Abstract
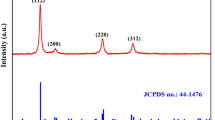
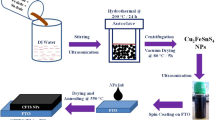
The novel chalcostibite CuSbS2 had gained unique attention due to their dynamic nature as less toxic, cost-effective and earth abundant materials for the synthesis of an absorber layer in solar cell application. Herein, a facile and effective solvothermal method was used to enhance the sphere-like grain growth in the presence of polyvinylpyrrolidone (PVP) along with other precursor’s, followed by the deposition of CuSbS2 thin films using drop casting method. The synthesized nanoparticles and the deposited films were characterized for their structural, morphological, optical and electrical properties using different characterization techniques. X-ray diffraction (XRD) and Raman analysis revealed that as the amount of PVP increased, the crystallinity improved and the impurity phase formation reduced. High-resolution transmission electron microscope (HRTEM) with reduced crystallite size in the range of 2–5 nm and field emission scanning electron microscope (FESEM), exhibited sphere-shaped grains indicating the effect of PVP as surfactant for the growth of CuSbS2 nanomaterials. The average elemental composition of the nanoparticles had been determined using EDX analysis, and the result yielded Cu rich in all the samples. Optical studies using UV–Vis-NIR diffuse reflectance spectroscopy revealed that obtained CuSbS2 nanoparticles were having the absorption in the entire visible region and the direct band gap energy was in the range of 1.25 eV to 1.53 eV and that of photoluminescence spectrum gave the emission in the near IR region. The hall measurement studies showed that the deposited CuSbS2 films exhibited p-type conductivity. Devices were fabricated with the configuration of FTO/n-TiO2/p-CuSbS2/Ag, and the electrical properties were studied by recording the current- voltage (I-V) characteristics of the heterojunction device structures.








Similar content being viewed by others
References
R B Balow, E J Sheets, M M Abu-Omar, and R Agrawal Chem. Mater. 27 2290 (2015)
S Suehiro, K Horita, M Yuasa, T Tanaka, K Fujita, Y Ishiwata, K Shimanoe, and T Kida Inorg. Chem. 54 7840 (2015)
L Shi, Y Li, C Wu, and Y Dai J. Alloys Compd. 648 507 (2015)
B Shu and Q Han 13 46 (2016)
A Wei, Z Yan, Y Zhao, M Zhuang, and J Liu Int. J. Hydrogen Energy 40 797 (2015)
M X Zhuang, A X Wei, Y Zhao, J Liu, Z Q Yan, and Z Liu Int. J. Hydrogen Energy 40 806 (2015)
S M McLeod, C J Hages, N J Carter, and R Agrawal Prog. Photovoltaics Res. Appl. 23 1550 (2015)
S Ahn, S Rehan, D G Moon, Y-J Eo, S Ahn, J H Yun, A Cho, and J Gwak Green Chem. 19 1268 (2017)
C K Miskin, W-C Yang, C J Hages, N J Carter, C S Joglekar, E A Stach, and R Agrawal Prog. Photovoltaics Res. Appl. 23 654 (2015)
C Wadia, A P Alivisatos and D M Kammen, Environ. Sci. Technol. 43 2072 (2009)
D B Mitzi, O Gunawan, T K Todorov, K Wang, and S Guha Sol. Energy Mater. Sol. Cells 95 1421 (2011)
S Chen, X G Gong, A Walsh, and S H Wei Appl. Phys. Lett. 96 4 (2010)
K Biswas, S Lany, and A Zunger Appl. Phys. Lett. 96 94 (2010)
Y Cao, M S Denny, J V Caspar, W E Farneth, Q Guo, A S Ionkin, L K Johnson, M Lu, I Malajovich, D Radu, D H Rosenfeld, K R Choudhury, and W Wu J. Am. Chem. Soc. 134 15644 (2012)
J Bincy, G Genifer Silvena, and A Leo Rajesh Phys. B Condens. Matter 537 243 (2018)
C L McCarthy, P Cottingham, K Abuyen, E C Schueller, S P Culver, and R L Brutchey J. Mater. Chem. C 4 6230 (2016)
K Ramasamy, H Sims, W H Butler, and A Gupta J. Am. Chem. Soc. 136 1587 (2014)
S Ikeda, S Sogawa, Y Tokai, W Septina, T Harada, and M Matsumura RSC Adv. 4 40969 (2014)
Z Zhang, C Zhou, Y Liu, J Li, Y Lai, and M Jia Int. J. Electrochem. Sci. 8 10059 (2013)
Y Zou and J Jiang Mater. Lett. 123 66 (2014)
Y Rodríguez-Lazcano, M T S Nair, and P K Nair J. Cryst. Growth 223 399 (2001)
J A Ramos Aquino, D L Rodriguez Vela, S Shaji, D A Avellaneda, and B Krishnan Phys. Status Solidi Curr. Top. Solid State Phys. 13 24 (2016)
A C Rastogi and N R Janardhana Thin Solid Films 565 285 (2014)
A Rabhi, M Kanzari, and B Rezig Mater. Lett. 62 3576 (2008)
D Colombara, L M Peter, K D Rogers, J D Painter, and S Roncallo Thin Solid Films 519 7438 (2011)
S Banu, S J Ahn, S K Ahn, K Yoon, and A Cho Sol. Energy Mater. Sol. Cells 151 14 (2016)
Z Liu, J Huang, J Han, T Hong, J Zhang, and Z Liu Phys. Chem. Chem. Phys. 18 16615 (2016)
K M Koczkur, S Mourdikoudis, L Polavarapu, and S E Skrabalak Dalt. Trans. 44 17883 (2015)
S V. Jadhav, D S Nikam, V M Khot, N D Thorat, M R Phadatare, R S Ningthoujam, A B Salunkhe, and S H Pawar New J. Chem. 37 3121 (2013)
G Lu, S Li, Z Guo, O K Farha, B G Hauser, X Qi, Y Wang, X Wang, S Han, X Liu, J S Duchene, H Zhang, Q Zhang, X Chen, J Ma, S C J Loo, W D Wei, Y Yang, J T Hupp, et al. Nat. Chem. 4 310 (2012)
D Liu, L Lin, S Ren, and S Fu J. Mater. Sci. 51 3111 (2016)
M Liu, Y Gong, Z Li, M Dou, and F Wang Appl. Surf. Sci. 387 790 (2016)
C Yan, Z Su, E Gu, T Cao, J Yang, J Liu, F Liu, Y Lai, J Li, and Y Liu RSC Adv. 2 10481 (2012)
T Rath, A J Maclachlan, M D Brown, and S A Haque J. Mater. Chem. A Mater. Energy Sustain. 3 24155 (2015)
K Ramasamy, R K Gupta, H Sims, S Palchoudhury, S Ivanov, and A Gupta J. Mater. Chem. A 3 13263 (2015)
J Bincy, G Genifer Silvena, and A Leo Rajesh, AIP Conf. Proc. 1832 50007 (2017)
H C Gupta, M K Singh, and L M Tiwari J. Phys. Chem. Solids 64 531 (2003)
X Y Ye, Y M Zhou, Y Q Sun, J Chen, and Z Q Wang J. Nanoparticle Res. 11 1159 (2009)
C An, K Tang, Q Yang, and Y Qian Inorg. Chem. 42 8081 (2003)
S Thiruvenkadam and A Leo Rajesh Int. J. Sci. Technol. Res. 3 38 (2014)
S Thiruvenkadam and A Leo Rajesh Ijser.Org 5 248 (2014)
J Bincy, G Genifer Silvena, and A Leo Rajesh Mater. Res. Bull. 95 267 (2017)
F Long, S Chi, J He, J Wang, X Wu, S Mo, and Z Zou J. Solid State Chem. 229 228 (2015)
S Moosakhani, A A Sabbagh Alvani, R Mohammadpour, P M Hannula, Y Ge, and S P Hannula Mater. Lett. 215 157 (2018)
K Takei, T Maeda, and T Wada Thin Solid Films 582 263 (2015)
A G Kannan, T E Manjulavalli, and J Chandrasekaran Procedia Eng. 141 15 (2016)
F E Loranca-Ramos, C J Diliegros-Godines, R Silva González, and M Pal Appl. Surf. Sci. 427 1099 (2018)
S A Manolache, L Andronic, A Duta, and A Enesca J. Optoelectron. Adv. Mater. 9 1269 (2007)
Acknowledgements
One of the authors Ms. Bincy John thanks the University Grants Commission of India, for providing research fellowship (Maulana Azad National Fellowship, Grant No: F1-17.1/2016-17/MANF-2015-17-KER-53161). The authors would like to thank Dr. G. Amarendra, Scientist-In-Charge, and Dr. G. M. Bhalerao, Scientist-E UGC-DAE Consortium for Scientific Research, Kalpakkam, Tamilnadu, India, for providing sophisticated instrumentation facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
John, B., Genifer Silvena, G., Hussain, S. et al. Surfactant-mediated solvothermal synthesis of CuSbS2 nanoparticles as p-type absorber material. Indian J Phys 93, 185–195 (2019). https://doi.org/10.1007/s12648-018-1288-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12648-018-1288-z
Keywords
- CuSbS2 nanoparticles
- Solvothermal method
- PVP surfactant
- Absorber layer
- Heterojunction
- Solar energy materials