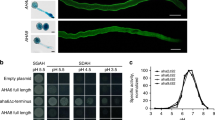
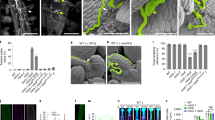
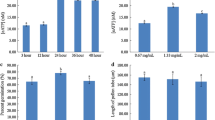
Abstract
The plasma membrane H+ ATPase is a member of the P-ATPase family transporting H+ from the cytosol to the extracellular space and thus energizing the plasma membrane for the uptake of ions and nutrients. As a housekeeping gene, this protein can be detected in almost every plant cell including the exclusive expression of specific isoforms in pollen grains and tubes where its activity is a prerequisite for successful germination and growth of pollen tubes. This review summarizes the current knowledge on pollen PM H+ ATPases and hypothesizes a central role for pollen-specific isoforms of this protein in tube growth. External as well as cytosolic signals from signal transduction and metabolic pathways are integrated by the PM H+ ATPase and directly translated to tube growth rates, allocating the PM H+ ATPase to an essential node in the signalling network of pollen tubes in their race to the ovule.





Similar content being viewed by others
References
Arango M, Gevaudant F, Oufattole M, Boutry M (2003) The plasma mebrane proton pump ATPase: the significance of gene subfamilies. Planta 216:355–365
Axelsen KB, Venema K, Jahn T, Baunsgaard L, Palmgren MG (1999) Molecular dissection of the C-terminal regulatory domain of the plant plasma membrane H+ ATPase AHA2: mapping of residues that when altered give rise to an activated enzyme. Biochemistry 38(22):7277–7234
Baunsgaard L, Fuglsang AT, Jahn T, Korthout HAAJ, De Boer AH, Palmgren MG (1998) The 14-3-3 proteins associate with the plant plasma membrane H+ ATPase to generate a fusicoccin binding complex and a fusicoccin responsive system. Plant J 13:661–671
Becker JD, Boavida LC, Carneiro J, Haury M, Feijo J (2003) Transcriptional profiling of Arabidopsis tissues reveals the unique characterictics of the pollen transcriptome. Plant Physiol 133:713–725
Bedinger P (1992) The remarkable biology of pollen. Plant Cell 4:879–887
Bobik K, Duby G, Nizet Y, Vandermeeren C, Stiernet P, Kanczewsky J, Boutry M (2010) Two widely expressed plasma membrane H+ ATPase isoforms of Nicotiana tabacum are differentially regulated by phosphorylation of their penultimate threonine. Plant J 62:291–301
Bock KW, Honys D, Ward JM, Padmanaban S, Nawrocki EP, Hirschi KD, Twell D, Sze H (2006) Integrating membrane transport with male gametophyte development and function through transcriptomics. Plant Physiol 140:1151–1168
Borch J, Bych K, Roepstorff P, Palmgren MG, Fuglsang AT (2002) Phosphorylation-independent interaction between 14-3-3 protein and the plant plasma membrane H+ ATPase. Biochem Soc Trans 30:411–415
Briskin DP (1990) The plasma membrane H+ ATPase of higher plant cells: biochemistry and transport function. Biochim Biophys Acta 1019:95–109
Briskin DP, Hanson JB (1992) How does the plasma membrane H+ ATPase pump protons? J Exp Bot 248:269–289
Briskin DP, Reynolds-Niesman I (1991) Determination of H+/ATP stoichiometry for the plasma membrane H+ ATPase from red beet (Beta vulgaris L.) storage tissue. Plant Physiol 95:242–250
Briskin DP, Leonard R, Hodges TK (1987) Isolation of the plasma membrane: membrane markers and general principles. Meth Enzymol 148:542–558
Buch-Pedersen MJ, Rudashevskaya EL, Berner TS, Venema K, Palmgren MG (2006) Potassium as an intrinsic uncoupler of the plasma membrane H+-ATPase. J Biol Chem 281:38285–38292
Buch-Pedersen MJ, Pedersen BP, Veierskov B, Nissen P, Palmgren MG (2009) Protons and how they are transported by proton pumps. Pflugers Arch 457:573–579
Caesar K, Elgass K, Chen Z, Hiuppenberger P, Witthöft J, Schleifenbaum F, Blatt MR, Oecking C, Harter K (2011) A fast brassinolide-regulated response pathway in the plasma membrane of Arabidopsis thaliana. Plant J 66:528–540
Cai G, Moscatelli A, Cresti M (1997) Cytoskeletal organization and pollen tube growth. Trends Plant Sci 2:86–91
Camoni L, Fullone MR, Marra M, Aducci P (1998) The plasma membrane H+ ATPase from maize roots is phosphorylated in the C-terminal domain by a calcium-dependent protein kinase. Physiol Plant 104:549–555
Camoni L, Iori V, Marra M, Aducci P (2000) Phosphorylation-dependent interaction between plant plasma membrane H+-ATPase and 14-3-3 proteins. J Biol Chem 275(14):9919–9923
Camoni L, Marra M, Garufi A, Visconti S, Aducci P (2006) The maize plasma membrane H+ ATPase is regulated by a sugar-induced transduction pathway. Plant Cell Physiol 47:743–747
Certal AC, Almeida RB, Carvalho LM, Wong E, Moreno N, Michard E, Carneiro J, Rodriguez-Leon J, Wu H-M, Cheung AY, Feijo J (2008) Exclusion of a proton ATPase from the apical membrane is associated with cell polarity and tip growth in Nicotiana tabacum pollen tubes. Plant Cell 20:614–634
Cheung AY, Wu H-M (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Ann Rev Plant Biol 59:547–572
Cid A, Perona R, Serrano R (1987) Replacement of the promoter of the yeast plasma membrane ATPase gene by a galactose-dependent promoter and its physiological consequences. Curr Genet 12:105–110
Cole RA, Fowler JE (2006) Polarized growth: maintaining focus on the tip. Curr Opin Plant Biol 9:579–588
Cresti M, Blackmore S, van Went JL (1992) Atlas of sexual reproduction in flowering plants. Springer Verlag, Berlin
Daher FB, Chebli Y, Geitmann A (2009) Optimization of conditions for germination of cold-stored Arabidopsis thaliana pollen. Plant Cell Rep 28:347–357
Dai S, Li L, Chen T, Chong K, Xue Y, Wang T (2006) Proteomic analysis of Oriza sativa pollen reveal novel proteins associated with pollen germination and tube growth. Proteomics 6:2504–2529
de Kerchove d'Exaerde A, Supply P, Dufour JP, Bogarts P, Thines D, Goffeau A, Boutry M (1995) Functional complementation of a null mutation of the yeast Saccharomyces cerevisae plasma membrane H+ ATPase by a plant H+ ATPase gene. J Biol Chem 270:23828–23837
Dickinson DB (1978) Influence of borate and penta-erythritol concentrations on germination and tube growth of Lilium longiflorum pollen. J Am Soc Hortic Sci 103:413–416
Ekberg K, Palmgren MG, Veierskov B, Buch-Pedersen MJ (2010) A novel mechanism of the P-type ATPase autoinhibition involving both termini of the protein. J Biol Chem 285:7344–7350
Feijó JA, Malhó R, Obermeyer G (1995) Ion dynamics and its possible role during in vitro pollen germination and tube growth. Protoplasma 187:155–167
Feijó JA, Sainhas J, Hackett G, Kunkel JG, Hepler PK (1999) Growing pollen tubes possess a constitutive alkaline band in the clear zone and a growth-dependent acidic tip. J Cell Biol 144:483–496
Feijó JA, Sainhas J, Holdaway-Clarke T, Cordeiro S, Kunkel JG, Hepler PK (2001) Cellular oscillations and the regulation of growth: the pollen tube paradigm. BioEssays 23(1):86–94
Fricker MD, White NS, Obermeyer G (1997) pH gradients are not associated with tip growth in pollen tubes of Lilium longiflorum. J Cell Sci 110:1729–1740
Fuglsang AT, Visconti S, Drumm K, Jahn T, Stensballe A, Mattei M, Jensen ON, Aducci P, Palmgren MG (1999) Binding of 14-3-3 protein to the plasma membrane H+ ATPase AHA2 involves the three C-terminal residues Tyr (946)–Thr–Val and requires phosphorylation of the THR (947). J Biol Chem 274:36774–36780
Fuglsang AT, Tulinius G, Cui N, Palmgren MG (2006) Protein phosphatase 2A-scaffolding subunit A interacts with plasma membrane H+-ATPase C-terminus in the same region as 14-3-3 protein. Physiol Plant 128:334–340
Fuglsang AT, Guo Y, Cuin TA, Qiu Q, Song C, Kristiansen KA, Bych K, Schulz A, Shabala S, Schumaker KS, Palmgren MG, Zhu J-K (2007) Arabidopsis protein kinase PKS5 inhibits the plasma membrane H+ ATPase by preventing interaction with 14-3-3 protein. Plant Cell 19:1617–1634
Fullone MR, Visconti S, Marra M, Fogliano V, Aducci P (1998) Fusicoccin effect on the in vitro interaction between plant 14-3-3 proteins and plasma membrane H+-ATPase. J Biol Chem 273(13):7698–7702
Gaxiola RA, Palmgren MG, Schumacher K (2007) Plant proton pumps. FEBS Lett 581:2204–2214
Gehwolf R, Griessner M, Pertl H, Obermeyer G (2002) First patch, then catch: measuring the activity and the mRNA transcripts of a proton pump in individual Lilium pollen protoplasts. FEBS Lett 512:152–156
Geitmann A, Wojciechowicz K, Cresti M (1996) Inhibition of intracellular pectin transport in pollen tubes by monensin, brefeldin A and cytochalasin D. Bot Acta 109:373–381
Giacometti S, Camoni L, Albumi C, Visconti S, De Michelis MI, Aducci P (2004) Tyrosine phosphorylation inhibits the interaction of 14-3-3 proteins with the plant plasma mebrane H+ ATPase. Plant Biol 6:422–431
Grobei MA, Qeli E, Brunner E, Rehrauer H, Zhang R, Roschitzki B, Basler K, Ahrens CH, Grossniklaus U (2009) Deterministic protein inference for shotgun proteomics data provides new insights into Arabidopsis pollen development and function. Genome Res 19:1786–1800
Hager A, Debus G, Edel H-G, Stransky H, Serrano R (1991) Auxin induces exocytosis and the rapid synthesis of a high-turnover pool of plasma-membrane H+ ATPase. Planta 185: 5327–5537
Hayashi Y, Nakamura S, Takemiya A, Takahashi Y, Shimazaki K-I, Kinoshita T (2010) Biochemical characterization of in vitro phosphorylation and dephosphorylation of the plasma membrane H+ ATPase. Plant Cell Physiol 51:1186–1196
Heslop-Harrison J (1979) An interpretation of the hydrodynamics of pollen. Am J Bot 66:737–743
Holdaway-Clarke T, Hepler PK (2003) Control of pollen tube growth: role of ion gradients and fluxes. New Phytol 159:539–563
Janicka-Russak M, Klobus G (2007) Modification of plasma membrane and vacuolar H+ ATPases in response to NaCl and ABA. Plant Physiol 164:295–302
Korthout HAAJ, DeBoer AH (1994) A fusicoccin binding protein belongs to the family of 14-3-3 brain protein homologs. Plant Cell 6:1681–1692
Kost B (2008) Spatial control of Rho (rRac.Rop) signaling in tip-growing plant cells. Trends Cell Biol 18:119–127
Larsson C, Widell S, Kjellbom P (1987) Preparation of high-purity plasma membranes. Meth Enzymol 148:350–382
Lee SH, Singh AP, Chung GC, Ahn SJ, Noh EK, Steudle E (2004) Exposure of roots of cucumber (Cucumis sativus) to low temperature severely reduces root pressure, hydraulic conductivity and active transport of nutrients. Physiol Plant 120:413–420
Lefebvre B, Arango M, Oufattole M, Crouzet J, Purnelle B, Boutry M (2005) Identification of a Nicotiana plumbaginifolia plasma membrane H+ ATPase gene expressed in the pollen tube. Plant Mol Biol 58:775–787
Lino B, Baizabel-Aguirre VM, Gonzalez de la Vara LE (1998) The plasma membrane H+ ATPase from beet root is inhibited by a calcium-dependent phosphorylation. Planta 204:352–359
Malhó R, Trewavas AJ (1996) Localized apical increases of cytosolic free calcium control pollen tube orientation. Plant Cell 8:1935–1949
Mascarenhas JP (1990) Gene activity during pollen development. Annu Rev Plant Physiol Plant Mol Biol 41:317–338
Maudoux O, Batoko H, Oecking C, Gevaert K, Vandekerckhove J, Boutry M, Morsomme P (2000) A plant plasma membrane H+-ATPase expressed in yeast is activated by phosphorylation at its penultimate residue and binding of 14-3-3 regulatory proteins in the absence of fusicoccin. J Biol Chem 275(23):17762–17770
Messerli M, Danuser G, Robinson KR (1999) Pulsatile influxes of H+, K+ and Ca2+ lag growth pulses of Lilium longiflorum pollen tubes. J Cell Sci 112:1497–1509
Michard E, Dias P, Feijo JA (2008) Tobacco pollen tubes as cellular models for ion dynamics: improved spatial and temporal resolution of extracellular flux and free cytosolic concentration of calcium and protons using pHluorin and YC3.1 CaMeleon. Sex Plant Reprod 21:169–181
Michard E, Alves F, Feijo JA (2009) The role of ion fluxes in polarized cell growth and morphogenesis: the pollen tube as an experimental paradigm. Int J Dev Biol 53:1609–1622
Morandini P, Valera M, Albumi C, Bonza MC, Giacometti S, Ravera G, Murgia I, Soave C, De Michelis MI (2002) A novel interaction partner of the C-terminus of Arabidopsis thaliana plasma membrane H+ ATPase (AHA1 isoform): site and mechanism of action on H+ ATPase activity differ from those of 14-3-3 proteins. Plant J 31:487–497
Morsomme P, Boutry M (2000) The plant plasma membrane H+ ATPase: structure, function and regulation. Biochim Biophys Acta 1465:1–16
Morsomme P, Kerchove d'Exaerde A, de Meester S, Thines D, Goffeau A, Boutry M (1996) Single point mutations in various domains of a plant plasma membrane H+ ATPase expressed in Saccharomyces cerevisiae increase H+ pumping and permit yeast growth at low pH. EMBO J 15:5513–5526
Muniz Garcia MN, Pais SM, Tellez-Inon MT, Capiati DA (2011) Characterization of StPPI1, a proton pump interactor from Solanum tuberosum L. that is up-regualted during tube formation and by abiotic stress. Planta 233:661–674
Obermeyer G, Weisenseel MH (1991) Calcium channel blocker and calmodulin antagonists affect the gradient of free calcium ions in lily pollen tubes. Eur J Cell Biol 56:319–327
Obermeyer G, Lützelschwab M, Heumann H-G, Weisenseel MH (1992) Immunolocalisation of H+ ATPases in the plasma membrane of pollen grains and pollen tubes of Lilium longiflorum. Protoplasma 171:55–63
Obermeyer G, Kriechbaumer R, Strasser D, Maschessnig A, Bentrup F-W (1996) Boric acid stimulates the plasma membrane H+ ATPase of ungerminated lily pollen grains. Physiol Plant 98:281–290
Obermeyer G, Klaushofer H, Nagl M, Höftberger M, Bentrup F-W (1998) In-vitro germination and growth of lily pollen tubes is affected by protein phosphatase inhibitors. Planta 207:303–312
Obermeyer G, Fragner L, Lang V, Weckwerth W (2013) Dynamic adaption of metabolic pathways during germination and growth of lily pollen tubes after inhibition of the lectron transport chain. Plant Physiol 162:1822–1833
Oecking C, Piotroski M, Hagemeier J, Hagemann K (1997) Topology and target interaction of the fusicoccin-binding 14-3-3 homologs of Commenlina communis. Plant J 12:441–453
Ottmann C, Marco S, Jaspert N, Marcon C, Schauer N, Weyand M, Vandermeeren C, Duby G, Boutry M, Wittinghofer A, Rigaud J-L, Oecking C (2007) Structure of a 14-3-3 coordinated hexamer of the plant plasma membrane H+ ATPase by combining X-ray crystallography and electron cryomicroscopy. Mol Cell 25:427–440
Palmgren MG (1990) An H+ ATPase assay: proton pumping and ATPase activity determined simultaneously in the same sample. Plant Physiol 94:882–886
Palmgren MG (1998) Proton gradients and plant growth: role of the plasma membrane H+ ATPase. Adv Bot Res 28:2–70
Palmgren MG (2001) Plant plasma membrane H+ ATPases: powerhouses for nutrient uptake. Annu Rev Plant Physiol Plant Mol Biol 52:817–845
Palmgren MG, Christensen G (1993) Complementation in situ of the yeast plasma membrane H+ ATPase gene pma1 by a H+ ATPase gene from a heterologous species. FEBS Lett 317:216–222
Palmgren MG, Harper JF (1999) Pumping with plant P-type ATPases. J Exp Bot 50:883–893
Parton RM, Fischer S, Malhó R, Papasouliotis O, Jelitto TC, Leonard T, Read ND (1997) Pronounced cytoplasmic pH gradients are not required for tip growth in plant and fungal cells. J Cell Sci 110:1187–1198
Paul A-L, Sehnke P, Ferl RJ (2005) Isoform-specific subcellular localisation among 14-3-3 proteins in Arabidopsis seems to be driven by client interaction. Mol Biol Cell 16:1735–1743
Pedersen BP, Buch-Pedersen MJ, Morth JP, Palmgren MG, Nissen P (2007) Crystal structure of the plasma membrane proton pump. Nature 450:1111–1117
Pedersen CNS, Axelsen KB, Harper JF, Palmgren MG (2012) Evolution of plant P-type ATPases. Front Plant Sci 3. DOI: 10.3389/fpls.2012.00031
Pertl H, Himly M, Gehwolf R, Kriechbaumer R, Strasser D, Michalke W, Richter K, Ferreira F, Obermeyer G (2001) Molecular and physiological characterisation of a 14-3-3 protein from lily pollen grains regulating the activity of the plasma membrane H+ ATPase during pollen grain germination and tube growth. Planta 213:132–141
Pertl H, Gehwolf R, Obermeyer G (2005) The distribution of membrane-bound 14-3-3 proteins in organelle-enriched fractions of germinating lily pollen. Plant Biol 7:140–147
Pertl H, Schulze WX, Obermeyer G (2009) The pollen organelle membrane proteome reveals highly spatial–temporal dynamics during germination and tube growth of lily pollen. J Proteome Res 8:5142–5152
Pertl H, Poeckl M, Blaschke C, Obermeyer G (2010) Osmoregulation in Lilium pollen grains occurs via modulation of the plasma membrane H+ ATPase activity by 14-3-3 proteins. Plant Physiol 154:1921–1928
Pertl H, Rittmann S, Schulze WX, Obermeyer G (2011) Identification of lily pollen 14-3-3 isoforms and their subcellular and time-dependent expression profile. Biol Chem 392:249–262
Pertl-Obermeyer H, Obermeyer G (2013) Pollen cultivation and preparation for proteomic studies. In: Jorrin-Novo JV (ed) Plant proteomics: methods and protocols, vol 1072. Methods in Molecular Biology, Springer Verlag. doi:10.1007/978-1-62703-631-3_30
Pierson ES, Miller DD, Callaham DA, Shipley AM, Rivers BA, Cresti M, Hepler PK (1994) Pollen tube growth is coupled to the extracellular calcium ion flux and the intracellular calcium gradient: Effect of BAPTA-type buffers and hypertonic media. Plant Cell 6:1815–1828
Piette A-S, Derua R, Waelkens E, Boutry M, Duby G (2011) A phosphorylation in the C-terminal auto-inhibitory domain of the plant plasma membrane H+ ATPase activates the enzyme with no requiremnet for regugulatory 14-3-3 proteins. J Biol Chem 286:18474–18482
Plätzer K, Obermeyer G, Bentrup F-W (1997) AC fields of low frequency and amplitude stimulate pollen tube growth possible via stimulation of the plasma membrane H+ ATPase. Bioelectrochem Bioenerg 44:95–102
Rathore KS, Cork RJ, Robinson KR (1991) A cytoplasmic gradient of Ca2+ is correlated with the growth of lily pollen tubes. Dev Biol 148:612–619
Regenberg B, Villalba JM, Lanfermeijer FC, Palmgren MG (1995) C-terminal deletion analysis of plant plasma membrane H+ ATPase: Yeast as a model system for solute transport across the plant plasma membrane. Plant Cell 7:1655–1666
Rodriguez-Rosales MP, Roldán M, Belver A, Donaire JP (1989) Correlation between in vitro germination capacity and proton extrusion in olive pollen. Plant Physiol Biochem 27:723–728
Rudashevskaya EL, Ye J, Jensen ON, Fuglsang AT, Palmgren MG (2012) Phosphosite mapping of P-type plasma membrane H+ ATPase in homologous and heterologous environments. J Biol Chem 287:4904–4913
Schrauwen JAM, de Groot PFM, Van Herpen MMA, van der Lee T, Reynen WH, Weterings K, Wullems GJ (1990) Stage-related expression of mRNAs during pollen development in lily and tobacco. Planta 182:298–304
Serrano R (1989) Structure and function of plasma membrane ATPase. Ann Rev Plant Physiol Plant Mol 40:61–94
Simon EW (1974) Phospholipids and plant membrane permeability. New Phytol 73:377–420
Southworth D (1983) pH changes during pollen germination in Lilium longiflorum. In: Mulcahy DL, Ottaviano E (eds) Pollen: biology and implications for plant breeding. Elsevier, New York, pp 61–65
Steinhorst L, Kudla J (2013) Calcium - a central regulator of pollen germination and tube growth. Biochim Biophys Acta 1833:1573–1581
Svennelid F, Olsson A, Piotroski M, Rosenquist M, Ottman C, Larsson C, Oecking C, Sommarin M (1999) Phosphorylation of Thr-948 at the C-terminus of the plasma membrane H+ ATPase creates a binding site for the regulatory 14-3-3 protein. Plant Cell 11:2379–2391
Tanaka I (1993) Development of male gametes in flowering plants. J Plant Res 106:55–63
Tupy J, Rihova L (1984) Changes and growth effect of pH in pollen tube culture. J Plant Physiol 115:1–10
Tupy J, Hrabetova E, Balatkova V (1977) A simple rapid method of determining pollen tube growth in mass culture. Plant Sci Lett 9:285–290
Turian G (1981) Decreasing pH-gradient toward the apex of germinating pollen tubes. Bot Helv 91:161–167
Villalba JM, Palmgren MG, Berberian GE, Ferguson C, Serrano R (1992) Functional expression of plant plasma membrane H+ ATPase in yeast endoplasmic reticulum. J Biol Chem 267:12341–12349
Viotti C, Luoni L, Morandini P, De Michaelis MI (2005) Characterization of the interaction between the plasma membrane H+ ATPase of Arabidopsis thaliana and a novel interactor (PPI1). FEBS J 272:5864–5871
Wei LQ, Xu WY, Deng ZY, Su Z, Xue Y, Wang T (2010) Genome-scale analysis and comparison of gene expression profiles in developing and germinated pollen in Oryza sativa. BMC Genomics 11:338
Weisenseel MH, Nuccitelli R, Jaffe LA (1975) Large electrical currents traverse growing pollen tubes. J Cell Biol 66:556–567
Yang Y, Qin Y, Xie C, Zhao F, Zhao J, Liu D, Chen S, Fuglsang AT, Palmgren MG, Schumaker KS, Deng XW, Guo Y (2010) The Arabidopsis chaperone J3 regulates the plasma membrane H+ ATPase through interaction with the PKS5 kinase. Plant Cell 22:1313–1332
Zonia L, Cordeiro S, Tupy J, Feijó JA (2002) Oscillatory chloride efflux at the pollen tube apex has a role in growth and cell volume regulation and is targeted by inositol 3,4,5,6-tetrakisphosphate. Plant Cell 14:2233–2249
Acknowledgements
Experimental work on pollen PM H+ ATPases was partially financed by a grant of the Austrian Science Fund (FWF, P21298).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Friedrich W. Bentrup
V. Lang and H. Pertl-Obermeyer contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lang, V., Pertl-Obermeyer, H., Safiarian, M.J. et al. Pump up the volume - a central role for the plasma membrane H+ pump in pollen germination and tube growth. Protoplasma 251, 477–488 (2014). https://doi.org/10.1007/s00709-013-0555-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-013-0555-2