Abstract
The epidermal growth factor receptor (EGFR) is an important cell surface receptor in normal physiology and disease. Recent work has shown that EGF-gold nanoparticle conjugates can influence cell behaviour, but the underlying mechanism at the receptor quaternary structural level remains poorly understood.
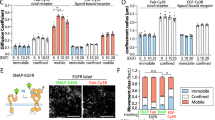
In the present work, the cluster density and cluster size of activated (phosphorylated) EGFR clusters in HeLa cells were determined with photobleaching image correlation spectroscopy. EGFR activation was probed via immunofluorescence-detected phosphorylation of tyrosines (pY-mAb) located in the kinase domain of EGFR (Y845) and at the EGFR cytoplasmic tail (Y1173). Cell activation was probed via nuclear extracellular-regulated kinase (ERK) phosphorylation. The cluster size of activated EGFR was 1.3–2.4 pY-mAb/cluster in unstimulated HeLa cells. EGF or nanorod treatment led to an increase in EGFR oligomers containing multiple phosphotyrosines (>2 phosphotyrosines per EGFR oligomer, average cluster size range = 3–5 pY-mAb/cluster) which paralleled increases in nuclear p-ERK. In contrast, EGF-nanorods decreased the contribution from higher-order phospho-clusters and decreased nuclear p-ERK relative to the nanorod control. These studies provide direct evidence that targeted nanotechnology can manipulate receptor organization and lead to changes in receptor activation and subsequent signalling processes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abulrob A, Lu Z, Baumann E et al (2010) Nanoscale imaging of epidermal growth factor receptor clustering: effects of inhibitors. J Biol Chem 285:3145–3156
Berkers JA, van Bergen en Henegouwen PM, Boonstra J (1991) Three classes of epidermal growth factor receptors on HeLa cells. J Biol Chem 266:922–927
Ciccotosto GD, Kozer N, Chow TTY et al (2013) Aggregation distributions on cells determined by photobleaching image correlation spectroscopy. Biophys J 104:1056–1064
Clayton AHA, Orchard SG, Nice EC et al (2008) Predominance of activated EGFR higher-order oligomers on the cell surface. Growth Factors 26:316–324
Clayton AHA, Tavarnesi ML, Johns TG (2007) Unligated epidermal growth factor receptor forms higher order oligomers within microclusters on A431 cells that are sensitive to tyrosine kinase inhibitor binding. Biochemistry 46:4589–4597
Clayton AHA, Walker F, Orchard SG et al (2005) Ligand-induced dimer-tetramer transition during the activation of the cell surface epidermal growth factor receptor – a multidimensional microscopy analysis. J Biol Chem 280:30392–30399
Crow MJ, Seekell K, Ostrander JH et al (2011) Monitoring of receptor dimerization using plasmonic coupling of gold nanoparticles. ACS Nano 11:8532–8540
Delcassian D, Depoil D, Rudnicka D et al (2013) Nanoscale ligand spacing influences receptor triggering in T cells and NK cells. Nano Lett 13:5608–5614
Dreaden EC, Alkilany AM, Huang X et al (2012) The golden age: gold nanoparticles for biomedicine. Chem Soc Rev 41:2740–2779
Flynn JF, Wong C, Wu J (2009) Anti-EGFR therapy: mechanism and advances in clinical efficacy in breast cancer. J Oncol . https://doi.org/10.1155/2009/526963
Huang Y, Bharill S, Karandur D et al (2016) Molecular basis for multimerization in the activation of the epidermal growth factor receptor. elife 5:e14107. https://doi.org/10.7554/eLife.14107
Ichinose J, Muratab M, Yanagida T et al (2004) EGF signalling amplification induced by dynamic clustering of EGFR. Biochem Biophys Res Comm 324:1143–1149
Kolch W, Pitt A (2010) Functional proteomics to dissect tyrosine kinase signalling pathways in cancer. Nat Rev Cancer 10:618–629
Kovacs E, Das R, Wang Q et al (2015) Analysis of the role of the C-terminal tail in the regulation of the epidermal growth factor receptor. J Mol Cell Biol 35:3083–3102
Kozer N, Barua D, Orchard S et al (2013) Exploring higher-order EGFR oligomerisation and phosphorylation – a combined experimental and theoretical approach. Mol BioSyst 9:1849–1863
Kozer N, Barua D, Henderson C et al (2014) Recruitment of the adaptor protein Grb2 to EGFR tetramers. Biochemistry 53:2594–2604
Lajevardipour A, Chon JWM, Clayton AHA et al (2015) Complex aggregation distributions by photobleaching image correlation spectroscopy. AIMS Biophys 2:1–7
Needham SR, Roberts SK, Arkhipov A et al (2016) EGFR oligomerization organizes kinase-active dimers into competent signalling platforms. Nat Commun 7:13307. https://doi.org/10.1038/ncomms13307
Oyarzún DA, Bramhall JL, Lopez-Caamal F et al (2014) The EGFR demonstrates linear signal transmission. Integr Biol 6:736–742
Paviolo C, Chon JWM, Clayton AHA (2015) Inhibiting EGFR clustering and cell proliferation with gold nanoparticles. Small 11:1638–1643
Peckys DB, Baudoin J, Eder M et al (2013) Epidermal growth factor receptor subunit locations determined in hydrated cells with environmental scanning electron microscopy. Sci Rep 3:2626
Rauch J, Kolch W, Mahmoudi M (2012) Cell type-specific activation of AKT and ERK signaling pathways by small negatively-charged magnetic nanoparticles. Sci Rep 2:1–9
Shaw A, Lundin V, Petrova E et al (2014) Spatial control of membrane receptor function using ligand nanocalipers. Nat Meth 11:841–846
Tice DA, Biscardi JS, Nickles AL et al (1999) Mechanism of biological synergy between cellular Src and epidermal growth factor receptor. PNAS 96:1415–1420
Wang Y, Gao J, Guo X et al (2014) Regulation of EGFR nanocluster formation by ionic protein-lipid interaction. Cell Res 24:959–976
Wilson KJ, Gilmore JL, Foley J et al (2009) Functional selectivity of EGF family peptide growth factors: implications for cancer. Pharmacol Ther 122:1–8
Acknowledgements
AHAC and JWMC gratefully acknowledge the Australian Research Council for funding this research (Grant Number: DP130101475).
Disclosure
The author reports no conflicts of interest in this work.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Paviolo, C., Chon, J.W.M., Clayton, A.H.A. (2018). The Effect of Nanoparticles on the Cluster Size Distributions of Activated EGFR Measured with Photobleaching Image Correlation Spectroscopy. In: Chattopadhyay, K., Basu, S. (eds) Biochemical and Biophysical Roles of Cell Surface Molecules. Advances in Experimental Medicine and Biology, vol 1112. Springer, Singapore. https://doi.org/10.1007/978-981-13-3065-0_4
Download citation
DOI: https://doi.org/10.1007/978-981-13-3065-0_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-3064-3
Online ISBN: 978-981-13-3065-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)