Abstract
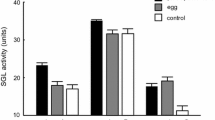
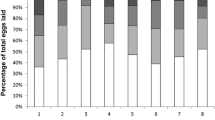
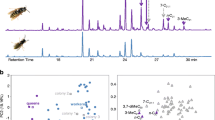
In social insects, the caste systems are based on reproductive division of labor; queens specialize in reproduction and workers primarily maintain the colony. Recently, a volatile pheromone containing n-butyl-n-butyrate and 2-methyl-1-butanol was identified as a termite queen pheromone that inhibits the differentiation of female neotenic reproductives (secondary queens). Although this volatile inhibitory pheromone regulates caste differentiation directly, the method by which it reaches members without direct contact with the queen in large colonies is not well understood. Therefore, additional mechanisms of indirect communication must exist, such as worker-mediated queen signal transport. We found that workers exposed to female reproductives did not mediate queens’ inhibitory signal in a termite Reticulitermes speratus. The experiment assessed worker transfer from direct to indirect contact groups and determined that the differentiation of new female reproductives in the indirect contact groups was not influenced by the direct contact groups, whereas direct contact with functional female reproductives and artificial queen pheromone did suppress neotenic differentiation. This suggests that worker transfer of the queen signal is unlikely and that for colony-wide inhibition direct contact by the majority of infertile members with reproductives or eggs, which emit the same volatiles as female reproductives, is necessary within a certain time interval.


Similar content being viewed by others
References
Afzal M. 1983. Radioisotope studies of trophallaxis in the drywood termite Bifiditermes beesoni (Gardner) (Isoptera). I: effect of group size on the rate of food exchange. Mater. Org. 18: 51-63
Arnold G., Leconte Y., Trouiller J., Hervet H., Chappe B. and Masson C. 1994. Inhibition of worker honeybee ovaries development by a mixture of fatty-acid esters from larvae. C.R. Acad. Sci. Paris Sci. Vie 317: 511-515
Brian M. and Rigby C. 1978. The trophic eggs of Myrmica rubra L. Insect. Soc. 25: 89-110.
Butler C.G., Callow R.K. and Johnston N.C. 1959. Extraction and purification of ‘queen substance’ from queen bees. Nature 184: 1871
Endler A., Liebig J., Schmitt T., Parker J.E., Jones G.R., Schreier P. and Hölldobler B. 2004. Surface hydrocarbons of queen eggs regulate worker reproduction in a social insect. Proc. Natl Acad. Sci. USA 101: 2945-2950. doi:10.1073/pnas.0308447101
Fletcher D.J.C. and Ross K.G. 1985. Regulation of reproduction in eusocial Hymenoptera. Annu. Rev. Entomol. 30: 319-343. doi:doi:10.1146/annurev.en.30.010185.001535
Fujita A. and Watanabe H. 2010. Inconspicuous matured males of worker form are produced in orphaned colonies of Reticulitermes speratus (Isoptera: Rhinotermitidae) and participate in reproduction. J. Insect Physiol. 56: 1510-1515
Hanus R., Vrkoslav V., Hrdý I., Cvačka J. and Šobotník J. 2010. Beyond cuticular hydrocarbons: evidence of proteinaceous secretion specific to termite kings and queens. Proc. R. Soc. Lond. B 277: 995-1002. doi:10.1098/rspb.2009.1857
Holman L., Jørgensen C.G., Nielsen J. and d’Ettorre P. 2010. Identification of an ant queen pheromone regulating worker sterility. Proc. R. Soc. Lond. B 277: 3793-3800. doi:10.1098/rspb.2010.0984
Jay S.C. 1970. The effect of various combinations of immature queen and worker bees on the ovary development of worker honeybees. Can. J. Zool. 48: 169-173
Kawabata S. and Tsuji K. 2005. The policing behavior ‘immobilization’ towards ovary-developed workers in the ant, Diacamma sp. from Japan. Insect. Soc. 52: 89-95. doi:10.1007/s00040-004-0778-5
Keller L. and Nonacs P. 1993. The role of queen pheromones in social insects: queen control or queen signal? Anim. Behav. 45: 787-794
Kikuchi T., Suwabe M. and Tsuji K. 2010. Durability of information concerning the presence of a gamergate in Diacamma sp. from Japan. Physiol. Entomol. 35: 93-97. doi:10.1111/j.1365-3032.2009.00710.x
Kindl J. and Hrdý I. 2005. Development of neotenics induced by a temporary absence of functional reproductives in Kalotermes flavicollis (Isoptera: Kalotermitidae). Eur. J. Entomol. 102: 307-311
Lenz M. 1994. Food resources, colony growth and caste development in wood-feeding termites. In: Nourishment and Evolution in Insect Societies (Hunt J.H. and Nalepa C.A., Eds), Westview Press, Boulder, Colorado, pp 159-210
Liebig J., Eliyahu D. and Brent C.S. 2009. Cuticular hydrocarbon profiles indicate reproductive status in the termite Zootermopsis nevadensis. Behav. Ecol. Sociobiol. 63: 1799-1807. doi:10.1007/s00265-009-0807-5
Liebig J., Monnin T. and Turillazzi S. 2005. Direct assessment of queen quality and lack of worker suppression in a paper wasp. Proc. R. Soc. Lond. B 272: 1339-1344. doi:10.1098/rspb.2005.3073
Liebig J., Peeters C. and Hölldobler B. 1999. Worker policing limits the number of reproductives in a ponerine ant. Proc. R. Soc. Lond. B 266: 1865-1870
Lüscher M. 1960. Hormonal control of caste differentiation in termites. Annals N.Y. Acad. Sci. 89: 549-563
Lüscher M. 1952. Die Produktion und Elimination von Ersatzgeschlechtstieren bei der Termite Kalotermes flavicollis Fabr. Z. Vergl. Physiol. 34: 123-141
Markin G.P. 1970. Food distribution within laboratory colonies of the Argentine ant, Iridomyrmex humilis (Mayr). Insect. Soc. 17: 127-158
Matsuura K. 2010. Sexual and asexual reproduction in termites. In: Biology of Termites: a Modern Synthesis (Bignell D.E., Roisin Y. and Lo N., Eds), Springer, Dordrecht, pp 255-277
Matsuura K., Himuro C., Yokoi T., Yamamoto Y., Vargo E.L. and Keller L. 2010. Identification of a pheromone regulating caste differentiation in termites. Proc. Natl Acad. Sci. USA 107: 12963-12968. doi:10.1073/pnas.1004675107
Matsuura K., Kobayashi N. and Yashiro T. 2007. Seasonal patterns of egg production in field colonies of the termite Reticulitermes speratus (Isoptera: Rhinotermitidae). Pop. Ecol. 49: 179-183. doi:10.1007/s10144-006-0030-4
Matsuura K., Vargo E.L., Kawatsu K., Labadie P.E., Nakano H., Yashiro T. and Tsuji K. 2009. Queen succession through asexual reproduction in termites. Science 323: 1687. doi:10.1126/science.1169702
Mohammedi A., Paris A., Crauser D. and Le Conte Y. 1998. Effect of aliphatic esters on ovary development of queenless bees (Apis mellifera L.). Naturwissenschaften 85: 455-458. doi:10.1007/s001140050531
Naumann K., Winston M.L., Slessor K.N., Prestwich G.D. and Webster F.X. 1991. Production and transmission of honey bee queen (Apis mellifera L.) mandibular gland pheromone. Behav. Ecol. Sociobiol. 29: 321-332. doi:10.1007/bf00165956
Pamilo P. 1991. Evolution of colony characteristics in social insects. II. Number of reproductive individuals. Am. Nat. 138: 412-433. doi:doi:10.1086/285224
Pettis J.S., Higo H.A., Pankiw T. and Winston M.L. 1997. Queen rearing suppression in the honey bee evidence for a fecundity signal. Insect. Soc. 44: 311-322
Ratnieks F.L.W. 1988. Reproductive harmony via mutual policing by workers in eusocial Hymenoptera. Am. Nat. 132: 217-236
Rosengaus R.B., Traniello J.F.A. and Levy C.K. 1986. Social transfer, elimination, and biological half-life of gamma-emitting radionuclides in the termite Reticulitermes flavipes Kol. J. Appl. Entomol. 101: 287-294
Seeley T.D. 1979. Queen substance dispersal by messenger workers in honeybee colonies. Behav. Ecol. Sociobiol. 5: 391-415. doi:10.1007/bf00292527
Shellman-Reeve J.S. 1997. The spectrum of eusociality in termites. In: The Evolution of Social Behaviour in Insects and Arachnids (Choe J.C. and Crespi B.J., Eds), Cambridge University Press, Cambridge, UK, pp 52-93
Springhetti A. 1972. I reali nella differenziazione delle caste di Kalotermes flavicollis (Fabr.) (Isoptera). Boll. Zool. 39: 83-87
Stuart A.M. 1979. The determination and regulation of the neotenic reproductive caste in the lower termites (Isoptera): with special reference to the genus Zootermopsis (Hagen). Sociobiology 4: 223-237
Suárez M.E. and Thorne B.L. 2000. Rate, amount, and distribution pattern of alimentary fluid transfer via trophallaxis in three species of termites (Isoptera: Rhinotermitidae, Termopsidae). Ann. Entomol. Soc. Am. 93: 145-155
Traniello J.F.A. 1977. Recruitment behavior, orientation, and the organization of foraging in the carpenter ant Camponotus pennsylvanicus DeGeer (Hymenoptera: Formicidae). Behav. Ecol. Sociobiol. 2: 61-79. doi:10.1007/bf00299289
Traniello J.F.A., Rosengaus R.B. and Levy C.K. 1985. Single and double isotope labeling of social insect colonies: gamma-emitting radionuclides as individually identifiable markers. Entomol. Exper. Appl. 38: 87-92
Tsuji K., Egashira K. and Hölldobler B. 1999. Regulation of worker reproduction by direct physical contact in the ant Diacamma sp. from Japan. Anim. Behav. 58: 337-343
Tsunoda K., Matsuoka H., Yoshimura T. and Tokoro M. 1999. Foraging populations and territories of Reticulitermes speratus (Isoptera: Rhinotermitidae). J. Econ. Entomol. 92: 604-609
Acknowledgments
We thank C. Himuro, T. Yokoi, and T. Yashiro for research assistance, and E.L. Vargo and L. Keller for helpful discussion. This work was supported by the Japan Society for the Promotion of Science (K.M.) and the Program for Promotion of Basic research Activities for Innovative Biosciences (K.M.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsuura, K., Yamamoto, Y. Workers do not mediate the inhibitory power of queens in a termite, Reticulitermes speratus (Isoptera, Rhinotermitidae). Insect. Soc. 58, 513–518 (2011). https://doi.org/10.1007/s00040-011-0170-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-011-0170-1