Abstract
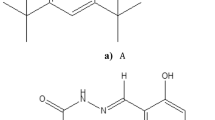
Protonated (or deuterated) all-trans, N-retyniliden, n-butylimine (NRB) with three different counter anions, (CF3)2CHO−, Cl− and BF4 − in various organic environments were used as model systems for protein-bound retinal in halorhodopsin. We have employed UV-Visible absorption and vibrational spectroscopies to study environmental and hydrogen bonding effects on the ground and excited state properties of NRB in order to gain more insight into the molecular interactions in the retinal binding pocket of the biological system.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Zinc, H., Baron, M. H., and Piart-Goypiron, A. (1995) Influence of the anion and solvent on the ion-pair stabitlity for (CH3)2CHCH=NH+CH(CH3)2 X− iminium salts (X−=Cl− or BF4−) Vibrational study, Spectrochim. Acta 51A, 457–469.
Lussicr, L. S., Sandorfy, C., Hoa Le-Thanh., and Vocelle, D. (1987) Effect of acids on the infrared spectra of Sniff basc of trans retinal, J. Phys. Chem. 91, 2282–2287.
Lopcz-Garriga, J. J., Babcock, G. T., and Harrison, J. F.(1986) Factors influencing the C=N stretching frequency in neutral protonated Schiffs base. J. Am. Chem. Soc. 108, 7241–7251.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1997 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Baron, M.H., Piart-Goypiron, A., Hildebrandt, P. (1997). Hydrogen Bonding and Environment Effects on N-Retinylidene N-Butyliminium Salts. In: Carmona, P., Navarro, R., Hernanz, A. (eds) Spectroscopy of Biological Molecules: Modern Trends. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-5622-6_55
Download citation
DOI: https://doi.org/10.1007/978-94-011-5622-6_55
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-6369-2
Online ISBN: 978-94-011-5622-6
eBook Packages: Springer Book Archive