Abstract
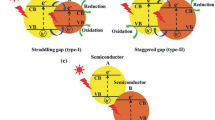
It has been shown on the example of semiconductor (CdS, TiO2) and metal complex (solvate complexes (V(III), Eu(III), Zr(IV)) photocatalysts that they can form a single photocatalytic system that is characterized by higher efficiency in the direction of the formation of hydrogen from alcohol-water solutions than the individual substances. Successful functioning of such a “hybrid” photocatalytic system is possible if the principle of energy correspondence is satisfied: the potential of the conduction zone of the semiconductor must be more negative than the redox potential of the Men+/Me(n−1)+ pair. In this case a photogenerated electron of the conduction zone is accepted by a metal ion Men+ and, thus, the electron-hole recombination process is suppressed. The resulting reduced form of the metal Me(n−1)+ in the presence of metallic palladium decomposes the protonated molecules of water with the formation of H2.
Similar content being viewed by others
Literature cited
K. I. Zamaraev and V. N. Parmon (editors), Photocatalytic Conversion of Solar Energy [in Russian], Part 1: Chemical and Biological Methods, Nauka, Novosibirsk (1985).
K. I. Zamaraev and V. N. Parmon (editors), Photocatalytic Conversion of Solar Energy [in Russian], Part 2: Molecular Systems for Decomposition of Water, Nauka, Novosibirsk (1985).
M. Grettsel (editor), Energy Resources through the Prism of Photochemistry and Catalysis [Russian translation], Mir, Moscow (1986).
N. Bühler, K. Meier, J.-F. Reber, “Photochemical hydrogen production with cadmium sulfide suspensions,” J. Phys. Chem., 88, No. 5, 3261–3268 (1984).
J. Calvert and J. Pitts, Photochemistry [Russian translation], Mir, Moscow (1986).
A. I. Kryukov, A. V. Korzhak, and S. Ya. Kuchmii, “Photocatalytic formation of hydrogen in alcoholic solutions of titanium tetrachloride,” Teor. Éksp. Khim., 20, No. 2, 169–177 (1984).
Z. A. Sinitsyna, Yu. I. Kiryukhin, E. V. Shepelin, and Kh. S. Bagdasar'yan, “Effect of conditions of preparation of cadmium sulfide on photochemical formation of hydrogen from methanol,” Khim. Vys. Énerg., 19, No. 2, 133–135 (1986).
B. V. Koryakin, T. S. Dzhabiev, and A. E. Shilov, “Photocatalytic formation of hydrogen from water-alcohol solutions of vanadium trichloride,” Dokl. Akad. Nauk SSSR, 229, No. 1, 128–130 (1976).
B. V. Koryakin and T. S. Dzhabiev, “Mechanism of the photogeneration of hydrogen from water-alcohol solutions of vanadium trichloride,” Izv. Akad. Nauk SSSR. Ser. Khim., No. 8, 1769–1773 (1980).
S. Ya. Kuchmii, A. V. Korzhak, Z. A. Tkachenko, andA. I. Kryukov, “Photochemical and dark reduction of chloride-alcoholate complexes of vanadium(V) in ethyl alcohol,” Teor. Éksp. Khim., 15, No. 4, 393–399 (1979).
D. Dobosh, Electrochemical Constants [in Russian], Mir, Moscow (1980).
J. Lilie, G. Beck, and A. Henglein, “Pulsradiolyse und Polarographie Halbstufenpotentiale fur die Oxidation und reduction von kurzlebigen organischen radikalen an der HgElektrode, Ber. Bunsenges. Phys. Chem., 75, No. 5, 458–465 (1971).
K. I. Zamaraev and V. N. Parmon, “Development of photocatalytic systems for the conversion of solar energy: Catalysts for the formation of hydrogen and oxygen from water,” Usp. Khim., 52, No. 9, 1433–1467 (1983).
E. Pelizzetti and M. Visca, “Bifunctional redox catalysts. Synthesis and activity in reactions of decomposition of water,” in: Energy Resources through the Prism of Photochemistry and Catalysis [Russian translation], Mir, Moscow (1986), pp. 288–322.
Author information
Authors and Affiliations
Additional information
Translated from Teoreticheskaya i Éksperimentalnaya Khimiya, Vol. 25, No. 4, pp. 452–459, July–August, 1989.
Rights and permissions
About this article
Cite this article
Kryukov, A.I., Kuchmii, S.Y., Korzhak, A.V. et al. “Hybrid” photocatalytic systems for production of molecular hydrogen. Theor Exp Chem 25, 420–426 (1990). https://doi.org/10.1007/BF00530435
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00530435