Abstract
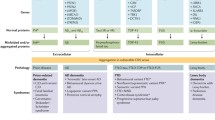
Dementia represents one of the most diffuse disorders of our Era. Alzheimer’s disease is the principle cause of dementia worldwide. Metabolic, infectious, autoimmune, inflammatory, and genetic dementias represent a not negligible number of disorders, with increasing numbers in younger subjects. Due to the heterogeneity of patients and disorders, the diagnosis of dementia is challenging. In the present article, we propose a practical diagnostic approach following the two-step investigation procedure. The first step includes basic blood tests and brain neuroimaging, performed on all patients. After this first-line investigation, it is then possible to rule out metabolic causes of dementia and to identify three main subgroups in dementia: predominant gray matter atrophy, white matter disease, basal ganglia pathologies. The predominant gray matter atrophy subgroup includes neurodegenerative causes of dementia and some lysosomal storage disorders. The white matter subgroup indicates a comprehensive list of vascular dementia causes, mitochondrial diseases, and leukodystrophies. Whereas, the basal ganglia alterations are due to metal accumulation pathologies, such as iron, copper, or calcium. Each category has specific clinical hallmarks, accurately reported in the article, and requires specific second-line investigation. Thus, we indicate the distinct second diagnostic step of each disease. The proposed diagnostic flow-chart follows the clinical reasoning and helps clinicians through the differential diagnosis of dementia.


Similar content being viewed by others
References
Mayeux R, Stern Y (2012) Epidemiology of Alzheimer disease. Cold Spring Harb Perspect Med 2(8)
Levine DA (2013) Young-onset dementia unanswered questions and unmet needs. JAMA Intern Med 173(17):1619–1620. https://doi.org/10.1001/jamainternmed.2013.8090
Kelley BJ, Boeve BF, Josephs KA (2008) Young-onset dementia: demographic and etiologic characteristics of 235 patients. Arch Neurol 65(11):1502–1508. https://doi.org/10.1001/archneur.65.11.1502
Tong T, Ledig C, Guerrero R, Schuh A, Koikkalainen J, Tolonen A, Rhodius H, Barkhof F, Tijms B, Lemstra AW, Soininen H, Remes AM, Waldemar G, Hasselbalch S, Mecocci P, Baroni M, Lötjönen J, Flier WV, Rueckert D (2017) Five-class differential diagnostics of neurodegenerative diseases using random undersampling boosting. Neuroimage Clin 15:613–624. https://doi.org/10.1016/j.nicl.2017.06.012
Rossor MN, Fox NC, Mummery CJ, Schott JM, Warren JD (2010) The diagnosis of young-onset dementia. Lancet Neurol 9(8):793–806. https://doi.org/10.1016/S1474-4422(10)70159-9
Sorbi S, Hort J, Erkinjuntti T, Fladby T, Gainotti G, Gurvit H, Nacmias B, Pasquier F, Popescu BO, Rektorova I, Religa D, Rusina R, Rossor M, Schmidt R, Stefanova E, Warren JD, Scheltens P, EFNS Scientist Panel on Dementia and Cognitive Neurology (2012) EFNS-ENS Guidelines on the diagnosis and management of disorders associated with dementia. Eur J Neurol 19(9):1159–1179
Hort J, O'Brien JT, Gainotti G, Pirttila T, Popescu BO, Rektorova I, Sorbi S, Scheltens P, EFNS Scientist Panel on Dementia (2010) EFNS guidelines for the diagnosis and management of Alzheimer’s disease. Eur J Neurol 17(10):1236–1248
Sampson EL, Warren JD, Rossor MN (2004) Young onset dementia. Postgrad Med J 80(941):125–139. https://doi.org/10.1136/pgmj.2003.011171
Snowden JS, Thompson JC, Stopford CL, Richardson AM, Gerhard A, Neary D, Mann DM (2011) The clinical diagnosis of early-onset dementias: diagnostic accuracy and clinicopathological relationships. Brain 134(Pt 9):2478–2492. https://doi.org/10.1093/brain/awr189
Power C, Selnes OA, Grim JA, McArthur JC (1995) HIV dementia scale: a rapid screening test. J Acquir Immune Defic Syndr Hum Retrovirol 8(3):273–278. https://doi.org/10.1097/00042560-199503010-00008
Titulaer MJ, McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, Honig LS, Benseler SM, Kawachi I, Martinez-Hernandez E, Aguilar E, Gresa-Arribas N, Ryan-Florance N, Torrents A, Saiz A, Rosenfeld MR, Balice-Gordon R, Graus F, Dalmau J (2013) Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol 12(2):157–165. https://doi.org/10.1016/S1474-4422(12)70310-1
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, Klunk WE, Koroshetz WJ, Manly JJ, Mayeux R, Mohs RC, Morris JC, Rossor MN, Scheltens P, Carrillo MC, Thies B, Weintraub S, Phelps CH (2011) The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7(3):263–269. https://doi.org/10.1016/j.jalz.2011.03.005
Lehmann M, Ghosh PM, Madison C, Laforce R Jr, Corbetta-Rastelli C, Weiner MW, Greicius MD, Seeley WW, Gorno-Tempini ML, Rosen HJ, Miller BL, Jagust WJ, Rabinovici GD (2013) Diverging patterns of amyloid deposition and hypometabolism in clinicalvariants of probable Alzheimer’s disease. Brain 136(Pt 3):844–858. https://doi.org/10.1093/brain/aws327
Albert MS, DeKosky ST, Dickson D, Dubois B, Feldman HH, Fox NC, Gamst A, Holtzman DM, Jagust WJ, Petersen RC, Snyder PJ, Carrillo MC, Thies B, Phelps CH (2011) The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Associationworkgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7(3):270–279
Morris E, Chalkidou A, Hammers A, Peacock J, Summers J, Keevil S (2016) Diagnostic accuracy of (18)F amyloid PET tracers for the diagnosis of Alzheimer's disease: a systematic review and meta-analysis. Eur J Nucl Med Mol Imaging 43(2):374–385
Höglund K, Fourier A, Perret-Liaudet A, Zetterberg H, Blennow K, Portelius E (2015) Alzheimer’s disease—recent biomarker developments in relation to updated diagnostic criteria. Clin Chim Acta 449:3–8. https://doi.org/10.1016/j.cca.2015.01.041
Rascovsky K, Hodges JR, Knopman D, Mendez MF, Kramer JH, Neuhaus J, van Swieten JC, Seelaar H, Dopper EG, Onyike CU, Hillis AE, Josephs KA, Boeve BF, Kertesz A, Seeley WW, Rankin KP, Johnson JK, Gorno-Tempini ML, Rosen H, Prioleau-Latham CE, Lee A, Kipps CM, Lillo P, Piguet O, Rohrer JD, Rossor MN, Warren JD, Fox NC, Galasko D, Salmon DP, Black SE, Mesulam M, Weintraub S, Dickerson BC, Diehl-Schmid J, Pasquier F, Deramecourt V, Lebert F, Pijnenburg Y, Chow TW, Manes F, Grafman J, Cappa SF, Freedman M, Grossman M, Miller BL (2011) Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain 134(9):2456–2477. https://doi.org/10.1093/brain/awr179
Gorno-Tempini ML, Hillis AE, Weintraub S, Kertesz A, Mendez M, Cappa SF, Ogar JM, Rohrer JD, Black S, Boeve BF, Manes F, Dronkers NF, Vandenberghe R, Rascovsky K, Patterson K, Miller BL, Knopman DS, Hodges JR, Mesulam MM, Grossman M (2011) Classification of primary progressive aphasia and its variants. Neurology 76(11):1006–1014. https://doi.org/10.1212/WNL.0b013e31821103e6
Kipps CM, Hodges JR, Fryer TD, Nestor PJ (2009) Combined magnetic resonance imaging and positron emission tomography brain imaging in behavioural variant frontotemporal degeneration: refining the clinical phenotype. Brain 132(Pt9):2566–2578. https://doi.org/10.1093/brain/awp077
Rohrer JD, Warren JD, Modat M (2009) Patterns of cortical thinning in the language variants of frontotemporal lobar degeneration. Neurology 72(18):1562–1569. https://doi.org/10.1212/WNL.0b013e3181a4124e
Borroni B, Padovani (2013) A new algorithm for molecular diagnostics in FTLD. Nat Rev Neurol 9(5):241–242. https://doi.org/10.1038/nrneurol.2013.72
McKeith IG, Boeve BF, Dickson DW, Halliday G, Taylor JP, Weintraub D, Aarsland D, Galvin J, Attems J, Ballard CG, Bayston A, Beach TG, Blanc F, Bohnen N, Bonanni L, Bras J, Brundin P, Burn D, Chen-Plotkin A, Duda JE, El-Agnaf O, Feldman H, Ferman TJ, Ffytche D, Fujishiro H, Galasko D, Goldman JG, Gomperts SN, Graff-Radford NR, Honig LS, Iranzo A, Kantarci K, Kaufer D, Kukull W, Lee VMY, Leverenz JB, Lewis S, Lippa C, Lunde A, Masellis M, Masliah E, McLean P, Mollenhauer B, Montine TJ, Moreno E, Mori E, Murray M, O'Brien JT, Orimo S, Postuma RB, Ramaswamy S, Ross OA, Salmon DP, Singleton A, Taylor A, Thomas A, Tiraboschi P, Toledo JB, Trojanowski JQ, Tsuang D, Walker Z, Yamada M, Kosaka K (2017) Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB Consortium. Neurology 89(1):88–100. https://doi.org/10.1212/WNL.0000000000004058
Armstrong MJ, Litvan I, Lang AE, Bak TH, Bhatia KP, Borroni B, Boxer AL, Dickson DW, Grossman M, Hallett M, Josephs KA, Kertesz A, Lee SE, Miller BL, Reich SG, Riley DE, Tolosa E, Tröster AI, Vidailhet M, Weiner WJ (2013) Criteria for the diagnosis of corticobasal degeneration. Neurology 80(5):496–503. https://doi.org/10.1212/WNL.0b013e31827f0fd1
Erkkinen MG, Kim MO, Geschwind MD (2017) Clinical Neurology and Epidemiology of the Major Neurodegenerative Diseases. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a033118
Höglinger GU, Respondek G, Stamelou M, Kurz C, Josephs KA, Lang AE, Mollenhauer B, Müller U, Nilsson C, Whitwell JL, Arzberger T, Englund E, Gelpi E, Giese A, Irwin DJ, Meissner WG, Pantelyat A, Rajput A, van Swieten JC, Troakes C, Antonini A, Bhatia KP, Bordelon Y, Compta Y, Corvol JC, Colosimo C, Dickson DW, Dodel R, Ferguson L, Grossman M, Kassubek J, Krismer F, Levin J, Lorenzl S, Morris HR, Nestor P, Oertel WH, Poewe W, Rabinovici G, Rowe JB, Schellenberg GD, Seppi K, van Eimeren T, Wenning GK, Boxer AL, Golbe LI, Litvan I, Movement Disorder Society-endorsed PSP Study Group (2017) Clinical diagnosis of progressive supranuclear palsy: The movement disorder society criteria. Mov Disord 32(6):853–864
Myers RH, Sax DS, Schoenfeld M, Bird ED, Wolf PA, Vonsattel JP, White RF, Martin JB (1985) Late onset of Huntington’s disease. J Neurol Neurosurg Psychiatry 48(6):530–534. https://doi.org/10.1136/jnnp.48.6.530
Seneca S, Fagnart D, Keymolen K, Lissens W, Hasaerts D, Debulpaep S, Desprechins B, Liebaers I, De Meirleir L (2004) Early onset Huntington disease: a neuronal degeneration syndrome. Eur J Pediatr 163(12):717–721. https://doi.org/10.1007/s00431-004-1537-3
Mondal B, Paul P, Paul M, Kumar H (2013) An update on spino-cerebellar ataxias. Ann Indian Acad Neurol 16(3):295–303. https://doi.org/10.4103/0972-2327.116896
Rossi M, Perez-Lloret S, Doldan L, Cerquetti D, Balej J, Millar Vernetti P, Hawkes H, Cammarota A, Merello M (2014) Autosomal dominant cerebellar ataxias: a systematic review of clinical features. Eur J Neurol 21(4):607–615. https://doi.org/10.1111/ene.12350
Murphy S, Gorman G, Beetz C, Byrne P, Dytko M, McMonagle P, Kinsella K, Farrell M, Hutchinson M (2009) Dementia in SPG4 hereditary spastic paraplegia: clinical, genetic, and neuropathologic evidence. Neurology 73(5):378–384. https://doi.org/10.1212/WNL.0b013e3181b04c6c
De Antonio M, Dogan C, Hamroun D (2016) Unravelling the myotonic dystrophy type 1 clinical spectrum: a systematic registry-based study with implications for disease classification. Rev Neurol 172(10):572–580. https://doi.org/10.1016/j.neurol.2016.08.003
Stirnemann J, Belmatoug N, Camou F, Serratrice C, Froissart R, Caillaud C, Levade T, Astudillo L, Serratrice J, Brassier A, Rose C, Billette de Villemeur T, Berger MG (2017) A review of Gaucher disease pathophysiology, clinical presentation and treatments. Int J Mol Sci 18(2). https://doi.org/10.3390/ijms18020441
Vanier MT (2010) Niemann-pick disease type C. Orphanet J Rare Dis 5(1):16. https://doi.org/10.1186/1750-1172-5-16
Neudorfer O, Pastores GM, Zeng BJ, Gianutsos J, Zaroff CM, Kolodny EH (2005) Late-onset Tay-Sachs disease: phenotypic characterization and genotypic correlations in 21 affected patients. Genet Med 7(2):119–123. https://doi.org/10.1097/01.GIM.0000154300.84107.75
Berkovic SF, Staropoli JF, Carpenter S, Oliver KL, Kmoch S, Anderson GW, Damiano JA, Hildebrand MS, Sims KB, Cotman SL, Bahlo M, Smith KR, Cadieux-Dion M, Cossette P, Jedličková I, Přistoupilová A, Mole SE, ANCL Gene Discovery Consortium (2016) Diagnosis and misdiagnosis of adult neuronal ceroid lipofuscinosis (Kufs disease). Neurology 87(6):579–584
Román GC, Tatemichi TK, Erkinjuntti T, Cummings JL, Masdeu JC, Garcia JH, Amaducci L, Orgogozo JM, Brun A, Hofman A (1993) Vascular dementia: diagnostic criteria for research studies. Report of the NINDS-AIREN international workshop. Neurology 43(2):250–260. https://doi.org/10.1212/WNL.43.2.250
Román GC, Erkinjuntti T, Wallin A, Pantoni L, Chui HC (2002) Subcortical ischaemic vascular dementia. Lancet Neurol 1(7):426–436. Review. https://doi.org/10.1016/S1474-4422(02)00190-4
Banerjee G, Carare R, Cordonnier C, Greenberg SM, Schneider JA, Smith EE, Buchem MV, Grond JV, Verbeek MM, Werring DJ (2017) The increasing impact of cerebral amyloid angiopathy: essential new insights for clinical practice. J Neurol Neurosurg Psychiatry 8(11):982–994. https://doi.org/10.1136/jnnp-2016-314697
Knudsen KA, Rosand J, Karluk D (2001) Clinical diagnosis of cerebral amyloid angiopathy: validation of the Boston criteria. Neurology 56(4):537–539. https://doi.org/10.1212/WNL.56.4.537
Pescini F, Nannucci S, Bertaccini B, Salvadori E, Bianchi S, Ragno M, Sarti C, Valenti R, Zicari E, Moretti M, Chiti S, Stromillo ML, De Stefano N, Dotti MT, Federico A, Inzitari D, Pantoni L (2012) The cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) Scale: a screening tool to select patients for NOTCH3 gene analysis. Stroke 43:2871–2876
Ferrari C, Nacmias B, Sorbi S (2014) Uncommon dementias. In: Galimberti D, Scarpini E (eds) Neurodegener Dis pp 193–226. https://doi.org/10.1007/978-1-4471-6380-0_12
Nannucci S, Donnini I, Pantoni L (2014) Inherited leukoencephalopathies with clinical onset in middle and old age. J Neurol Sci 347(1–2):1–13. https://doi.org/10.1016/j.jns.2014.09.020
Dallabona C, Diodato D, Kevelam SH, Haack TB, Wong LJ, Salomons GS (2014) Novel (ovario) leukodystrophy related to AARS2 mutations. Neurology 82(23):2063–2071. https://doi.org/10.1212/WNL.0000000000000497
van der Knaap MS, Naidu S, Breiter SN, Blaser S, Stroink H, Springer S, Begeer JC, van Coster R, Barth PG, Thomas NH, Valk J, Powers JM (2001) Alexander disease: diagnosis with MR imaging. AJNR Am J Neuroradiol 22(3):541–552
van der Knaap MS, Pronk JC, Scheper GC (2006) Vanishing white matter disease. Lancet Neurol 5(5):413–423. https://doi.org/10.1016/S1474-4422(06)70440-9
Yatsuga S, Povalko N, Nishioka J, Katayama K, Kakimoto N, Matsuishi T, Kakuma T, Koga Y, Taro Matsuoka for MELAS Study Group in Japan (2012, 1820) MELAS: a nationwide prospective cohort study of 96 patients in Japan. Biochim Biophys Acta:619–624
Brunberg JA, Jacquemont S, Hagerman RJ, Berry-Kravis EM, Grigsby J, Leehey MA, Tassone F, Brown WT, Greco CM, Hagerman PJ (2002) Fragile X premutation carriers: characteristic MR imaging findings of adult male patients with progressive cerebellar and cognitive dysfunction. AJNR Am J Neuroradiol 23(10):1757–1766
Manji H, Jäger HR, Winston A (2013) HIV, dementia and antiretroviral drugs: 30 years of an epidemic. J Neurol Neurosurg Psychiatry 84(10):1126–1137. https://doi.org/10.1136/jnnp-2012-304022
Abdel Razek AA, Alvarez H, Bagg S, Refaat S, Castillo M (2014) Imaging spectrum of CNS vasculitis. Radiographics (34, 4):873–894
Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, Fujihara K, Havrdova E, Hutchinson M, Kappos L, Lublin FD, Montalban X, O'Connor P, Sandberg-Wollheim M, Thompson AJ, Waubant E, Weinshenker B, Wolinsky JS (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69(2):292–302. https://doi.org/10.1002/ana.22366
Kaneko M, Sano K, Nakayama J, Amano N (2010) Nasu-Hakola disease: the first case reported by Nasu and review: the 50th anniversary of Japanese Society of Europathology. Neuropathology 30(5):463–470
Taglia I, Formichi P, Battisti C, Peppoloni G, Barghigiani M, Tessa A, Federico A (2017) Primary familial brain calcification with a novel SLC20A2 mutation: analysis of PiT-2 expression and localization. J Cell Physiol. https://doi.org/10.1002/jcp.26104
Tsakanikas D, Relkin (2007) N Normal pressure hydrocephalus. Semin Neurol 27(1):58–65. https://doi.org/10.1055/s-2006-956756
Shprecher D, Schwalb J, Kurlan R (2008) Normal pressure hydrocephalus: diagnosis and treatment. Curr Neurol Neurosci Rep 8(5):371–376. https://doi.org/10.1007/s11910-008-0058-2
Bradley WG Jr (2016) Magnetic resonance imaging of normal pressure hydrocephalus. Semin Ultrasound CT MR 37(2):120–128. https://doi.org/10.1053/j.sult.2016.01.005
Ishikawa M, Yamada S, Yamamoto K (2016) Early and delayed assessments of quantitative gait measures to improve the tap test as a predictor of shunt effectiveness in idiopathic normal pressure hydrocephalus. Fluids Barriers CNS 13(1):20. https://doi.org/10.1186/s12987-016-0044-z
Snowden JS, Stopford CL, Julien CL, Thompson JC, Davidson Y, Gibbons L, Pritchard A, Lendon CL, Richardson AM, Varma A, Neary D, Mann D (2007) Cognitive phenotypes in Alzheimer’s disease and genetic risk. Cortex 43(7):835–845. https://doi.org/10.1016/S0010-9452(08)70683-X
McMonagle P, Deering F, Berliner Y, Kertesz A (2006) The cognitive profile of posterior cortical atrophy. Neurology 66(3):331–338. https://doi.org/10.1212/01.wnl.0000196477.78548.db
Alladi S, Xuereb J, Bak T, Nestor P, Knibb J, Patterson K, Hodges JR (2007) Focal cortical presentations of Alzheimer’s disease. Brain 130(10):2636–2645. https://doi.org/10.1093/brain/awm213
Ossenkoppele R, Pijnenburg YA, Perry DC, Cohn-Sheehy BI, Scheltens NM, Vogel JW, Kramer JH, van der Vlies AE, La Joie R, Rosen HJ, van der Flier WM, Grinberg LT, Rozemuller AJ, Huang EJ, van Berckel BN, Miller BL, Barkhof F, Jagust WJ, Scheltens P, Seeley WW, Rabinovici GD (2015) The behavioural/dysexecutive variant of Alzheimer’s disease: clinical, neuroimaging and pathological features. Brain 138(9):2732–2749. https://doi.org/10.1093/brain/awv191
Crutch SJ, Schott JM, Rabinovici GD, Murray M, Snowden JS, van der Flier WM, Dickerson BC, Vandenberghe R, Ahmed S, Bak TH, Boeve BF, Butler C, Cappa SF, Ceccaldi M, de Souza LC, Dubois B, Felician O, Galasko D, Graff-Radford J, Graff-Radford NR, Hof PR, Krolak-Salmon P, Lehmann M, Magnin E, Mendez MF, Nestor PJ, Onyike CU, Pelak VS, Pijnenburg Y, Primativo S, Rossor MN, Ryan NS, Scheltens P, Shakespeare TJ, Suárez González A, Tang-Wai DF, Yong KX, Carrillo M, Fox NC, Alzheimer’s Association ISTAART Atypical Alzheimer’s Disease and Associated Syndromes Professional Interest Area (2017) Consensus classification of posterior cortical atrophy. Alzheimers Dement 13(8):S1552–S5260. https://doi.org/10.1016/j.jalz.2017.01.014
Gallo M, Frangipane F, Cupidi C, De Bartolo M, Turone S, Ferrari C, Nacmias B, Grimaldi G, Laganà V, Colao R, Bernardi L, Anfossi M, Conidi ME, Vasso F, Curcio SAM, Mirabelli M, Smirne N, Torchia G, Muraca MG, Puccio G, Di Lorenzo R, Piccininni M, Tedde A, Maletta RG, Sorbi S, Bruni AC (2017) The novel PSEN1 M84V mutation associated to frontal dysexecutive syndrome, spastic paraparesis, and cerebellar atrophy in a dominant Alzheimer's disease family. Neurobiol Aging 56:213.e7–213.e12
Wardlaw JM, Smith C, Dichgans M (2013) Mechanisms of sporadic cerebral small vessel disease: insights from neuroimaging. Lancet Neurol 12(5):483–497. https://doi.org/10.1016/S1474-4422(13)70060-7
O'Brien JT, Thomas A (2015) Vascular dementia. Lancet 386(10004):1698–1706. https://doi.org/10.1016/S0140-6736(15)00463-8
Ballard C, Gauthier S, Corbett A (2011) Alzheimer’s disease. Lancet 377:1019–1031
Sudre CH, Bocchetta M, Cash D, Thomas DL, Woollacott I, Dick KM, van Swieten J, Borroni B, Galimberti D, Masellis M, Tartaglia MC, Rowe JB, Graff C, Tagliavini F, Frisoni G, Laforce R Jr, Finger E, de Mendonça A, Sorbi S, Ourselin S, Cardoso MJ, Rohrer JD, Genetic FTD, Initiative GENFI (2017) White matter hyperintensities are seen only in GRN mutation carriers in the GENFI cohort. Neuroimage Clin 15:171–180. https://doi.org/10.1016/j.nicl.2017.04.015
Regal P (2017) Delirium: a guide for the general physician. Clin Med (Lond) 17(4):381. https://doi.org/10.7861/clinmedicine.17-4-381
Geschwind MD (2016) Rapidly progressive dementia. Continuum (Minneap Minn) 22(2):510–537
Geschwind MD (2015) Prion diseases. Continuum (Minneap Minn) 21(6 Neuroinfectious disease):1612–1638
Lattanzio F, Abu-Rumeileh S, Franceschini A, Kai H, Amore G, Poggiolini I, Rossi M, Baiardi S, McGuire L, Ladogana A, Pocchiari M, Green A, Capellari S, Parchi P (2017) Prion-specific and surrogate CSF biomarkers in Creutzfeldt-Jakob disease: diagnostic accuracy in relation to molecular subtypes and analysis of neuropathological correlates of p-tau and Aβ42 levels. Acta Neuropathol 133(4):559–578. https://doi.org/10.1007/s00401-017-1683-0
Zhou JY, Xu B, Lopes J, Blamoun J, Li L (2017) Hashimoto encephalopathy: literature review. Acta Neurol Scand 135(3):285–290. https://doi.org/10.1111/ane.12618
Graus F, Saiz A (2008) Limbic encephalitis: an expanding concept. Neurology 70(7):500–501. https://doi.org/10.1212/01.wnl.0000299189.43164.96
Dalmau J, Gleichman AJ, Hughes EG (2008) AntiNMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol 7(12):1091–1098. https://doi.org/10.1016/S1474-4422(08)70224-2
Nishimoto A, Usery J, Winton JC, Twilla J (2017) High-dose parenteral thiamine in treatment of Wernicke's encephalopathy: case series and review of the literature. In Vivo 31(1):121–124
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ferrari, C., Nacmias, B. & Sorbi, S. The diagnosis of dementias: a practical tool not to miss rare causes. Neurol Sci 39, 615–627 (2018). https://doi.org/10.1007/s10072-017-3206-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-017-3206-0