Summary
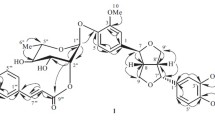
Two new esters of the new sesquiterpene alcohol juniferol — juniferin and juniferinin — have been isolated from the roots ofFerula juniperina. On the basis of the products of chemical transformations and spectral characteristics the structure of 2,5-dihydroxy-1,1,4,8-tetramethylcycloundeca-3,7-diene has been proposed for juniferol.
It has been shown that juniferin is 5-O-vanilloyljuniferol, and juniferinin is 2-O-acetyl-5-O-p-hydroxybenoyljuniferol.
Similar content being viewed by others
Literature cited
G. V. Sagitdinova, A. I. Saidkhodzhaeva, and G. K. Nikonov, Khim. Prirodn. Soedin., 547 (1976).
N. P. Damodaran and Sukh Dev, Tetrahedron,24, 4113 (1968).
B. R. Chhadra, R. S. Dhillon, M. S. Wadia, and P. S. Kalsi, Indian J. Chem.,13, 222 (1975).
K. Jano and T. Nishijama, Phytochemistry,13, 1207 (1974).
L. A. Smedman, E. Zavarin, and R. Teranishi, Phytochemistry,8, 1457 (1969).
S. Dev, Tetrahedron Lett., 12 (1959).
J. Poos, G. E. Arth, R. E. Beyler, and L. H. Sarett, J. Am. Chem. Soc.,75, 422 (1953).
A. I. Saidkhodzhaev and G. K. Nikonov, Khim. Prirodn. Soedin., 559 (1972).
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Uzbek SSR, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 6, pp. 790–796, November–December, 1977.
Rights and permissions
About this article
Cite this article
Sagitdinova, G.V., Saidkhodzhaev, A.I. The structures of juniferin and juniferinin. Chem Nat Compd 13, 665–670 (1977). https://doi.org/10.1007/BF00565514
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00565514