Abstract
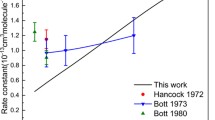
The three-dimensional problem of energy exchange between translational and rotational degrees of freedom in collisions of diatomic molecules with structureless particles is solved numerically within classical mechanics. The intermolecular interaction potentials were taken as exponential repulsions between the molecular atoms and the incident particles, while well-known experimental information [1] was used for the potential parameters. The results obtained were used for numerical Monte Carlo evaluation of the collision integral, determining the rotational relaxation time. Molecular nitrogen is chosen as an example. The results of calculation are in quite satisfactory agreement with experimental data.
Similar content being viewed by others
Literature cited
V. B. Leonas, “Studies of short-range intermolecular forces,” Usp. Fiz. Nauk,107, 29 (1972).
C. S. Wang-Chang, G. E. Uhlenbeck, and J. de Boer, “The heat conductivity of polyatomic gases,” in: Studies in Statistical Mechanics, Vol. 2 (1964).
I. S. Berezin and N. P. Zhidkov, Numerical Methods [in Russian], Vol. 1, Nauka, Moscow (1966).
C. F. Hansen and W. E. Peason, “Three-dimensional model of collision-induced vibration transitions in homonuclear diatomic molecules,” J. Chem. Phys.,53, No. 9 (1970).
E. H. Carnevale, C. Carey, and G. Larson, “Ultrasonic determination of rotational collision numbers and vibration relaxation times of polyatomic gases at high temperatures,” J. Chem. Phys.,47, No. 8 (1967).
J. Parker, “Rotational and vibrational relaxation in diatomic gases,” in: Gasdynamics and heat transfer in Chemical Reactions [Russian translation], IL, Moscow (1962).
Author information
Authors and Affiliations
Additional information
Translated from Zhurnal Prikladnoi Mekhaniki Tekhnicheskoi Fiziki, No. 1, pp. 13–18, January–February, 1975.
The authors are grateful to Yu. N. Belyaev and E. G. Kolesnichenko for useful discussion of the results.
Rights and permissions
About this article
Cite this article
Gerasimov, G.Y., Makarov, V.N. Theory of rotational relaxation in a diatomic gas. J Appl Mech Tech Phys 16, 8–12 (1975). https://doi.org/10.1007/BF00853532
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00853532