Abstract
The effect of the North Atlantic Oscillation (NAO) on fisheries production has received attention in the last 2 decades, especially in relation to the inter-annual fluctuation of the important North Atlantic cod fisheries. Results of these studies show that the effects of NAO on cod stocks vary geographically, with opposite patterns in the western and eastern North Atlantic and that NAO affects mainly the recruitment success of cod, especially on heavily fished stocks. In the Mediterranean sea the number of studies is much more limited, due in part to the paucity of sufficiently long and reliable data series for Mediterranean fisheries. A recent study on the interannual variation of landings of the red shrimp Aristeus antennatus in Catalonia shows that the NAO explains a large part of the variability in population abundance with a lag of 2–3 years. New results presented here show that, at the scale of the entire Mediterranean, stocks of hake (Merluccius merluccius) respond to the NAO with different trends in the northwestern and southeastern stocks. Analysis of detailed life history data of this species in the Spanish Mediterranean coast allows to postulate the hypothesis that positive NAO years enhance hake fishery production through increasing the individual size of recruits, as well as the individual weight and abundance of adult hake. No effect on other demographic parameters such as recruitment strength or natural mortality could be demonstrated.
Similar content being viewed by others
Keywords
1 Introduction
Fisheries production depends on the interaction between human inputs (fishing effort) and the biomass of fish. While fishing effort may change over time due to socioeconomic factors, such as market interests, changing costs of effort or other reasons, fish biomass fluctuates over time due to environmental factors, such as biological productivity of its prey resources, or abiotic factors such as changes in temperature or salinity (Bakun, 1996; Cushing, 1996; Ottersen et al., 2004).
The abundance of marine fish is difficult to determine and many different methods have been developed over time (Hilborn and Walters, 1992). Briefly, fish abundance can be estimated from two basic sources: fisheries dependent data (commercial catches) or fisheries-independent data. In the first case, data series of catches are relatively easy to obtain because most countries maintain relatively long data bases on fish production by species (Evans and Grainger, 2001). However, to interpret catch data it is often necessary to know the evolution of fishing effort and catchability, which are often not available or difficult to estimate (Hilborn and Walters, 1992). Fisheries-independent programmes are more reliable but expensive to maintain and rarely have the long time spans required to assess the relationship between environmental variations and fish abundance (Sparre and Venema, 1998). In the European North Atlantic, there exist data series derived from scientific trawl surveys longer than 40 years (International Bottom Trawl Survey: http://datras.ices.dk), but in the Mediterranean, the only continuous fisheries monitoring programme (MEDITS: Mediterranean International Trawl Surveys: http://www.ifremer.fr/Medits_indices) to cover all EU member states started in 1994 and the data series is thus considerably shorter than in the Atlantic (Bertrand et al., 2002). In any case both types of data sources, commercial and trawl surveys, have advantages and disadvantages and it is convenient to use the two types of data concurrently when possible.
It is known that the changes in total biomass of a fishery target species can be due to individual changes in (one or more) individual demographic parameters. For instance, the abundance of a given year class may fluctuate from year to year due to environmental factors that cause enhanced survival (or conversely, increased mortality) of the individuals of that particular year class (Cushing, 1996; Myers, 2002; Watanabe et al., 2003). This is specially important in the first age class recruited to the fishery (known as recruitment), and several studies have shown the relationship between interannual fluctuations in recruitment and environmental variability (e.g. Mendelsohn and Mendo, 1987; Watanabe et al., 1996; Stige et al., 2006). Other key life history parameters affected by environmental variability are the individual growth rate, the natural mortality or the reproductive success (Hänninen et al., 1999; Jobling, 2002).
It is often difficult to determine the causes of the relationship between large scale climatic indices and fisheries due to the paucity of data, but also due to the complexity of the mechanisms involved (Cushing, 1996). In recent years, the usefulness of employing large scale climatic indices such as the NAO index as a proxy for complex space-time variations in ecological variables has been suggested (Hallett et al., 2004; Stenseth and Mysterud, 2005) and indeed the relationship between climatic indices and fisheries is well documented in non-Mediterranean settings, particularly in upwelling ecosystems (e.g. anchovy/sardine substitutions in the Pacific Kuroshio and Humboldt Current ecosystems and teleconnections among fish populations, reviewed in Alheit and Bakun, 2010).
The effects of NAO on North Atlantic fisheries has received attention in recent decades, especially in the last 10 years (Reid et al., 2001; Beaugrand, 2004), with studies focused particularly on cod (Gadus morhua, Mann and Drinkwater, 1994; Beaugrand et al., 2003; Stige et al., 2006). The results of these studies show that the NAO affects cod fisheries mainly by modulating the strength of cod recruitment with a time lag of 2 years (Brodziak and O’Brien, 2005). It has been shown also that the effect is stronger when spawning stock biomass (SSB) is low (Brander, 2005). Most significantly, some of these studies showed that the effect of the NAO varies spatially across North Atlantic cod stocks (synthesized in the model by Stige et al., 2006): while cod recruitment is enhanced in Newfoundland, Greenland, Iceland, northern Norway and the Faroes during positive NAO years, it is decreased in the eastern US, around the British Isles, in the North sea and in the Baltic sea (figure 4 in Stige et al., 2006).
In many cases the relationship between the NAO and fisheries production has been shown to be mediated by changes in the composition and abundance of zooplankton (Fromentin and Planque, 1996; Ottersen and Stenseth, 2001; Reid et al., 2003; Molinero et al., 2008).
In the Mediterranean, the relationship between the NAO and fisheries has received less attention, probably because data series are generally shorter than in Atlantic fisheries. Lloret et al. (2001) assessed the relationship between river runoff in 13 coastal fishery species of the Catalan coasts. They found that catches per unit of effort were positively correlated with the wind mixing index in the Gulf of Lions and the runoff of local rivers, which varied synchronously with the NAO index: negative NAO indices were associated with higher river runoff (probably because during negative NAO years precipitation increases in the NW Mediterranean, Mariotti et al., 2002). Maynou (2008) showed that the detrended landings of the deepwater red shrimp Aristeus antennatus were positively correlated with the NAO index with time lags of 2 and 3 years. An ecological mechanism to explain this correlation was produced, based on current knowledge of the biology of this species and the water mass dynamics in the study area (NW Mediterranean): positive NAO years are correlated with decreased rainfall in the NW Mediterranean sea, especially in the Gulf of Lions, the Ligurian sea and adjacent hinterland (Mariotti et al., 2002). Decreased rainfall is linked to enhanced vertical mixing of the water masses in the Gulf of Lions (Demirov and Pinardi, 2002; Jordi and Hameed, 2009), due to the saltier (hence, more dense) surface water mass in low rainfall years. Knowing that food supply in winter is key to the reproductive success of A. antennatus, because during this period females switch from their normal generalist diet to a high-energy content diet based on zooplankton (Cartes et al., 2008; Maynou, 2008) postulated that positive NAO years would enhance egg production and higher abundance of recruits to the fishery.
The objective of this contribution is to assess the effects of NAO in another important Mediterranean demersal fishery resource, the hake Merluccius merluccius at the scale of the entire Mediterranean basin, in order to investigate whether ecological mechanisms linking the NAO with fisheries productivity demonstrated in local areas (Catalan sea, Ligurian sea) can be extrapolated to wider areas.
2 Results
European hake (Merluccius merluccius) is an important resource of the Mediterranean demersal fishery, with catches fluctuating between 20 and 50,000 t in recent decades (FAO Fisheries Department, 2000). The biology of this species is well-known (e.g. the review by Oliver and Massutí, 1995; Recasens et al., 1998). Hake is distributed mainly over the continental shelf between 50 and 200 m depth, although occasional large individuals are caught down to 800 m. It is a heavily exploited species and catches are at present mainly composed of juvenile individuals of age classes 0–2. The recruitment period of hake varies across Mediterranean subareas and is relatively protracted, but it occurs mainly in early summer (June–July) in the NW Mediterranean (Recasens et al., 1998).
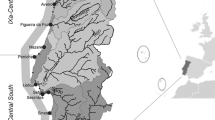
I analyzed the annual data series of hake catches for the period 1970–2005 in the seven geographical areas of the Mediterranean used by the FAO for fisheries statistics purposes (Fig. 1) based on the FISHSTAT database (FAO Fisheries Department, 2000).
As shown in Fig. 2, the data series of hake catches are very noisy, although some general patterns emerge. First, hake catches increased from 1970 until the early 1990s in most areas (Gulf of Lions, Adriatic, Ionian and Aegean). In these areas, and also in the Levant, hake production decreased since the early 1990s to the present due probably to overfishing (Lleonart, 2008). In the Balearic area no overall trend can be distinguished, but periods of high catches and low catches can be discerned, while in the Sardinia area hake catches have been decreasing since the beginning of the time series (Fig. 2).
Data series of hake (Merluccius merluccius) landings in the seven Mediterranean FAO fisheries divisions for the period 1970–2005 (data extracted from FISHSTAT, FAO Fisheries Department, 2000)
To analyze these data sets, a Generalized Additive Model (GAM) was set up for each statistical area, using year as the main effect (because it is expected that long-term trends in interannual variations in catches are mainly due to the observed increase in fishing effort, Lleonart, 2008) and the NAO index as additional effect with time lags of 1 and 2 years (nao1 and nao2). GAMs are a flexible class of statistical regression models which allow to assess linear or nonlinear relationships between a set of predictors (in our case, year, nao1 and nao2) and a dependent variable (in our case hake catch) (Wood, 2006):
I used smoothing splines to represent the possibly nonlinear effect of predictors and Generalized Cross Validation (GCV) to establish the optimal degree of the smooth terms. Model selection was achieved by means of Akaike’s Information Criteria to assess the goodness of fit (Wood, 2006).
The GAM models fitted for hake catch in the seven statistical subareas for the period 1970–2005 helped explain between 62% (Gulf of Lions) and 96% (Aegean sea) of the deviance in the data series (Table 1). These results show that most of the variability in the catch data series can be attributed to the year effect, which mostly reflects changes in fishing effort over time. More interestingly, the results of the GAM analysis show that the lagged NAO index explains between 2 and 8% of the deviance of the models and that some of the functional non-parametric terms are statistically significant (Table 1). The shape of these nonparametric terms can be examined in Fig. 3, which shows that hake stocks in the western and northern Mediterranean areas (Gulf of Lions, Balearic and Adriatic) are positively correlated with the NAO index at lags 1 and 2 years, while hake stocks in the eastern Mediterranean (Aegean and Levant) would be negatively correlated with these NAO indices (Fig. 3). Hake stocks in the central Mediterranean show weak correlations (Ionian) or of opposite sign (Sardinian).
To investigate the causes linking NAO with hake fisheries variability I analyzed the correlation between several life-history indicators derived from fisheries independent trawl surveys in the western Mediterranean (MEDITS surveys carried out along the continental coast of Spain, Bertrand et al., 2002) by means of autocorrelation function analysis (Venables and Ripley, 2002). The span of this data set (1994–2009) is shorter than the FAO production statistics, but includes a critical transitional period from positive values of the NAO index in the 1990s to negative or zero values later on. I extracted several demographic indicators from the MEDITS database (http://www.ifremer.fr/Medits_indices): abundance by age class, natural mortality by age class, total biomass, spawning stock biomass, and average individual length and weight (Table 2).
The results of autocorrelation function analysis (Fig. 4) show that the mean length of recruits (hake of age class 0) is positively correlated with NAO at lag 1, i.e. recruits are larger than average after positive NAO years. Also the abundance of adults (age classes 3–6) of hake is increased 1 and 2 years after positive NAO years, while the average weight of adults is positively correlated with NAO at lag 1. Despite this increase in abundance and mean weight of adults, no increase in spawning stock biomass could be shown in positive NAO years, probably because the biomass of the adult fraction of the population is much lower than the juvenile fraction (age classes 0–2) in this heavily exploited population. Natural mortality was not correlated to the NAO. These results show that in the Balearic subarea, and probably also in the Gulf of Lions and Adriatic, positive NAO years have a positive effect in at least 3 critical indicators of the hake life history (length of recruits, abundance of adults and weight of adults) suggesting that the positive effects on the catches shown earlier from the FAO production data series can be related to increase size and weight of the hake population and not, for instance, to decreased natural mortality or increased reproductive success, as in the red shrimp (Maynou, 2008).
3 Conclusions
The North Atlantic Oscillation has profound influences on fisheries productivity, mediated by complex dynamics of ocean processes and biological productivity, as shown in North Atlantic fish stocks (Ottersen and Stenseth, 2001; Reid et al., 2001, 2003; Stenseth and Mysterud, 2005; Stige et al., 2006), but also in the Mediterranean (Lloret et al., 2001; Maynou, 2008).
In fisheries research the influence of environmental variation on stock productivity may be difficult to detect due to the lack of long and consistent time series of the main oceanographic and ecological parameters at appropriate scales. Large-scale climatic indices such as the NAO are often better proxies of ecological processes than local weather variables, because they reduce the complexities of time and space variability in environmental phenomena to simple measures (Hallett et al., 2004; Stenseth and Mysterud, 2005), and are specially useful for fish abundance data where comparable environmental data sets are non existent or expensive to obtain (e.g. for deep-sea fishery resources, Maynou, 2008).
The influence of the NAO on fish stocks has been shown to vary geographically, with impacts of different sign in cod stocks of, broadly, the western Atlantic and the eastern Atlantic (Stige et al., 2006). In Mediterranean hake stocks, present results suggest also geographically varying impacts of the NAO. A N/S – W/E trend in the effects of NAO has been evidenced: in the N and W areas (Balearic sea, Gulf of Lions, Adriatic sea) the effect is close to linear and positive (enhanced hake catches at positive NAO values). In the E and S subareas (Aegean sea, Levantine sea) the effects is negative (enhanced catches at negative NAO values), while in the two central subareas (Sardinian seas and Ionian sea) the effect is weak but mainly positive. Focusing on the Balearic area, I used a shorter data set derived from fisheries independent surveys to investigate the possible biological causes of enhanced catches by NAO in this area. I showed that the positive relationship between NAO and catches of hake is related to the positive effect of NAO (with temporal lags of 1 or 2 years) on at least 3 critical indicators of the hake life history: length of recruits, abundance of adults and weight of adults. These results suggest that the positive effects of NAO on Mediterranean hake catches can be related to increased size and weight of the hake population and not, for instance, to decreased natural mortality nor increased reproductive success, as in the red shrimp (Maynou, 2008).
Although the effect of the NAO on fisheries of Mediterranean stocks has been shown in a few studies (Lloret et al., 2001; Maynou, 2008; present work for hake), further research is needed to investigate the relationships linking the North Atlantic Oscillation, Mediterranean oceanography and biological productivity. Although it is likely that the effect of the NAO on higher trophic levels (which constitute the fisheries target species) is mediated by the effects of NAO on zooplankton (Fromentin and Planque, 1996; Maynou, 2008; Molinero et al., 2008) the details of these relationships are often mere working hypotheses that need to be refined and tested.
References
Alheit J, Bakun A (2010) Population synchronies within and between ocean basins: apparent teleconnections and implications as to physical-biological linkage mechanisms. J Mar Syst 79:267–285
Bakun A (1996) Patterns in the ocean: ocean processes and marine population dynamics. California Sea Grant College System/Centro de Investigaciones Biológicas del Noroeste, La Paz, 323 pp
Beaugrand G (2004) The North Sea regime shift: evidence, mechanisms and consequences. Prog Oceanogr 60:245–262
Beaugrand G, Brander KM, Lindley JA, Souissi S, Reid PC (2003) Plankton effect on cod recruitment. Science 426:661–664
Bertrand JA, Gil de Sola L, Papaconstantinou C, Relini G, Souplet A (2002) The general specifications of the MEDITS surveys. Sci Marina 66:9–17
Brander K (2005) Cod recruitment is strongly affected by climate when stock biomass is low. ICES J Mar Sci 62:339–343
Brodziak J, O’Brien L (2005) Do environmental factors affect recruits per spawner anomalies of New England groundfish? ICES J Mar Sci 62:1394–1407
Cartes JE, Madurell T, Fanelli E, López-Jurado JL (2008) Dynamics of suprabenthos-zooplankton communities around the Balearic Islands (western Mediterranean): influence of environmental variables and effects on the biological cycle of Aristeus antennatus. J Mar Syst 71:316–335
Cushing DH (1996) Towards a science of recruitment in fish populations. International Ecology Institute, Oldendorf/Luhe
Demirov E, Pinardi N (2002) Simulation of the Mediterranean Sea circulation from 1979 to 1993: Part I. The interannual variability. J Mar Syst 33–34:23–50
Evans D, Grainger R (2001) Gathering data for resource monitoring and fisheries management. In: Hart PJB, Reynolds JD (eds) Handbook of fish biology and fisheries, vol 2, chapter 5. Blackwell Publishing, Oxford
FAO Fisheries Department (2000) FISHSTAT plus: universal software for fishery statistical time series. Version 2.30. FAO Fisheries Department, Fishery Information, Data and Statistics Unit
Fromentin JM, Planque B (1996) Calanus and environment in the eastern North Atlantic. II. Influence of the North Atlantic Oscillation on C. finmarchicus and C. helgolandicus. Mar Ecol Prog Ser 134:111–118
Hallett TB, Coulson T, Pilkington JG, Clutton-Brock TH, Pemberton JM, Grenfell BT (2004) Why large-scale climate indices seem to predict ecological processes better than local weather. Nature 430:71–75
Hänninen J, Vuorinen I, Hjelt P (1999) Climatic factors in the Atlantic control the oceanographic and ecological changes in the Baltic Sea. Limnol Oceanogr 45:703–710
Hilborn R, Walters CJ (1992) Quantitative fish stock assessment: choice, dynamics and uncertainty. Chapman and Hall, London
Jobling M (2002) Environmental factors and rates of development and growth. In: Hart PJB, Reynolds JD (eds) Handbook of fish biology and fisheries, vol 1, chapter 5. Blackwell Publishing, Oxford
Jordi A, Hameed S (2009) Influence of the Icelandic low on the variability of surface air temperature in the Gulf of Lion: implications for intermediate water formation. J Phys Oceanogr 39:3228–3232
Lleonart J (2008) Review of the state of Mediterranean and Black Sea fishery resources. In: Basurco B (ed) The Mediterranean fisheries sector. Options méditerranéennes, Série B: Études et recherches, n. 62. CIHEAM/FAO/GFCM, Rome, 179 pp
Lloret J, Lleonart J, Solé I, Fromentin J-M (2001) Fluctuations of landings and environmental conditions in the north-western Mediterranean Sea. Fish Oceanogr 10:33–50
Mann KH, Drinkwater KF (1994) Environmental influences on fish and shellfish production in the Northwest Atlantic. Environ Rev 2:16–32
Mariotti A, Struglia MV, Zeng N, Lau KM (2002) The hydrological cycle in the Mediterranean region and implications for the water budget of the Mediterranean Sea. J Clim 15:1674–1690
Maynou F (2008) Influence of the North Atlantic Oscillation on Mediterranean deep-sea shrimp landings. Clim Res 36:253–257
Mendelsohn R, Mendo J (1987) Exploratory analysis of anchoveta recruitment off Peru and related environmental series. In: Pauly D, Tsukuyama I (eds) The Peruvian anchoveta and its upwelling ecosystem: three decades of change. ICLARM studies and reviews, Instituto del Mar de Peru, Callao (Peru), vol 15. pp 109–116
Molinero JC, Ibanez F, Souissi S, Buecher E, Dallot S, Nival P (2008) Climate control on the long-term anomalous changes of zooplankton communities in the northwestern Mediterranean. Glob Change Biol 14:11–26
Myers R (2002) Recruitment: understanding density-dependence in fish populations. In: Hart PJB, Reynolds JD (eds) Handbook of fish biology and fisheries, vol 1, chapter 6. Blackwell Publishing, London
Oliver P, Massutí E (1995) Biology and fisheries of western Mediterranean hake (M. merluccius). In: Alheit J, Pitcher TJ (eds) Hake: biology, fisheries and markets. Chapman and Hall, London, pp 181–202
Ottersen G, Alheit J, Drinkwater K, Friedland K, Hagen E, Stenseth NC (2004) The responses of fish populations to ocean climate fluctuations. In: Stenseth NC, Ottersen G, Hurrell J, Belgrano A (eds) Marine ecosystems and climate variation: the North Atlantic. Oxford University Press, Oxford, pp 73–94
Ottersen G, Stenseth NC (2001) Atlantic climate governs oceanographic and ecological variability in the Barents Sea. Limnol Oceanogr 46:1774–1780
Recasens L, Lombarte A, Morales-Nin B, Torres GJ (1998) Spatiotemporal variation in the population structure of the European hake in the NW Mediterranean. J Fish Biol 53:387–401
Reid PC, Borges MF, Svendsen E (2001) A regime shift in the North Sea circa 1988 linked to changes in the North Sea horse mackerel fishery. Fish Res 50:163–171
Reid PC, Edwards M, Beaugrand G, Skogen M, Stevens D (2003) Periodic changes in the zooplankton of the North Sea during the 20th century linked to oceanic inflow. Fish Oceanogr 12:260–269
Sparre P, Venema SC (1998) Introduction to tropical fish stock assessment – Part 1: Manual. FAO Fisheries Technical Paper 306/1
Stenseth NC, Mysterud A (2005) Weather packages: finding the right scale and composition of climate in ecology. J Animal Ecol 74:1195–1198
Stige LC, Ottersen G, Brander K, Chan KS, Stenseth NC (2006) Cod and climate: effect of the Northa Atlantic Oscillation on recruitment in the North Atlantic. Mar Ecol Prog Ser 325:227–241
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, Berlin
Watanabe Y, Kurita Y, Noto M, Oozeki Y, Kitagawa D (2003) Growth and survival of Pacific saury (Cololabis saira) in the Kuroshio-Oyashio transitional waters. J Oceanogr 59:403–414
Watanabe Y, Zenitani H, Kimura R (1996) Causes of population decline in the Japanese sardine (Sardinops melanostictus): overfishing or early mortality? In: Watanabe Y, Yamashita Y, Oozeki Y (eds) Survival strategies in early life stages of marine resources. Balkema, Rotterdam, pp 83–94
Wood S (2006) Generalized additive models: an introduction with R. Chapman and Hall/CRC, London, p 410
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
Maynou, F. (2011). Impact of NAO on Mediterranean Fisheries. In: Vicente-Serrano, S., Trigo, R. (eds) Hydrological, Socioeconomic and Ecological Impacts of the North Atlantic Oscillation in the Mediterranean Region. Advances in Global Change Research, vol 46. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-1372-7_7
Download citation
DOI: https://doi.org/10.1007/978-94-007-1372-7_7
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-1371-0
Online ISBN: 978-94-007-1372-7
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)