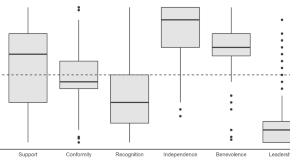
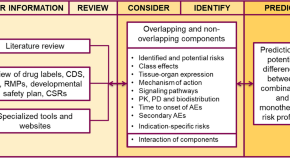
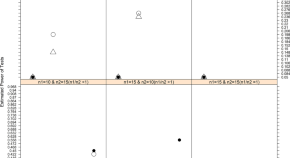
Meaningful Within-Patient Change in Subjective Total Sleep Time in Patients with Insomnia Disorder: An Analysis of the Sleep Diary Questionnaire Using Data from Open-Label and Phase III Clinical Trials
Authors (first, second and last of 5)

Collection