Abstract
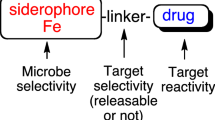
Bacterial iron uptake machinery can be hijacked for the targeted delivery of antibiotics into pathogens by attaching antibiotics to siderophores, iron chelators that are employed by bacteria to obtain this essential nutrient. We synthesized and evaluated Ent–SS–Cipro, a siderophore–antibiotic conjugate comprised of the triscatecholate siderophore enterobactin and the fluoroquinolone antibiotic ciprofloxacin that contains a self-immolative disulfide linker. This linker is designed to be cleaved after uptake into the reducing environment of the bacterial cytoplasm. We show that the disulfide bond of Ent–SS–Cipro is cleaved by reducing agents, including the cellular reductant glutathione, which results in release of the unmodified fluoroquinolone antibiotic. Antibacterial activity assays against a panel of Escherichia coli show that Ent–SS–Cipro exhibits activity against some, but not all, E. coli. This work informs the design of siderophore–antibiotic conjugates, particularly those carrying antibiotics with cytoplasmic targets that require release after uptake into bacterial cells, and indicates that disulfide linkers may not be generally applicable for conjugation strategies of antibiotics.
Graphical abstract








Similar content being viewed by others
Abbreviations
- DHBS:
-
2,3-Dihydroxybenzoyl serine
- DTT:
-
1,4-Dithiothreitol
- Ent:
-
Enterobactin
- GSH:
-
Glutathione
- GSSG:
-
Glutathione disulfide
- TCEP:
-
Tris(2-carboxyethyl)phosphine
References
Clatworthy AE, Pierson E, Hung DT (2007) Nat Chem Biol 3:541–548
Lewis K (2013) Nat Rev Drug Discovery 12:371–387
Hood MI, Skaar EP (2012) Nat Rev Microbiol 10:525–537
Weinberg ED (1975) JAMA 231:39–41
Palmer LD, Skaar EP (2016) Annu Rev Genet 50:67–91
Miethke M, Marahiel MA (2007) Microbiol Mol Biol Rev 71:413–451
Hider RC, Kong X (2010) Nat Prod Rep 27:637–657
Chu BC, Garcia-Herrero A, Johanson TH, Krewulak KD, Lau CK, Peacock RS, Slavinskaya Z, Vogel HJ (2010) Biometals 23:601–611
Wencewicz TA, Miller MJ (2017) Top Med Chem. Springer, Berlin, Heidelberg, pp 1–33
Braun V, Pramanik A, Gwinner T, Köberle M, Bohn E (2009) Biometals 22:3–13
Zahner H, Diddens H, Keller-Schierlein W, Nägeli HU (1977) Jpn J Antibiot 30:S201–S206
Diarra MS, Lavoie MC, Jacques M, Darwish I, Dolence EK, Dolence JA, Ghosh A, Ghosh M, Miller MJ, Malouin F (1996) Antimicrob Agents Chemother 40:2610–2617
Ji C, Juárez-Hernández RE, Miller MJ (2012) Future Med Chem 4:297–313
Page MGP (2013) Ann NY Acad Sci 1277:115–126
Tillotson GS (2016) Infect Dis (Auckl) 9:45–52
Watanabe N-A, Nagasu T, Katsu K, Kitoh K (1987) Antimicrob Agents Chemother 31:497–504
Curtis NAC, Eisenstadt RL, East SJ, Cornford RJ, Walker LA, White AJ (1988) Antimicrob Agents Chemother 32:1879–1886
Silley P, Griffiths JW, Monsey D, Harris AM (1990) Antimicrob Agents Chemother 34:1806–1808
Hashizume T, Sanada M, Nakagawa S, Tanaka N (1990) J Antibiot 43:1617–1620
Nikaido H, Rosenberg EY (1990) J Bacteriol 172:1361–1367
McKee JA, Sharma SK, Miller MJ (1991) Bioconjugate Chem 2:281–291
Dolence EK, Minnick AA, Lin C-E, Miller MJ, Payne SM (1991) J Med Chem 34:968–978
Ji C, Miller PA, Miller MJ (2012) J Am Chem Soc 134:9898–9901
Zheng T, Nolan EM (2014) J Am Chem Soc 136:9677–9691
Kohira N, West J, Ito A, Ito-Horiyama T, Nakamura R, Sato T, Rittenhouse S, Tsuji M, Yamano Y (2016) Antimicrob Agents Chemother 60:729–734
Chairatana P, Zheng T, Nolan EM (2015) Chem Sci 6:4458–4471
Ruiz N, Kahne D, Silhavy TJ (2006) Nat Rev Microbiol 4:57–66
Silhavy TJ, Kahne D, Walker S (2010) Cold Spring Harb Perspect Biol 2:a000414
Hennard C, Truong QC, Desnottes J-F, Paris J-M, Moreau NJ, Abdallah MA (2001) J Med Chem 44:2139–2151
Rivault F, Liébert C, Burger A, Hoegy F, Abdallah MA, Schalk IJ, Mislin GLA (2007) Bioorg Med Chem Lett 17:640–644
Wencewicz TA, Möllmann U, Long TE, Miller MJ (2009) Biometals 22:633–648
Md-Saleh SR, Chilvers EC, Kerr KG, Milner SJ, Snelling AM, Weber JP, Thomas GH, Duhme-Klair A-K, Routledge A (2009) Bioorg Med Chem Lett 19:1496–1498
Noël S, Gasser V, Pesset B, Hoegy F, Rognan D, Schalk IJ, Mislin GLA (2011) Org Biomol Chem 9:8288–8300
Juárez-Hernández RE, Miller PA, Miller MJ (2012) ACS Med Chem Lett 3:799–803
Ji C, Miller MJ (2012) Bioorg Med Chem 20:3828–3836
Wencewicz TA, Miller MJ (2013) J Med Chem 56:4044–4052
Wencewicz TA, Long TE, Möllmann U, Miller MJ (2013) Bioconjugate Chem 24:473–486
Milner SJ, Seve A, Snelling AM, Thomas GH, Kerr KG, Routledge A, Duhme-Klair A-K (2013) Org Biomol Chem 11:3461–3468
Souto A, Montaos MA, Balado M, Osorio CR, Rodríguez J, Lemos ML, Jiménez C (2013) Bioorg Med Chem 21:295–302
Milner SJ, Snelling AM, Kerr KG, Abd-El-Aziz A, Thomas GH, Hubbard RE, Routledge A, Duhme-Klair A-K (2014) Bioorg Med Chem 22:4499–4505
Fardeau S, Dassonville-Klimpt A, Audic N, Sasaki A, Pillon M, Baudrin E, Mullié C, Sonnet P (2014) Bioorg Med Chem 22:4049–4060
Ji C, Miller MJ (2015) Biometals 28:541–551
Braun V, Günthner K, Hantke K, Zimmermann L (1983) J Bacteriol 156:308–315
Neumann W, Sassone-Corsi M, Raffatellu M, Nolan EM (2018) J Am Chem Soc 140:5193–5201
Lin H, Fischbach MA, Liu DR, Walsh CT (2005) J Am Chem Soc 127:11075–11084
Zhu M, Valdebenito M, Winkelmann G, Hantke K (2005) Microbiology 151:2363–2372
Zheng T, Nolan EM (2015) Bioorg Med Chem Lett 25:4987–4991
Paulen A, Gasser V, Hoegy F, Perraud Q, Pesset B, Schalk IJ, Mislin GLA (2015) Org Biomol Chem 13:11567–11579
Paulen A, Hoegy F, Roche B, Schalk IJ, Mislin GLA (2017) Bioorg Med Chem Lett 27:4867–4870
Liu R, Miller PA, Vakulenko SB, Stewart NK, Boggess WC, Miller MJ (2018) J Med Chem 61:3845–3854
Zheng T, Bullock JL, Nolan EM (2012) J Am Chem Soc 134:18388–18400
Vrudhula VM, MacMaster JF, Li Z, Kerr DE, Senter PD (2002) Bioorg Med Chem Lett 12:3591–3594
Vlahov IR, Leamon CP (2012) Bioconjugate Chem 23:1357–1369
Brezden A, Mohamed MF, Nepal M, Harwood JS, Kuriakose J, Seleem MN, Chmielewski J (2016) J Am Chem Soc 138:10945–10949
Kim HS, Song WY, Kim HJ (2015) Org Biomol Chem 13:73–76
Ritz D, Beckwith J (2001) Annu Rev Microbiol 55:21–48
Masip L, Veeravalli K, Georgiou G (2006) Antioxid Redox Signaling 8:753–762
Van Loi V, Rossius M, Antelmann H (2015) Front Microbiol 6:187
Serjeant EP, Dempsey B, International Union of Pure and Applied Chemistry (IUPAC) (1979) IUPAC Chemical Data Series, No 23. Pergamon Press, Oxford, New York
Acknowledgements
We thank the National Institutes of Health (NIH Grants 1R21AI126465 and 1R01AI114625) for financial support; Lynette Cegelski for providing E. coli UTI89; Manuela Raffatellu for providing E. coli JB2; Ardeypharm GmbH for providing E. coli Nissle 1917. W.N. acknowledges the German National Academy of Sciences Leopoldina for a postdoctoral fellowship (LPDS 2015-08). MS instrumentation maintained by the MIT Center for Environmental Health Sciences (CEHS) is supported by a core center grant from the National Institute of Environmental Health Sciences, National Institutes of Health (NIEHS grant P30-ES002109).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Neumann, W., Nolan, E.M. Evaluation of a reducible disulfide linker for siderophore-mediated delivery of antibiotics. J Biol Inorg Chem 23, 1025–1036 (2018). https://doi.org/10.1007/s00775-018-1588-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-018-1588-y