Abstract
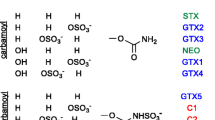
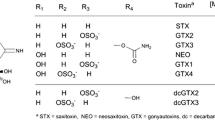
Polar marine toxins are more challenging to analyze by mass spectrometry-based methods than lipophilic marine toxins, which are now routinely measured in shellfish by multiclass reversed-phase liquid chromatography–tandem mass spectrometry (MS/MS) methods. Capillary electrophoresis (CE)–MS/MS is a technique that is well suited for the analysis of polar marine toxins, and has the potential of providing very high resolution separation. Here, we present a CE–MS/MS method developed, with use of a custom-built interface, for the sensitive multiclass analysis of paralytic shellfish toxins, tetrodotoxins, and domoic acid in seafood. A novel, highly acidic background electrolyte (5 M formic acid) was designed to maximize protonation of analytes and to allow a high degree of sample stacking to improve the limits of detection. The method was applied to a wide range of regulated and less common toxin analogues, and exhibited a high degree of selectivity between toxin isomers and matrix interference. The limits of detection in mussel tissue were 0.0052 mg/kg for tetrodotoxins, 0.160 mg/kg for domoic acid, and between 0.0018 and 0.120 mg/kg for paralytic shellfish toxins, all of which showed good linearity. Minimal ionization suppression was observed when the response from neat and mussel-matrix-matched standards was corrected with multiple internal standards. Analysis of shellfish matrix reference materials and spiked samples demonstrated good accuracy and precision. Finally, the method was transferred to a commercial CE–MS/MS system to demonstrate its widespread applicability for use in both R & D and routine regulatory settings. The approach of using a highly acidic background electrolyte is of broad interest, and can be considered generally applicable to simultaneous analysis of other classes of small, polar molecules with differing pKa values.

ᅟ









Similar content being viewed by others
References
Sanseverino I, Conduto D, Pozzoli L, Dobricic S, Lettieri T. Algal bloom and its economic impact. Report no. EUR 27905 EN. Ispra: Joint Research Centre; 2016.
Sobel J, Painter J. Illnesses caused by marine toxins. Clin Infect Dis. 2005;41:1290–6.
Starr M, Lair S, Michaud S, Scarratt M, Quilliam M, Lefaivre D, et al. Multispecies mass mortality of marine fauna linked to a toxic dinoflagellate bloom. PLoS One. 2017;12:e0176299.
Wells ML, Trainer VL, Smayda TJ, Karlson BS, Trick CG, Kudela RM, et al. Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae. 2015;49:68–93.
Impellizzeri G, Mangiafico S, Oriente G, Piattelli M, Sciuto S, Fattorusso E, et al. Constituents of red algae. I. Amino acids and low-molecular-weight carbohydrates of some marine red algae. Phytochemistry. 1975;14:1549–57.
Bates SS, Bird CJ, de Freitas ASW, Foxall R, Gilgan M, Hanic LA, et al. Pennate diatom Nitzschia pungens as the primary source of domoic acid, a toxin in shellfish from Eastern Prince Edward Island. Canada. Can J Fish Aquat Sci. 1989;46:1203–15.
Quilliam MA, Wright JLC. The amnesic shellfish poisoning mystery. Anal Chem. 1989;61:1053A–60A.
Lefebvre KA, Kendrick PS, Ladiges W, Hiolski EM, Ferriss BE, Smith DR, et al. Chronic low-level exposure to the common seafood toxin domoic acid causes cognitive deficits in mice. Harmful Algae. 2017;64:20–9.
Wiese M, D'Agostino PM, Mihali TK, Moffitt MC, Neilan BA. Neurotoxic alkaloids: saxitoxin and its analogs. Mar Drugs. 2010;8:2185–211.
Bane V, Lehane M, Dikshit M, O'Riordan A, Furey A. Tetrodotoxin: chemistry, toxicity, source, distribution and detection. Toxins. 2014;6:693–755.
Chau R, Kalaitzis JA, Neilan BA. On the origins and biosynthesis of tetrodotoxin. Aquat Toxicol. 2011;104:61–72.
Andrew D, Turner AP. A Schofield, D N Lees, C Baker-Austin. Detection of the pufferfish toxin tetrodotoxin in European bivalves, England, 2013 to 2014. Euro Surveillance. 2015;20:1–7.
Vlamis A, Katikou P, Rodriguez I, Rey V, Alfonso A, Papazachariou A, et al. First detection of tetrodotoxin in Greek shellfish by UPLC-MS/MS potentially linked to the presence of the dinoflagellate Prorocentrum minimum. Toxins. 2015;7:1779–807.
Knutsen HK, Alexander J, Barregård L, Bignami M, Brüschweiler B, Ceccatelli S, et al. Risks for public health related to the presence of tetrodotoxin (TTX) and TTX analogues in marine bivalves and gastropods. EFSA J. 2017;15:1–65.
Turner AD, Dhanji-Rapkova M, Coates L, Bickerstaff L, Milligan S, O'Neill A, Faulkner D, McEneny H, Baker-Austin C, Lees DN, Algoet M. Detection of tetrodotoxin shellfish poisoning (TSP) toxins and causative factors in bivalve molluscs from the UK. Mar Drugs. 2017;15:277-294.
AOAC International. AOAC official method 959.08. Paralytic shellfish poison, biological method. Gaithersburg: AOAC International; 2000.
AOAC International. AOAC official method 2011.02. Quantitative determination of paralytic shellfish toxins in mussels, clams, oysters, and scallops using liquid chromatography and post-column oxidation with fluorescence detection. Gaithersburg: AOAC International; 2011.
AOAC International. AOAC official method 2005.06, Quantitative determination of paralytic shellfish poisoning toxins in shellfish using pre-chromatographic oxidation and liquid chromatography with fluorescence detection. Gaithersburg: AOAC International; 2005.
van den Top HJ, Gerssen A, McCarron P, van Egmond HP. Quantitative determination of marine lipophilic toxins in mussels, oysters and cockles using liquid chromatography-mass spectrometry: inter-laboratory validation study. Food Addit Contam A. 2011;28:1745–57.
McCarron P, Wright E, Quilliam MA. Liquid chromatography/mass spectrometry of domoic acid and lipophilic shellfish toxins with selected reaction monitoring and optional confirmation by library searching of product ion spectra. J AOAC Int. 2014;97:316–24.
Thomas KM, Beach DG, Reeves KL, Gibbs RS, Kerrin ES, McCarron P, et al. Hydrophilic interaction liquid chromatography-tandem mass spectrometry for quantitation of paralytic shellfish toxins: validation and application to reference materials. Anal Bioanal Chem. 2017;409:5675–87.
Boundy MJ, Selwood AI, Harwood DT, McNabb PS, Turner AD. Development of a sensitive and selective liquid chromatography–mass spectrometry method for high throughput analysis of paralytic shellfish toxins using graphitised carbon solid phase extraction. J Chromatogr A. 2015;1387:1–12.
Coleman R, Lemire SW, Bragg W, Garrett A, Ojeda-Torres G, Hamelin E, et al. Development and validation of a high-throughput online solid phase extraction - liquid chromatography - tandem mass spectrometry method for the detection of tetrodotoxin in human urine. Toxicon. 2016;119:64–71.
Turner AD, Boundy MJ, Rapkova MD. Development and single-laboratory validation of a liquid chromatography tandem mass spectrometry method for quantitation of tetrodotoxin in mussels and oysters. J AOAC Int. 2017;100:1469–82.
Shoji Y, Yotsu-Yamashita M, Miyazawa T, Yasumoto T. Electrospray ionization mass spectrometry of tetrodotoxin and its analogs: liquid chromatography/mass spectrometry, tandem mass spectrometry, and liquid chromatography/tandem mass spectrometry. Anal Biochem. 2001;290:10–7.
van de Riet J, Gibbs RS, Muggah PM, Rourke WA, JD MN, Quilliam MA, et al. Liquid chromatography post-column oxidation (PCOX) method for the determination of paralytic shellfish toxins in mussels, clams, oysters, and scallops: collaborative study. J AOAC Int. 2011;94:1154–76.
Turner AD, Hatfield RG, Rapkova M, Higman W, Algoet M, Suarez-Isla BA, et al. Comparison of AOAC 2005.06 LC official method with other methodologies for the quantitation of paralytic shellfish poisoning toxins in UK shellfish species. Anal Bioanal Chem. 2011;399:1257–70.
de la Iglesia P, Barber E, Gimenez G, Rodriguez-Velasco ML, Villar-Gonzalez A, Diogene J. High-throughput analysis of amnesic shellfish poisoning toxins in shellfish by ultra-performance rapid resolution LC-MS/MS. J AOAC Int. 2011;94:555–64.
Beach DG, Walsh CM, Cantrell P, Rourke W, O'Brien S, Reeves K, et al. Laser ablation electrospray ionization high-resolution mass spectrometry for regulatory screening of domoic acid in shellfish. Rapid Commun Mass Spectrom. 2016;30:2379–87.
Olivares JA, Nguyen NT, Yonker CR, Smith RD. On-line mass spectrometric detection for capillary zone electrophoresis. Anal Chem. 1987;59:1232–6.
Smith RD, Udseth HR, Loo JA, Wright BW, Ross GA. Sample introduction and separation in capillary electrophoresis, and combination with mass spectrometric detection. Talanta. 1989;36:161–9.
Thibault P, Pleasance S, Laycock MV. Analysis of paralytic shellfish poisons by capillary electrophoresis. J Chromatogr. 1991;542:483–501.
Nguyen A-L, Luong JH, Masson C. Capillary electrophoresis for detection and quantitation of domoic acid in mussels. Anal Lett. 1990;23:1621–34.
Locke SJ, Thibault P. Improvement in detection limits for the determination of paralytic shellfish poisoning toxins in shellfish tissues using capillary electrophoresis/electrospray mass spectrometry and discontinuous buffer systems. Anal Chem. 1994;66:3436–46.
Pleasance S, Ayer SW, Laycock MV, Thibault P. Ionspray mass spectrometry of marine toxins. III. Analysis of paralytic shellfish poisoning toxins by flow-injection analysis, liquid chromatography/mass spectrometry and capillary electrophoresis/mass spectrometry. Rapid Commun Mass Spectrom. 1992;6:14–24.
Pleasance S, Thibault P, Kelly J. Comparison of liquid-junction and coaxial interfaces for capillary electrophoresis-mass spectrometry with application to compounds of concern to the aquaculture industry. J Chromatogr. 1992;591:325–39.
Keyon AS, Guijt RM, Gaspar A, Kazarian AA, Nesterenko PN, Bolch CJ, et al. Capillary electrophoresis for the analysis of paralytic shellfish poisoning toxins in shellfish: comparison of detection methods. Electrophoresis. 2014;35:1496–503.
Gago-Martinez A, Rodriguez-Vazquez JA, Thibault P, Quilliam MA. Simultaneous occurrence of diarrhetic and paralytic shellfish poisoning toxins in Spanish mussels in 1993. Nat Toxins. 1996;4:72–9.
Zhao J-Y, Thibault P, Quilliam MA. Analysis of domoic acid and isomers in seafood by capillary electrophoresis. Electrophoresis. 1997;18:268–76.
Kerrin ES, White RL, Quilliam MA. Quantitative determination of the neurotoxin beta-N-methylamino-L-alanine (BMAA) by capillary electrophoresis-tandem mass spectrometry. Anal Bioanal Chem. 2017;409:1481–91.
Burton IW, Quilliam MA, Walter JA. Quantitative 1H NMR with external standards: use in preparation of calibration solutions for algal toxins and other natural products. Anal Chem. 2005;77:3123–31.
Reeves K, Thomas K, Quilliam MA. A mussel tissue certified reference material for paralytic shellfish poisoning toxins. In: Henshilwood K, Deegan B, McMahon T, Cusack C, Keaveney S, Silke J, et al., editors. Proceedings of the 5th international conference on molluscan shellfish safety. Galway: Marine Institute; 2006. p. 116-22.
Ding L, Qiu J, Li A. Proposed biotransformation pathways for new metabolites of paralytic shellfish toxins based on field and experimental mussel samples. J Agric Food Chem. 2017;65:5494–502.
Rossi R, Arace O, Buonomo MG, Capozzo D, Castellano V, Imbimbo S, et al. Monitoring the presence of domoic acid in the production areas of bivalve molluscs. Ital J Food Saf. 2016;5:5706.
Saito T, Miura N, Namera A, Ota S, Miyazaki S, Inokuchi S. A rapid sample preparation procedure using MonoSpin CBA and amide columns for tetrodotoxin detection in serum and urine using LC–MS/MS analysis. Chromatographia. 2014;77:687–93.
Shimizu Y, Hsu C, Fallon WE, Oshima Y, Miura I, Nakanishi K. Structure of neosaxitoxin. J Am Chem Soc. 1981;100:6791–3.
Shimizu Y, Hsu C, Genenah A. Structure of saxitoxin in solutions and stereochemistry of dihydrosaxitoxins. J Am Chem Soc. 1981;103:605–9.
Rogers RS, Rapoport H. The pKa's of saxitoxin. J Am Chem Soc. 1980;102:7335–9.
Goto T, Kishi Y, Takahashi S, Hirata Y. Tetrodotoxin. Tetrahedron. 1965;21:2059–88.
Walter JA, Leek DM, Falk M. NMR study of the protonation of domoic acid. Can J Chem. 1992;70:1156–61.
Selwood AI, Waugh C, Harwood DT, Rhodes LL, Reeve J, Sim J, et al. Acute toxicities of the saxitoxin congeners gonyautoxin 5, gonyautoxin 6, decarbamoyl gonyautoxin 2&3, decarbamoyl neosaxitoxin, C-1&2 and C-3&4 to mice by various routes of administration. Toxins. 2017;9:73–80.
Osbourn DM, Weiss DJ, Lunte CE. On-line preconcentration methods for capillary electrophoresis. Electrophoresis. 2000;21:2768–79.
Beach DG, Kerrin ES, Giddings SD, Quilliam MA, McCarron P. Differential mobility-mass spectrometry double spike isotope dilution study of release of b-methylaminoalanine and proteinogenic amino acids during biological sample hydrolysis. Sci Rep. 2018;8:117.
Landsberg JH, Hall S, Johannessen JN, White KD, Conrad SM, Abbott JP, et al. Saxitoxin puffer fish poisoning in the United States, with the first report of Pyrodinium bahamense as the putative toxin source. Environ Health Perspect. 2006;114:1502–7.
V. González-Ruiz, N. Drouin, E. Reginato, S. Rudaz, J. Schappler. zeecalc 1.0. https://epgl.unige.ch/labs/fanal/zeecalc. Accessed 5 April, 2018.
Acknowledgements
The authors thank Michael Boundy from the Cawthron Institute and Aifeng Li from Ocean University of China for generously providing the sea slug and pufferfish tissue, respectively. Thanks are due to Jean-Francois Roy, Sylvie Larocque, and Dat Phan of Agilent Technologies for arranging access to the commercial capillary electrophoresis–tandem mass spectrometry system and for technical assistance with the related experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Published in the topical collection Food Safety Analysis with guest editor Steven J. Lehotay.
Electronic supplementary material
ESM 1
(PDF 420 kb)
Rights and permissions
About this article
Cite this article
Beach, D.G., Kerrin, E.S., Thomas, K. et al. Capillary electrophoresis–tandem mass spectrometry for multiclass analysis of polar marine toxins. Anal Bioanal Chem 410, 5405–5420 (2018). https://doi.org/10.1007/s00216-018-1089-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-1089-4