Abstract
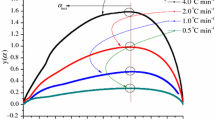
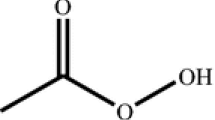
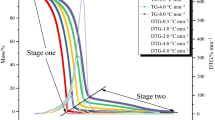
Tert-butyl peroxybenzoate (TBPB) is one of the sensitive and hazardous chemicals which have been popularly employed in petrifaction industries in the past. This study attempted to elucidate its unsafe characteristics and thermally sensitive structure so as to help prevent runaway reactions, fires or explosions in the process environment. We employed differential scanning calorimetry (DSC) to assess the kinetic parameters (such as exothermic onset temperature (T 0), heat of reaction (ΔH), frequency factor (A)), and the other safety parameters using four different scanning rates (1, 2, 4 and 10°C min−1) combined with curve-fitting method.
The results indicated that TBPB becomes very dangerous during decomposition reactions; the onset temperature and reaction heat were about 100°C and 1300 J g−1, respectively. Through this study, TBPB accidents could be reduced to an accepted level with safety parameters under control. According to the findings in the study and the concept of inherent safety, TBPB runaway reactions could be thoroughly prevented in the relevant plants.
Similar content being viewed by others
References
Material safety data sheet, Akzo Nobel Chemicals bv, Stationsplein 4, P.O. Box 247, 3800 AE Amersfoort, The Netherlands 2006.
P. H. Bolton and D. R. Kearns, J. Phys. Chem., 78 (1974) 1896.
R. Baron, A. Darchen and D. Hauchard, Electrochim. Acta, 51 (2006) 1336.
Safety and handling of organic peroxides: a guide, organic peroxide producers safety division, The Society of the Plastics Industry (SPI), Inc., Washington, DC, USA 1999.
Recommendations on the transportation of dangerous goods. Manual of test and criteria, 3rd Ed., United Nations (UN), New York, USA and Geneva, Switzerland 1999.
D. Swern, Organic peroxides, John Wiley and Sons, Inc., 1972.
V. Logvinenko, K. Mikhailov and Yu. Yukhin, J. Therm. Anal. Cal., 88 (2007) 47.
A. Ioitescu, G. Vlase, T. Vlase and N. Doca, J. Therm. Anal. Cal., 88 (2007) 121.
J. M. Tseng, C. M. Shu, J. J. Horng, C. M. Kuan and H. I. Hsu, Process Saf. Environ. Prot., 85 (2007) 125.
D. I. Townsend and J. C. Tou, Thermochim. Acta, 37 (1980) 1.
H. G. Fisher and D. D. Goetz, J. Loss Prev. Process Ind., 6 (1993) 183.
R. Andreozzi, V. Caprio, S. Crescitelli and G. Russo, J. Hazard. Mater., 17 (1988) 305.
R. H. Chang, J. M. Tseng, J. M. Jehng, C. M. Shu and H. Y. Hou, J. Therm. Anal. Cal., 83 (2006) 57.
J. M. Tseng, C. M. Shu and Y. C. Yu, Korean J. Chem. Eng., 22 (2005) 797.
P. C. Bowes, Self-heating: evaluating and controlling the hazards, Elsevier Science Publishing Company, Inc., New York, NY 10017, USA 1984.
A. A. Kossoy, Inherently safer and assessment of reaction hazards technology workshop, Yunlin, Taiwan, ROC 2002.
H. G. Fisher and D. D. Goetz, J. Loss Prev. Process Ind., 4 (1991) 305.
A. A. Kossoy, A. Benin and Y. Akhmetshin, J. Hazard. Mater., 118 (2005) 9.
A. A. Kossoy and Y. Akhmetshin, Process Saf. Prog., 26 (2007) 209.
STARe software with solaris operating system, Operating instructions; Mettler Toledo, Sweden 2004.
R. H. Chang, Master Thesis, NYUST, Yunlin, Taiwan, ROC 2003.
H. Y. Hou, T. S. Liao, Y. S. Duh and C. M. Shu, J. Therm. Anal. Cal., 83 (2006) 167.
B. D. Leila and H. Fierz, J. Hazard. Mater., 93 (2002) 137.
M. H. Yuan, C. M. Shu and A. A. Kossoy, Thermochim. Acta, 430 (2005) 67.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheng, S.Y., Tseng, J.M., Lin, S.Y. et al. Runaway reaction on tert-butyl peroxybenzoate by DSC tests. J Therm Anal Calorim 93, 121–126 (2008). https://doi.org/10.1007/s10973-007-8831-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8831-z