Parfrey LW, Knight R (2012) The impact of the gut microbiota on human health: an integrative view. Cell 148(6):1258–1270
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67(3):345–366
Eaton AD, Franson MAH (2005) Standard methods for the examination of water & wastewate. American Public Health Association, Water Environment Federation
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27(16):2194–2200
FAO (2010) The state of world fisheries and aquaculture. FAO Rome, Italy
Farzanfar A (2006) The use of probiotics in shrimp aquaculture. FEMS Immunol Med Mic 48(2):149–158
Frank DN, Amand ALS, Feldman RA, Boedeker EC, Harpaz N, Pace NR (2007) Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci U S A 104(34):13780–13785
Goldfarb KC, Karaoz U, Hanson CA, Santee CA, Bradford MA, Treseder KK, Wallenstein MD, Brodie EL (2011) Differential growth responses of soil bacterial taxa to carbon substrates of varying chemical recalcitrance. Front Microbiol 2:194
Guarner F, Malagelada JR (2003) Gut flora in health and disease. Lancet 361(9356):512–519
Jia W, Li H, Zhao L, Nicholson JK (2008) Gut microbiota: a potential new territory for drug targeting. Nat Rev Drug Discov 7(2):123–129
Leser TD, Mølbak L (2009) Better living through microbial action: the benefits of the mammalian gastrointestinal microbiota on the host. Environ Microbiol 11(9):2194–2206
Leung TL, Bates AE (2013) More rapid and severe disease outbreaks for aquaculture at the tropics: implications for food security. J Appl Ecol 50(1):215–222
Logares R, Lindström ES, Langenheder S, Logue JB, Paterson H, Laybourn-Parry J, Rengefors K, Tranvik L, Bertilsson S (2013) Biogeography of bacterial communities exposed to progressive long-term environmental change. ISME J 7(5):937–948
Luo Y, Hui D, Zhang D (2006) Elevated CO2 stimulates net accumulations of carbon and nitrogen in land ecosystems: a meta-analysis. Ecology 87(1):53–63
Mack DR, Michail S, Wei S, McDougall L, Hollingsworth MA (1999) Probiotics inhibit enteropathogenic E. coli adherence in vitro by inducing intestinal mucin gene expression. Am J Physiol-Gastr L 276(4):G941–G950
Nerstedt A, Nilsson EC, Ohlson K, Håkansson J, Thomas Svensson L, Löwenadler B, Svensson UK, Mahlapuu M (2007) Administration of Lactobacillus evokes coordinated changes in the intestinal expression profile of genes regulating energy homeostasis and immune phenotype in mice. Brit J Nutr 97(06):1117–1127
Pandit SN, Kolasa J, Cottenie K (2009) Contrasts between habitat generalists and specialists: an empirical extension to the basic metacommunity framework. Ecology 90(8):2253–2262
Roeselers G, Mittge EK, Stephens WZ, Parichy DM, Cavanaugh CM, Guillemin K, Rawls JF (2011) Evidence for a core gut microbiota in the zebrafish. ISME J 5(10):1595–1608
Round JL, Mazmanian SK (2009) The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol 9(5):313–323
Rungrassamee W, Klanchui A, Maibunkaew S, Chaiyapechara S, Jiravanichpaisal P, Karoonuthaisiri N (2014) Characterization of intestinal bacteria in wild and domesticated adult black tiger shrimp (Penaeus monodon). PLoS One 9(3):e91853
Schryver PD, Vadstein O (2014) Ecological theory as a foundation to control pathogenic invasion in aquaculture. ISME J 8(12):2360–2368
Snieszko S (1974) The effects of environmental stress on outbreaks of infectious diseases of fishes. J Fish Biol 6(2):197–208
Sullam KE, Essinger SD, Lozupone CA, O’Connor MP, Rosen GL, Knight R, Kilham SS, Russell JA (2012) Environmental and ecological factors that shape the gut bacterial communities of fish: a meta‐analysis. Mol Ecol 21(13):3363–3378
Sung H, Hsu S, Chen C, Ting Y, Chao W (2001) Relationships between disease outbreak in cultured tiger shrimp (Penaeus monodon) and the composition of Vibrio communities in pond water and shrimp hepatopancreas during cultivation. Aquaculture 192(2):101–110
Team RC (2005) R: A language and environment for statistical computing. R foundation for Statistical Computing
Ter Braak CJ (1986) Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67(5):1167–1179
Tseng I, Chen JC (2004) The immune response of white shrimp Litopenaeus vannamei and its susceptibility to Vibrio alginolyticus under nitrite stress. Fish Shellfish Immunol 17(4):325–333
Vaseeharan B, Ramasamy P (2003) Abundance of potentially pathogenic microorganisms in (Penaeus monodon) larvae rearing systems in India. Microbiol Res 158(4):299–308
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73(16):5261–5267
Xiong J, Ye X, Wang K, Chen H, Hu C, Zhu J, Zhang D (2014a) The biogeography of the sediment bacterial community responds to a nitrogen pollution gradient in the East China Sea. Appl Environ Microbiol 80(6):1919–1925
Xiong J, Zhu J, Wang K, Wang X, Ye X, Liu L, Zhao Q, Hou M, Qiuqian L, Zhang D (2014b) The temporal scaling of bacterioplankton composition: high turnover and predictability during shrimp cultivation. Microb Ecol 67(2):256–264
Xiong J, Zhu J, Zhang D (2014c) The application of bacterial indicator phylotypes to predict shrimp health status. Appl Microbiol Biotechnol 98:8291–8299
Xiong J, He Z, Shi S, Kent A, Deng Y, Wu L, Van Nostrand JD, Zhou J (2015) Elevated CO2 shifts the functional structure and metabolic potentials of soil microbial communities in a C4 agroecosystem. Sci Rep 5:9316
Zhang D, Wang X, Xiong J, Zhu J, Wang Y, Zhao Q, Chen H, Guo A, Wu J, Dai H (2014) Bacterioplankton assemblages as biological indicators of shrimp health status. Ecol Indic 38:218–224
Zhou H, Li D, Tam N, Jiang H, Sheng H, Qin J, Liu X, Zou F (2011) BIPES, a cost-effective highthroughput method foe seeessing microbial diversity. ISME J 5:741–749
Zhou J, Fang W, Yang X, Zhou S, Hu L, Li X, Qi X, Su H, Xie L (2012) A nonluminescent and highly virulent Vibrio harveyi strain is associated with “bacterial white tail disease” of Litopenaeus vannamei shrimp. PLoS One 7(2):e29961
Acknowledgments
This work was financially supported by the National High Technology Research and Development Program of China (863 Program, 2012AA092000), the Natural Science Foundation of Ningbo City (2013A610169), the Natural Science Foundation (XYL14004), the Experimental Technology Research and Development Project (SYJS201405), and the KC Wong Magna Fund of Ningbo University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 450 kb)
Rights and permissions
About this article
Cite this article
Xiong, J., Wang, K., Wu, J. et al. Changes in intestinal bacterial communities are closely associated with shrimp disease severity. Appl Microbiol Biotechnol 99, 6911–6919 (2015). https://doi.org/10.1007/s00253-015-6632-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6632-z