Collection
Regulatory Science
- Submission status
- Open
- Open for submission from
- 19 July 2023
- Submission deadline
- Ongoing
A collection of articles that are relevant to regulatory professionals.
Articles (23 in this collection)
-

-
“Regulatory Sandboxes” Could Solve the Regulatory Problems Encountered in Europe and Arising from Innovation in Biological Medicinal Products
Authors
- Mathieu Guerriaud
- Content type: Current Opinion
- Published: 09 December 2023
- Pages: 19 - 23
-
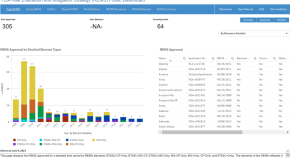
Correction to: The Risk Evaluation and Mitigation Strategy (REMS) Public Dashboard: Improving Transparency of Regulatory Activities
Authors
- Gita A. Toyserkani
- Joann H. Lee
- Esther H. Zhou
- Content type: Correction
- Published: 07 August 2023
- Pages: 355 - 355
-
The Risk Evaluation and Mitigation Strategy (REMS) Public Dashboard: Improving Transparency of Regulatory Activities
Authors
- Gita A. Toyserkani
- Joann H. Lee
- Esther H. Zhou
- Content type: Leading Article
- Published: 08 July 2023
- Pages: 349 - 353
-
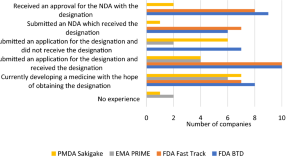
A Survey of Industry Perceptions of Facilitated Regulatory Pathways in Drug Development in Australia
Authors (first, second and last of 4)
- Alina Yoffe
- Johnson Liu
- Orin Chisholm
- Content type: Original Research Article
- Open Access
- Published: 27 June 2023
- Pages: 385 - 394
-
Where is European Regulation 536/2014 Taking Us?
Authors
- Anthony W. Fox
- Content type: Current Opinion
- Published: 24 June 2023
- Pages: 343 - 347
-
Current Status and Challenges of Pharmacovigilance of Traditional Medicines in French-Speaking West African (UEMOA) Countries
Authors (first, second and last of 5)
- Kampadilemba Ouoba
- Hélène Lehmann
- Rasmané Semdé
- Content type: Original Research Article
- Published: 22 May 2023
- Pages: 305 - 318
-
Clinical and Regulatory Challenges and Opportunities for Monoclonal Antibodies in Low- and Middle-Income Countries: Lessons from COVID-19 and Beyond
Authors (first, second and last of 4)
- Lisa Gieber
- Vincent Muturi-Kioi
- Ayesha Sitlani
- Content type: Current Opinion
- Published: 28 April 2023
- Pages: 203 - 214
-
Evaluation of Risk-Based Approaches to the Registration of Medicines: Current Status Among African Regulatory Authorities
Authors (first, second and last of 6)
- Neil McAuslane
- Magda Bujar
- Stuart Walker
- Content type: Original Research Article
- Published: 19 April 2023
- Pages: 251 - 260
-
The Implementation of a Risk-Based Assessment Approach by the South African Health Products Regulatory Authority (SAHPRA)
Authors
- Lerato Moeti
- Madira Litedu
- Jacques Joubert
- Content type: Original Research Article
- Open Access
- Published: 04 January 2023
- Pages: 71 - 91
-
Are the Current Processes and Regulations Fit for Purpose to Deliver Novel Therapies During Pandemics? A Perspective on COVID-19 from the UK
Authors
- Rajmeet Jandu
- Carl Naraynassamy
- Nadarajah Sreeharan
- Content type: Current Opinion
- Published: 28 July 2022
- Pages: 275 - 278
-
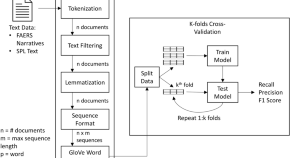
Utilizing Deep Learning for Detecting Adverse Drug Events in Structured and Unstructured Regulatory Drug Data Sets
Authors
- Benjamin M. Knisely
- Qais Hatim
- Monifa Vaughn-Cooke
- Content type: Original Research Article
- Published: 24 July 2022
- Pages: 307 - 317
-
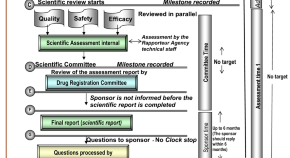
Evaluation of the Performance of the Gulf Cooperation Council Centralised Regulatory Review Process: Strategies to Improve Product Authorisation Efficiency and Quality
Authors (first, second and last of 7)
- Hajed M. Hashan
- Sarah K. Al-Muteb
- Stuart R. Walker
- Content type: Original Research Article
- Open Access
- Published: 29 June 2022
- Pages: 223 - 231
-
Barriers to Chimeric Antigen Receptor T-Cell (CAR-T) Therapies in Clinical Practice
Authors (first, second and last of 6)
- Ajeet Gajra
- Abigail Zalenski
- Ankit Kansagra
- Content type: Current Opinion
- Open Access
- Published: 07 June 2022
- Pages: 163 - 171
-
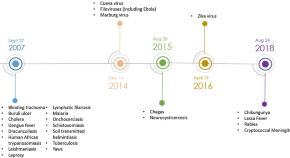
The Impact of the Priority Review Voucher on Research and Development for Tropical Diseases
Authors (first, second and last of 4)
- Celine Aerts
- Eliana Barrenho
- Elisa Sicuri
- Content type: Original Research Article
- Open Access
- Published: 19 May 2022
- Pages: 189 - 197
-
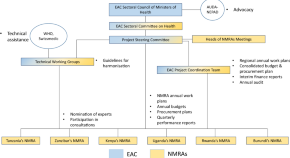
Medicines Regulatory Science Expertise in Africa: Workforce Capacity Development and Harmonisation Activities Towards the Establishment of the African Medicines Agency
Authors
- Bakani Mark Ncube
- Admire Dube
- Kim Ward
- Content type: Current Opinion
- Published: 05 April 2022
- Pages: 83 - 97
-
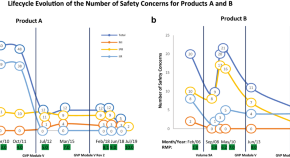
Impact of Changing Regulations and the Dynamic Nature of European Risk Management Plans for Human Medicines on the Lifecycle of Safety Concerns
Authors
- Julia Elvira Johanna Holm
- Jan Gustav Ruppert
- Samuel David Ramsden
- Content type: Original Research Article
- Open Access
- Published: 31 January 2022
- Pages: 33 - 46
-
How the Global COVID-19 Pandemic Brought Drug and Vaccine Development into the Public Mainstream
Authors
- Theodore J. Witek Jr.
- Content type: Leading Article
- Published: 27 September 2021
- Pages: 287 - 295
-
Evaluation of Drug Lags in Development Initiation, New Drug Application and Approval Between Japan and the USA and the Impact of Local Versus Multi-regional Clinical Trials
Authors
- Satoshi Ushijima
- Naoki Matsumaru
- Katsura Tsukamoto
- Content type: Original Research Article
- Published: 22 July 2021
- Pages: 253 - 260

-
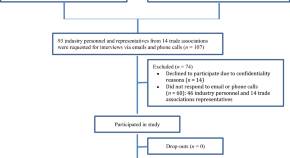
Challenges Faced by the Biopharmaceutical Industry in the Development and Marketing Authorization of Biosimilar Medicines in BRICS-TM Countries: An Exploratory Study
Authors (first, second and last of 5)
- Hasumati Rahalkar
- Alan Sheppard
- Sam Salek
- Content type: Original Research Article
- Open Access
- Published: 22 July 2021
- Pages: 235 - 251
-
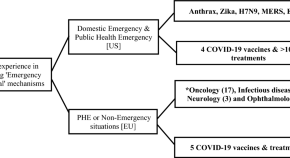
The Emergency Use Authorization of Pharmaceuticals: History and Utility During the COVID-19 Pandemic
Authors
- Allan Tran
- Theodore J. Witek Jr
- Content type: Leading Article
- Published: 28 August 2021
- Pages: 203 - 213
-
Regulatory Approval Scenario of Biosimilars in Pediatric Patients in the United States and European Union
Authors (first, second and last of 4)
- Swetha Radhakrishna
- Meghana Dahiya
- Vinu Jose
- Content type: Review Article
- Published: 17 February 2021
- Pages: 93 - 112

-
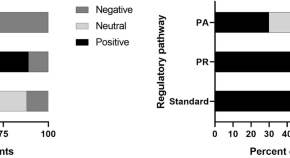
The Qualitative Value of Facilitated Regulatory Pathways in Europe, USA, and Japan: Benefits, Barriers to Utilization, and Suggested Solutions
Authors
- Magdalena Bujar
- Neil McAuslane
- Lawrence Liberti
- Content type: Original Research Article
- Open Access
- Published: 04 February 2021
- Pages: 113 - 122