Abstract
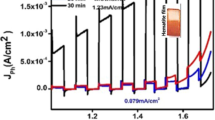
Photoelectrochemical (PEC) water splitting is a promising approach to producing H2 and O2. Hematite (α-Fe2O3) is considered one of the most promising photoelectrodes for PEC water splitting, due to its good photochemical stability, non-toxicity, abundance in earth, and suitable bandgap (Eg∼2.1 eV). However, the PEC water splitting efficiency of hematite is severely hampered by its short hole diffusion length (2–4 nm), poor conductivity, and ultrafast recombination of photogenerated carriers (about 10 ps). Here, we show a novel and effective method for significantly improving the PEC water splitting performance of hematite by Au ion implantation and the following high-temperature annealing process. Based on a series of characterizations and analyses, we have found Fe2+ species and tightly attached Au particles were produced at Au-implanted hematite. As a result, the charge separation and charge injection efficiency of Au-implanted Fe2O3 are markedly increased. The photocurrent density of optimized Au-implanted Fe2O3 could reach 1.16 mA cm−2 at 1.5 V vs. RHE which was nearly 300 times higher than that of the pristine Fe2O3 (4 μA cm−2). Furthermore, the Au-implanted Fe2O3 photoelectrode exhibited great stability for the 8-hour PEC water splitting test without photocurrent decay.
摘要
光电化学催化分解水是一种极具发展前景的生产H2和O2的方法. α-Fe2O3由于其具有优良的光电化学稳定性、 无毒害、 地球储量大以及合适的能带宽度Eg∼2.1 eV等优点, 被认为是最有潜力的光电催化分解水的材料之一. 然而由于其空穴传输距离短(约2∼4 nm)、 导电性差、 光生载流子复合速度极快等原因, α-Fe2O3的光电催化分解水的效率受到了极大限制. 在此, 我们报道了一种新颖并且高效的离子注 入Au元素并且退火的方法显著提高了α-Fe2O3光电化学分解水的效率. 根据一系列的表征和分析, 发现Fe2+和紧密接触的金颗粒在金注入的α-Fe2O3样品中产生. 因此, 金注入的α-Fe2O3样品的载流子分离效率和注入效率得到了显著提高. 该样品光电流密度在1.5 V vs. RHE偏压下可以达到1.16 mA cm−2, 相对原始样品光电流密度提升接近300倍(4 μA cm−2). 此外, 金注入的α-Fe2O3样品在8 h的光电催化分解水的测试中没有出现光电流衰减的现象, 表现了良好的稳定性.
Similar content being viewed by others
References
Wheeler DA, Wang G, Ling Y, et al. Nanostructured hematite: synthesis, characterization, charge carrier dynamics, and photoelectrochemical properties. Energy Environ Sci, 2012, 5: 6682–6702
Fujishima A, Honda K. Electrochemical photolysis of water at a semiconductor electrode. Nature, 1972, 238: 37–38
Yang C, Wang Z, Lin T, et al. Core-shell nanostructured “black” rutile titania as excellent catalyst for hydrogen production enhanced by sulfur doping. J Am Chem Soc, 2013, 135: 17831–17838
Warren SC, Voïtchovsky K, Dotan H, et al. Identifying champion nanostructures for solar water-splitting. Nat Mater, 2013, 12: 842–849
Liu J, Liu Y, Liu N, et al. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science, 2015, 347: 970–974
Qiu Y, Liu W, Chen W, et al. Efficient solar-driven water splitting by nanocone BiVO4-perovskite tandem cells. Sci Adv, 2016, 2: e1501764
Morales-Guio CG, Tilley SD, Vrubel H, et al. Hydrogen evolution from a copper (I) oxide photocathode coated with an amorphous molybdenum sulphide catalyst. Nat Commun, 2014, 5: 3059
Wu H, Ren F, Xing Z, et al. Cathodic shift of onset potential for water oxidation of WO3 photoanode by Zr+ ions implantation. J Appl Phys, 2017, 121: 085305
Li Z, Luo W, Zhang M, et al. Photoelectrochemical cells for solar hydrogen production: current state of promising photoelectrodes, methods to improve their properties, and outlook. Energy Environ Sci, 2013, 6: 347–370
Han T, Chen Y, Tian G, et al. Hydrogenated TiO2/SrTiO3 porous microspheres with tunable band structure for solar-light photocatalytic H2 and O2 evolution. Sci China Mater, 2016, 59: 1003–1016
Liu H, Ma H, Joo J, et al. Contribution of multiple reflections to light utilization efficiency of submicron hollow TiO2 photocatalyst. Sci China Mater, 2016, 59: 1017–1026
Pozun ZD, Henkelman G. Hybrid density functional theory band structure engineering in hematite. J Chem Phys, 2011, 134: 224706
Rozenberg GK, Dubrovinsky LS, Pasternak MP, et al. High-pressure structural studies of hematite Fe2O3. Phys Rev B, 2002, 65: 064112
Glasscock JA, Barnes PRF, Plumb IC, et al. Enhancement of photoelectrochemical hydrogen production from hematite thin films by the introduction of Ti and Si. J Phys Chem C, 2007, 111: 16477–16488
Aroutiounian VM, Arakelyan VM, Shahnazaryan GE, et al. Photoelectrochemistry of tin-doped iron oxide electrodes. Sol Energy, 2007, 81: 1369–1376
Annamalai A, Shinde PS, Jeon TH, et al. Fabrication of superior a-Fe2O3 nanorod photoanodes through ex-situ Sn-doping for solar water splitting. Sol Energy Mater Sol Cells, 2016, 144: 247–255
Ling Y, Wang G, Wheeler DA, et al. Sn-doped hematite nanostructures for photoelectrochemical water splitting. Nano Lett, 2011, 11: 2119–2125
Abel AJ, Patel AM, Smolin SY, et al. Enhanced photoelectrochemical water splitting via SILAR-deposited Ti-doped hematite thin films with an FeOOH overlayer. J Mater Chem A, 2016, 4: 6495–6504
Kay A, Cesar I, Grätzel M. New benchmark for water photooxidation by nanostructured a-Fe2O3 Films. J Am Chem Soc, 2006, 128: 15714–15721
Li M, Yang Y, Ling Y, et al. Morphology and doping engineering of Sn-doped hematite nanowire photoanodes. Nano Lett, 2017, 17: 2490–2495
Ling Y, Wang G, Reddy J, et al. The influence of oxygen content on the thermal activation of hematite nanowires. Angew Chem, 2012, 124: 4150–4155
Forster M, Potter RJ, Ling Y, et al. Oxygen deficient a-Fe2O3 photoelectrodes: a balance between enhanced electrical properties and trap-mediated losses. Chem Sci, 2015, 6: 4009–4016
Wang G, Ling Y, Li Y. Oxygen-deficient metal oxide nanostructures for photoelectrochemical water oxidation and other applications. Nanoscale, 2012, 4: 6682–6691
Ling Y, Wang G, Wang H, et al. Low-temperature activation of hematite nanowires for photoelectrochemical water oxidation. ChemSusChem, 2014, 7: 848–853
Li M, Deng J, Pu A, et al. Hydrogen-treated hematite nanostructures with low onset potential for highly efficient solar water oxidation. J Mater Chem A, 2014, 2: 6727–6733
Kiejna A, Pabisiak T. Surface properties of clean and Au or Pd covered hematite (a-Fe2O3) (0001). J Phys-Condens Matter, 2012, 24: 095003
Warwick MEA, Barreca D, Bontempi E, et al. Pt-functionalized Fe2O3 photoanodes for solar water splitting: the role of hematite nano-organization and the platinum redox state. Phys Chem Chem Phys, 2015, 17: 12899–12907
Liu W, Liu Z, Wang G, et al. Carbon coated Au/TiO2 mesoporous microspheres: a novel selective photocatalyst. Sci China Mater, 2017, 60: 438–448
Dearnaley G. Ion implantation. Nature, 1975, 256: 701–705
Wang G, Xiao X, Li W, et al. Significantly enhanced visible light photoelectrochemical activity in TiO2 nanowire arrays by nitrogen implantation. Nano Lett, 2015, 15: 4692–4698
Vayssieres L, Beermann N, Lindquist SE, et al. Controlled aqueous chemical growth of oriented three-dimensional crystalline nanorod arrays: application to iron (III) oxides. Chem Mater, 2001, 13: 233–235
Liu Y, Ren F, Cai G, et al. Energy dependence on formation of TiO2 nanofilms by Ti ion implantation and annealing. Mater Res Bull, 2014, 51: 376–380
Zheng X, Shen S, Ren F, et al. Irradiation-induced TiO2 nanorods for photoelectrochemical hydrogen production. Int J Hydrogen Energy, 2015, 40: 5034–5041
Kim JY, Magesh G, Youn DH, et al. Single-crystalline, wormlike hematite photoanodes for efficient solar water splitting. Sci Rep, 2013, 3: 2681
de Faria DLA, Venâncio Silva S, de Oliveira MT. Raman microspectroscopy of some iron oxides and oxyhydroxides. J Raman Spectrosc, 1997, 28: 873–878
Ding SY, Yi J, Li JF, et al. Nanostructure-based plasmon-enhanced Raman spectroscopy for surface analysis of materials. Nat Rev Mater, 2016, 1: 16021
Malviya KD, Dotan H, Shlenkevich D, et al. Systematic comparison of different dopants in thin film hematite (a-Fe2O3) photoanodes for solar water splitting. J Mater Chem A, 2016, 4: 3091–3099
Sander D, Schmidthals C, Enders A, et al. Stress and structure of Ni monolayers on W(110): the importance of lattice mismatch. Phys Rev B, 1998, 57: 1406–1409
Li W, Sheehan SW, He D, et al. Hematite-based solar water splitting in acidic solutions: functionalization by mono-and multilayers of iridium oxygen-evolution catalysts. Angew Chem, 2015, 127: 11590–11594
Fujii T, de Groot FMF, Sawatzky GA, et al. In situ XPS analysis of various iron oxide films grown by NO2-assisted molecular-beam epitaxy. Phys Rev B, 1999, 59: 3195–3202
Morrish R, Rahman M, MacElroy JMD, et al. Activation of hematite nanorod arrays for photoelectrochemical water splitting. ChemSusChem, 2011, 4: 474–479
Xiao R, Pelenovich VO, Fu D. Spin cycloid destruction in Pr doped BiFeO3 films studied by conversion-electron Mossbauer spectroscopy. Appl Phys Lett, 2013, 103: 012901
Klingelhöfer G, Morris RV, Bernhardt B, et al. Jarosite and hematite at meridiani planum from opportunity’s mossbauer spectrometer. Science, 2004, 306: 1740–1745
Zheng X, Ren F, Zhang S, et al. A general method for large-scale fabrication of semiconducting oxides with high SERS sensitivity. ACS Appl Mater Interfaces, 2017, 9: 14534–14544
Pu A, Deng J, Li M, et al. Coupling Ti-doping and oxygen vacancies in hematite nanostructures for solar water oxidation with high efficiency. J Mater Chem A, 2014, 2: 2491–2497
Turner NH, Single AM. Determination of peak positions and areas from wide-scan XPS spectra. Surf Interface Anal, 1990, 15: 215–222
Wang H, Turner JA. Characterization of hematite thin films for photoelectrochemical water splitting in a dual photoelectrode device. J Electrochem Soc, 2010, 157: F173
Dotan H, Sivula K, Grätzel M, et al. Probing the photoelectrochemical properties of hematite (a-Fe2O3) electrodes using hydrogen peroxide as a hole scavenger. Energy Environ Sci, 2011, 4: 958–964
Xi L, Chiam SY, Mak WF, et al. A novel strategy for surface treatment on hematite photoanode for efficient water oxidation. Chem Sci, 2013, 4: 164–169
Acknowledgements
This work was supported by the National Natural Science Foundation of China (51371131, 11375134, 51571153 and 11722543), and the Fundamental Research Funds for the Central Universities (2042017kf0168).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dong He obtained his bachelaor degree from Hubei University in 2015. He is a PhD candidate of Wuhan University under the supervision of Profs. Xiangheng Xiao and Changzhong Jiang. His research interests are mainly focused on photoelectrochemical water splitting and electrocatalytic water splitting.
Xiangheng Xiao received his BS degree and PhD degree from Wuhan University. He worked as a visiting scholar at the Department of Chemistry and Biochemistry, University of California, Los Angeles, from 2013 to 2014. Currently, he has been a full professor in the Department of Physics, Wuhan University, since 2015. The research fields are mainly focused on photoelectrochemical water splitting, surface-enhanced spectroscopy, nanophotonics and ion beam modification of nanoscale material devices. For details please see the lab website: http://physics.whu.edu.cn/xiaoxiangheng.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
He, D., Song, X., Ke, Z. et al. Construct Fe2+ species and Au particles for significantly enhanced photoelectrochemical performance of α-Fe2O3 by ion implantation. Sci. China Mater. 61, 878–886 (2018). https://doi.org/10.1007/s40843-017-9155-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-017-9155-9