Abstract
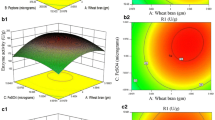
A central composite experimental design leading to a set of 16 experiments with different combinations of pH and temperature was performed to attain the optimal activities of xylose reductase (XR) and xylitol dehydrogenase (XDH) enzymes from Candida mogii cell extract. Under optimized conditions (pH 6.5 and 38°C), the XR and XDH activities were found to be 0.48 U/ml and 0.22 U/ml, respectively, resulting in an XR to XDH ratio of 2.2. Stability, cofactor specificity and kinetic parameters of the enzyme XR were also evaluated. XR activity remained stable for 3 h under 4 and 38°C and for 4 months of storage at −18°C. Studies on cofactor specificity showed that only NADPH-dependent XR was obtained under the cultivation conditions employed. The XR present in C. mogii extracts showed a superior K m value for xylose when compared with other yeast strains. Besides, this parameter was not modified after enzyme extraction by aqueous two-phase system.






Similar content being viewed by others
References
Andersson M, Adlercreutz P (1999) Evaluation of simple enzyme kinetics by response surface modeling. Biotechnol Tech 13:903–907
Chiang C, Knight SG (1960) Metabolism of d-xylose by moulds. Nature 188:79–81
Bolen PL, Roth KA, Freer SN (1986) Affinity purifications of aldose reductase and xylitol dehydrogenase from the xylose-fermenting yeast Pachysolen tannophilus. Appl Environ Microbiol 52:660–664
Boyaci IH (2005) A new approach for determination of enzyme kinetic constants using response surface methodology. Biochem Eng J 25:55–62
Cortez EV, Felipe MGA, Roberto IC, Pessoa Jr A, Vitolo M (2001) Extraction by reversed micelles of the intracellular enzyme xylose reductase. Appl Biochem Biotechnol 91/93:753–759
Ditzelmüller G, Kubicek CP, Wöhrer W, Röhr M (1984) Xylose metabolism in Pachysolen tannophilus: purification and properties of xylose reductase. Can J Microbiol 30:1330–1336
Gírio FM, Roseiro JC, Sá-Machado P, Duarte-Reis AR, Amaral-Collaço MT (1994) Effect of oxygen transfer rate on levels of key enzymes of xylose metabolism in Debaryomyces hansenii. Enzyme Microb Technol 16:1074–1078
Gírio FM, Pelica F, Amaral-Collaço MT (1996) Characterization of xylitol dehydrogenase from Debaryomyces hansenii. Appl Biochem Biotechnol 56:79–87
Granström T, Leisola M (2002) Controlled transient changes reveal differences in metabolite production in two Candida yeasts. Appl Microbiol Biotechnol 58:511–517
Ho NWY, Lin FP, Huang S, Andrews PC, Tsao GT (1990) Purification, characterization, and amino terminal sequence of xylose reductase from Candida shehatae. Enzyme Microb Technol 12:33–39
Hyvönen L, Koivistoinen P (1982) Food technological evaluation of xylitol. Adv Food Res 28:373–403
Kern M, Haltrich D, Nidetzky B, Kulbe DK (1997) Induction of aldose reductase and xylitol dehydrogenase activities in Candida tenuis CBS 4435. FEMS Microbiol Lett 149:31–37
Mayerhoff ZDVL, Roberto IC, Silva SS (1997) Xylitol production from rice straw hemicellulose hydrolysate using different yeast strains. Biotechnol Lett 19:407–409
Mayerhoff ZDVL, Roberto IC, Franco TT (2001) Activity of xylose reductase from Candida mogii grown in media containing different concentrations of rice straw hydrolysate. Appl Biochem Biotechnol 91–93:729–736
Mayerhoff ZDVL, Roberto IC, Franco TT (2004) Purification of xylose reductase from Candida mogii in aqueous two-phase systems. Biochem Eng J 18:217–223
Neuhauser W, Haltrich D, Kulbe KD, Nidetzky B (1997) NAD(P)H-dependent aldose reductase from the xylose-assimilating yeast Candida tenuis. Isolation, characterization and biochemical properties of the enzyme. Biochem J 326:683–692
Nidetzky B, Klimacek M, Mayr P (2001) Transient-state and steady-state kinetic studies of the mechanism of NADH-dependent aldehyde reduction catalyzed by xylose reductase from the yeast Candida tenuis. Biochemistry 40:10371–10381
Scopes RK (1994) Protein purification: principles and practice. Springer, Berlin Heidelberg New York
Takamizawa K, Uchida S, Hatsu M, Suzuki T, Kawai K (2000) Development of a xylitol biosensor composed of xylitol dehydrogenase and diaphorase. Can J Microbiol 46:350–357
Vandeska E, Kuzmanova S, Jeffries TW (1995) Xylitol formation and key enzyme activities in Candida boidinii under different oxygen transfer rates. J Ferment Bioeng 80:513–516
Woodyer R, Simurdiak M, van der Donk WA, Zhao H (2005) Heterologous expression, purification, and characterization of a highly active xylose reductase from Neurospora crassa. Appl Environ Microbiol 71:1642–1647
Acknowledgements
The authors gratefully acknowledge financial support from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) Brazil.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mayerhoff, Z.D.V.L., Roberto, I.C. & Franco, T.T. Response surface methodology as an approach to determine the optimal activities of xylose reductase and xylitol dehydrogenase enzymes from Candida Mogii . Appl Microbiol Biotechnol 70, 761–767 (2006). https://doi.org/10.1007/s00253-005-0304-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-005-0304-3