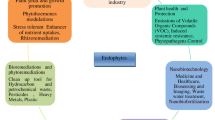
Abstract
Endophytes are the organisms that exist within the living tissues of most plant species without inducing any pathogenic symptoms in the host. Their association with the host plant may be symbiotic, mutualistic, commensalistic, trophobiotic and their interaction with them may be biochemical and genetic as well. It is their enormous diversity and specialized biotopes that make them a stimulating field of study. The fact that endophytes can mimic some of the phytochemicals originally characteristic of the host has changed the scope of endophytic biology. As a result of this fact, various classes of natural products that have been discovered from endophytes which include antitumour, antibacterial, antifungal, antiviral and various different industrial enzymes. Even some unusual molecules that act as immunomodulatory, antidiabetic, insecticidal, herbicidal, etc. have been isolated from them. Endophyte–plant relationship can be exploited to promote plant health as it also plays an important role in low-input sustainable agricultural applications for both food and nonfood crops. Recent studies on endophytes have shown the beneficial roles of host-endophyte associations as protection against mammals, resistance to pathogenic fungi and other insect herbivores, increased growth and development, nutrient uptake and stress tolerance in various plants including agriculturally important crops. The main objective of this review is to explore the potential of endophytes in varied fields that can prove beneficial to mankind. As a result, the role of endophytes has been discussed in some of the key fields of interest such as biotransformation, biodegradation, phytoremediation, seed production, seed predation, plant growth promotion, plant stress relievers, biocatalysis, biofuel production, biocontrol agents, agricultural importance, source of novel natural products etc. The present review is an attempt to unlock the enormous benefits that the microbial endophytes can offer.
Similar content being viewed by others
Keywords
1 Endophytes
Endophytes have over the period of time evolved from being defined just as the microbes living within plants indicating not only their location but also the type of association that they have with the host. They are the organisms that live asymptomatically within the internal tissues of the plant. Endophytes live for its life or part of it without causing any apparent disease to the host plant. The word endophytes (Greek endo = within + phytes = plants) has also been defined as the microorganisms inhabiting inside of healthy plant tissues. Petrini (1991) described them as all organisms that inhabit or colonize internal plant tissues at some part of their life without any immediate deleterious effect on their host. They are now considered as ubiquitous symbionts of plants from their common detection to many plant species.
Endophytes have now been considered as an important component of microbial biodiversity, as they are found in virtually every plant on earth ranging from terrestrial to aquatic. Common endophytes include a variety of bacteria, fungi and actinomycetes which can be isolated from wild (Brooks et al. 1994) or cultivated crops (Liu and Tang 1996) of either the monocotyledonous (Fisher et al. 1992) or dicotyledonous plant groups (El-Shanshoury et al. 1996). A single plant can harbour several to hundreds of endophyte species, some of which may also show host specificity. They exhibit a variety of relationships with their host ranging from symbiotic to pathogenic. The observed biodiversity of endophytes suggests that they can also be aggressive saprophytes or opportunistic pathogens. Endophytes usually inhabit above-ground plant tissues (leaves, stems, bark, petioles and reproductive structures) which distinguish them from better known mycorrhizal symbionts. The distinction is not so firm, because endophytes may also inhabit root tissues.
The type of symbiosis exhibited between plant and endophyte is often related to the reproductive mode of the endophyte. Most of the endophytes are horizontally transmitted. These have sexual mode of transmission via spores and can be spread by wind, insects and vectors; so, these can evolve a more pathogenic lifestyle as they can escape their host plant, whereas vertically transmitted endophytes are asexual and transmit via fungal hyphae penetrating the host’s seed (e.g. Neotyphodium).
Endophytes have a very intimate and also a coevolutionary relationship with their host. Therefore, they have the potential to influence the physiology of the plant. They confer enhanced fitness to the host plants by producing functional metabolites in lieu of the nutrition and protection from the host. They offer a plethora of benefits to the plant ranging from resistance against diseases to insect herbivory.
2 Endophytes as Source of Novel Bioactive Metabolites
Endophytes can be considered as potential, reliable and promising source of novel organic natural metabolites exhibiting a variety of biological activities viz. antimicrobial, antiparasitic, cytotoxic, neuroprotective, antioxidant, insulin mimetic, enzyme inhibitive, immunosuppressive and many more. A broad variety of bioactive secondary metabolites with unique structures, including alkaloids, benzopyranones, chinones, flavonoids, phenolic acids, quinones, steroids, terpenoids, tetralones, xanthones and many more, are the members of the diverse metabolite bank of endophytes. Many reports witness the various usages of endophytic fungal metabolites in different human diseases due to their highly functionalized activity. Various fungal endophytes from selected medicinal plants of western Himalayas have been reported to show antimicrobial and immunomodulatory activities (Qadri et al. 2013). Antibacterial activity of four fungal endophytes (Cladosporium sp., Aspergillus flavus, Aspergillus sp., Curvularia lunata) isolated from Kigelia africana has been reported by Idris et al. (2013). Fungal endophytes isolated from the plant Aralia elata have also been reported as potential antibacterial agents (Wu et al. 2012). Metabolites of endophytes isolated from Artemisia annua have been reported to show anti-acetylcholinesterase activity and can therefore be used in the treatment of Alzheimer’s disease (Aly et al. 2011). Some of the endophytes have the potential to produce the same bioactive metabolites for which the host plant is known, hypericin, camptothecin, podophyllotoxin, paclitaxel and diosgenin (Zhao et al. 2010). There is an increasing interest in studying fungal endophytes as alternative potential source of functional metabolites. So far, about 19 genera from different plant sources have been screened for the production of paclitaxel and its analogues. Cephalotheca faveolata, a fungal endophyte isolated from Eugenia jambolina has been reported to exhibit anticancerous activity (Giridharan et al. 2012). Therefore, keen studies on endophytes are required for the commercial and economical production of taxol and other host origin chemicals from endophytic microbes, as their production from host plant is comparatively costlier, time consuming and exerts biodiversity pressure.
Endophytic fungi isolated from six species of Indonesian medicinal plants Mahoni (Swietenia mahagoni Jacq.), Sambiloto (Andrographis paniculata Ness.), Kumis kucing (Orthosiphon spicatus BBS), Mengkudu (Morinda citrifolia L.), Sirih merah (Piper crocatum L.) and Sirih hitam (Piper ornatum sp.) have the potential to produce antidiabetic agents (Dompeipen 2011). The foliar endophytes of conifers have been reported to synthesize anti-insect toxins (Sumarah et al. 2009). So, strategic use of specific endophytes or their metabolites can be used in plant–insect/pest management programmes.
Some of the important bioactive metabolites isolated recently from endophytes and their bioactive functions are mentioned in detail in Table 4.1. For more information readers are suggested to read reviews by Tan and Zou (2001); Schulz et al. (2002); Strobel and Daisy (2003); Strobel et al. (2004); Joseph and Priya (2011); Kharwar et al. (2011); Premjanu and Jayanthy (2012).
3 Seed Production and Seed Predation
Presence or absence of endophytes influences the production and predation of seeds among different plants, especially the grasses. Saari et al. (2010) studied the grass populations of tall fescue and meadow fescue for the effect of presence or absence of endophytes on production and predation of seeds. Endophyte infected (E+) and endophyte free (E−) tall fescue plants did not show any difference in the mean seed mass or production of seeds, but seeds from E+ plants showed significantly lower proportions of seed predation compared to seeds from E− plants. In contrast, endophyte infected (E+) meadow fescue plants produced marginally more seeds than endophyte free (E−) plants. Seed predation of meadow fescue plants by cocksfoot moth did not differ between endophyte infected and endophyte free plants. These results suggest that endophyte infection benefits tall fescue grasses by enhancing host resistance to seed predators rather than through enhanced reproductive effort. On the other hand, it also suggests that endophyte infection benefits meadow fescue grasses by enhancing their capabilities of seed production rather than enhancement of host resistance to seed predators. The possible reasons for this variability in the effects of endophytes is firstly, the high genetic variability among wild grasses; secondly, different species of endophytes infecting meadow fescue and tall fescue grasses may have different effects on various invertebrate and vertebrate herbivores (Cheplick and Faeth 2009); thirdly, the production of mycotoxins by endophytes, especially alkaloids, is conditional on environmental factors such as weather conditions or soil nutrients (Saikkonen et al. 2006; Saikkonen et al. 2010) and the genotype of the plant and endophyte (Cheplick and Faeth 2009).
Endophytes also aid in the seed production in particular types of grasses such as bird deterrent grasses or grass swards used at airports (Pyke et al. 2010). These grasses can deter birds either by reducing insect populations for insectivorous birds or by deterring the feeding of herbivorous birds causing them to shift to some other areas for feeding. These deterrent properties of the grasses are provided by endophytic fungi specifically selected for these characteristic features. Endophytic fungi grow within the host plant into the developing seed and when these seeds are sown, they develop into the new grass seedlings that confer bird deterrence to the developing sward (sward grass).
4 Phytoremediation
Benefits of industrialization, modernization and urbanization are always associated with some harmful effects such as accumulation of a wide variety of anthropogenic chemicals which includes halogenated hydrocarbons, petroleum hydrocarbons, polycyclic aromatic hydrocarbons, pesticides, heavy metals, solvents, etc. in the environment. This has caused havoc by enhancing the environmental problems (Meagher 2000; Rajkumar et al. 2010; Ma et al. 2011a, b). Phytoremediation by endophytes has been found to be a promising technology for the remediation of contaminated soils (Li et al. 2012). For the phytoremediation of heavy metals, the endophytes resistant to heavy metals can decrease metal phytotoxicity, enhance plant growth and affect metal accumulation and translocation in plants. Endophytes have application in the phytoremediation of organic contaminants produce various enzymes that degrade these contaminants thereby reducing both the evapotranspiration and phytotoxicity of volatile compounds. Li et al. (2012) have also discussed about various endophytes (bacteria and fungi) that aid in phytoremediation of heavy metals and organic contaminants. Various metal resistant bacterial endophytes belong to the taxa Bacillus, Clostridium, Arthrobacter, Enterobacter, Leifsonia, Microbacterium, Paenibacillus, Pseudomonas, Xanthomonadaceae, Staphylococcus, etc. Likewise, various reports of metal-resistant fungal endophytes include species of Alternaria, Microsphaeropsis, Steganosporium, Mucor, Phoma, Peyronellaea and Aspergillus. Microbial endophytes that are able to degrade organic contaminants include Pseudomonas rhodesiae, Bacillus megaterium, Paenibacillus amylolyticus, Burkholderia macroides, Achromobacter xylosoxidans, Pseudomonas tolaasii and Pseudomonas putida. Two main groups of contaminants that are being degraded by endophyte-assisted phytoremediation include heavy metals (Ni, Pb, Cu, Cd, Cr, Zn, Co, etc.) and organic contaminants (trichloroethylene, naphthalene, catechol, phenol, alkanes, BTEX (benzene, toluene, ethylbenzene and xylene), etc.).
Engineered bacterial endophytes are being incorporated into the plants in order to improve the phytoremediation of contaminants particularly organic ones, so that they can efficiently degrade these organic contaminants (Taghavi et al. 2011). These engineered bacterial endophytes have also been tested to improve the phytoremediation of heavy metals but this study is still in its infancy (Lodewyckx et al. 2001).
Phytoremediation of organic compounds especially benzene, toluene, ethylbenzene, and xylene (BTEX) or trichloroethylene (TCE) brings about the evaporation of a significant amount of metabolites from these contaminants into the atmosphere thereby increasing their levels much above the optimal amounts. Therefore, to prevent the environment from such hazards, there is a need for certain alternative means by which these contaminants can be minimized. One of the best and safe ways is to inoculate plants with engineered bacterial endophytes so that they can optimally metabolize the contaminants of interest, thereby improving the overall process of phytoremediation.
5 Biotransformation
Biotransformation may simply be defined as any change or conversion of any substance by or in a biological system. It has been found that endophytes have excellent biotransformation abilities as they adapt so well with their host plants thereby having a strong tolerance towards the unique metabolites including toxins of their hosts (Wang and Dai 2011).
To control the excessive growth of endophytes, plants sometimes produce certain toxins against them. Endophytes have gradually formed a variety of tolerance mechanisms towards hosts’ metabolites over their long period of coevolution, and have acquired a powerful ability to transform complex compounds. These endophytes may degrade the hosts’ macromolecules into simpler ones or convert toxins to other substances thereby increasing their adaptability. In addition to plants, sometimes even endophytes become aggressive towards their host plants thereby producing mycotoxins or exoenzymes in order to colonize a particular host (Schulz et al. 2002). Several cases of endophytic virulence have also been reported (Peters et al. 1998; Costa et al. 2000). Likewise endophytes, plants also have defense mechanisms against the virulence effects of endophytes. Thus, from these studies, it is concluded that both the host plant and the endophyte produce unique metabolites and enzymes against each other and even in their defence reactions thereby making this host–endophyte relationship much more complex (Saikkonen et al. 2004).
The process of biotransformation mediated by endophytes has various applications, some of which are discussed as under:
5.1 Detoxification of Toxic Metabolites Produced by Host Plants
One of the important abilities of the endophytes is the detoxification of highly bioactive defence metabolites of host plant and converting them to simpler compounds to be easily metabolized. For example, plants usually produce unique class of compounds like benzoxazinones for defence against pests such as fungi, bacteria and insects. Once the plant defence response is triggered on attack of pests, hydroxamic acids are chemically transformed into toxic benzoxazolinon 2-benzoxazolinone (BOA) or the less toxic 2-hydroxy-1,4-benzoxazin-3-(2H)-one (HBOA) and some other toxic compounds. These toxins are detoxified by the biotransformation abilities of endophytes as reported by Zikmundova et al. (2002). They isolated four fungal endophytes, among which Fusarium sambucinum detoxified (BOA) to N-(2- hydroxyphenyl) malonamic acid and the rest detoxified (HBOA) to 2-Hydroxy-N-(2-hydroxyphenyl) acetamide, N-(2-hydroxyphenyl) acetamide and some other structural analogues.
5.2 Stereoselective Biotransformation Mediated by Endophytes
Endophytes are being exploited to obtain more active metabolites because fungal endophytes as a result of their effective biotransformation enzymes have been employed to change the three-dimensional conformation of compounds, e.g. thioridazine (THD), a phenothiazine neuroleptic drug, is commercially available as a racemic mixture of the enantiomers. Borges et al. (2008) isolated four fungal endophytes that showed stereoselective biotransformation potential, thereby transforming THD into pharmacologically active metabolites. Other examples of endophyte mediated stereoselective biotransformation includes transformation of flavans, i.e. stereoselective oxidation of flavans at C-4 by the endophytic fungus Diaporthe sp. isolated from a tea plant Camelia sinensis (Agusta et al. 2005) and biotransformation of tetrahydrofuran lignans by the endophytic fungus Phomopsis sp. inhabiting Viguiera arenaria (Verza et al. 2009).
5.3 In Vivo Simulation of Drug Metabolism in Mammals by Endophytes
It has been found that sometimes, the endophyte-mediated enzymatic reactions are similar to metabolic responses in mammals. Thus, these metabolic pathways and the active metabolites involved therein could be very much useful in studies on metabolism in mammals, e.g. two naturally occurring triterpenes: betulinic acid and betulonic acid found in many plants, exhibit important biological properties like antiviral and antineuroblastomic activity. Bastos et al. (2007) isolated four fungal endophytes Arthrobotrys sp., Chaetophoma sp., Dematium sp. and Colletotrichum sp. that showed certain oxidation reactions thereby converting betulinic acid to many oxygenated derivatives and some of these oxidizing reactions also occur in mammals.
6 A Potential Tool for Biodegradation
A large number of extracellular enzymes are being produced by endophytes including proteinases, cellulases, lipoidases, pectinases, phenoloxidases and lignin catabolic enzymes (Tan and Zou 2001; Oses et al. 2006; Bischoff et al. 2009). Such broad spectrum of enzyme production helps the endophytes in degradation and decomposition of plant debris. Four fungal endophytes from Chilean tree showed a nonselective white-rot wood decay pattern (Oses et al. 2006). Jordaan et al. (2006) isolated three different fungal endophytes (Alternaria sp., Phoma sp. and Phomopsis sp.) from surface-sterilized pods of Colophospermum mopane showing lignocellulolytic enzyme activity that helps them to accelerate the decay of pods. By doing this, they allowed the germination of seeds more effectively under favourable conditions. Endophytic fungi are now being exploited for the degradation of environmental pollutants like polycyclic aromatic hydrocarbons (PAHs). Dai et al. (2010) isolated a novel endophytic fungus Ceratobasidum stevensii from the euphorbiaceae plant that metabolized 89.51 % of environmental pollutant phenanthrene. Likewise, various other novel fungal endophytes harboured by different plants are being studied for the degradation of various energetic compounds like 2,4,6-trinitrotoluene (TNT), octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine (HMX), hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX), etc. (Aken et al. 2004).
7 Endophytes as a Source of Biocatalysts
It has been found that the endophytes produce a large number of enzymes that aid in the catabolism of a wide variety of complex organic molecules such as proteins, cellulose, chitin, etc. This is due to the absorptive mode of nutrition of these fungi and a wide variety of substrates which they exploit for their growth and thus produce a diverse array of extracellular enzymes like cellulases, chitinases, amylases, lipases and proteases, etc. (Suryanarayanan et al. 2012). Ostergaard and Olsen (2010), concluded that around 60 % of the industrial enzymes used currently are of fungal origin with a wide variety of applications, e.g. enzymes produced by endophytes are being used for meat processing, fermentation of coffee beans, baking, manufacturing corn syrup, hydrolysing milk proteins, removing stains, separating racemic mixtures of amino acids, bioremediation and biosensing. Hawksworth (1991) found that there are only five genera of fungi (Penicillium, Rhizopus, Aspergillus, Humicola and Trichoderma) out of 1.5 million estimated members which account for almost three-quarters of the 60 % industrial enzymes produced by endophytic fungi (Ostergaard and Olsen 2010). This leads to the immediacy for screening of novel and more efficient biocatalysts from different ecological groups of fungi (Peterson et al. 2011). There are a wide variety of industrial enzymes derived from endophytes acting as biocatalysts with diverse applications. Some of the common enzymes isolated from endophytes are as follows:
7.1 Alkaline Protease
Suryanarayanan et al. (2012) isolated several fungal endophytes from the leaves of dicotyledonous trees which showed positive results for alkaline protease activity using agar plate assay. It has been found that many fungal endophytes belonging to the genera Nodulisporium, Corynespora, Colletotrichum, Xylaria, Curvularia and Robillarda produce alkaline proteases using dot-blot assay (Thirunavukkarasu and Suryanarayanan unpublished). This industrial enzyme can be exploited for production of protein hydrolysates, mediation of drug release, brewing, baking, detergent additive, denture cleaners, tanning processes, cosmetics, animal feeds, waste water treatment, etc. (Gupta et al. 2002).
7.2 Chitinase and Chitosanase
Suryanarayanan et al. (2012) isolated foliar fungal endophytes inhabiting the trees of different forests in the Western Ghats mountain ranges (in southern India) that produced various extracellular enzymes including chitinases and chitosanases. These enzymes can be used in drug preparations for treating cancer, diabetes, asthma, inflammation, immunity modulation, anti-inflammatory, bacterial/fungal infections, wounds, sialic acid production, etc. (Hartl et al. 2012).
7.3 Laccase
Kumaresan et al. (2002) isolated two fungal endophytes Pestalotiopsis sp. and Glomerella sp. inhabiting Rhizophora apiculata and screened them for the production of extracellular enzymes such as laccase and cellulase. Promputtha et al. (2010) also reported the production of laccase from Corynespora cassiicola, endophytic in Magnolia liliifera. This enzyme can be exploited for processing of wine, fruit juice and beer, bioremediation of phenolic compounds, delignification, bio-bleaching, etc. (Kunamneni et al. 2008).
7.4 Chitin Deacetylase
Suryanarayanan et al. (2012) observed the production of chitin deacetylase from the fungal endophytes Pestalotiopsis sp. and Sordaria sp. inhabiting the tropical forest trees. This enzyme is used for biological control of insect pests, human and plant pathogens and can even be exploited for preparation of chitosan from chitin (Zhao et al. 2010).
7.5 Acidic Protease
Many foliar fungal endophytes, i.e. Nodulisporium, Xylaria, Colletotrichum, Corynespora and Curvularia show positive results for the production of acidic proteases (Suryanarayanan et al. 2012). This enzyme aids in preparation of fermented foods and seasoning material as well as in digestion (Rao et al. 1998).
7.6 β-glucosidase
Suryanarayanan et al. (2012) have observed the production of β-glucosidases from the endophytic Phomopsis sp. Chauve et al. (2010) reported that this enzyme is mainly used for biofuel production from lignocellulosic biomass.
7.7 Tannase
Production of tannase enzyme from the endophytic Phomopsis sp. has been reported by Suryanarayanan et al. (2012). This enzyme is used as a clarifying agent in instant tea, fruit juices and wine (Ramirez-Coronel et al. 2003).
8 Agricultural Importance of Endophytes
In order to feed the growing human population, the use of man-made fertilizers (particularly N and P fertilizers) have increased manifold to increase the crop production (Vance 2001). People are mainly interested in large-scale crop production, thereby using wide varieties of pesticides, fungicides and bactericides without caring about their harmful effects on ground and surface water (Cook 1992).
One of the best alternative methods involves the use of endophytes as biofertilizers in agriculture. In the recent times, use of endophytes in agriculture have received a substantial attention due to their broad host range, tolerance to a vast range of environmental conditions and promotion of vegetative growth including seed production of various economically important plants (Singh et al. 2000; Shende et al. 2006). Endophytes have become a valuable tool for agriculture due to their capability to colonize the internal tissues of the host and to contribute towards plant growth.
Over the last 40 years, nutrient use efficiency (NUE) in crop production has decreased considerably (Hirel et al. 2007). Due to this and rapid depletion of mines mineral deposits (particularly Potassium and phosphorus) along with their increasing costs have raised threat to current crop production and future food security (Tilman et al. 2002; Cordell et al. 2009). Nutrient use efficiency in crop plants may significantly be increased by root-colonizing endophytes. These endophytes help the crop plants in two ways:
-
a.
Direct mode of action through nitrogen fixation, phosphorus and micronutrient uptake, etc.
-
b.
Indirect mode of action through antagonism against soil- and seed-borne pathogens, induction of plant resistance, etc.
Nowadays, a strongly growing field of land use is desert agriculture, but the problems caused by soil-borne pathogens limit the yield. However, endophytic biological control agents (BCAs) are the promising candidates for plant protection that can suppress these soil-borne pathogens. But for desert agriculture, only specific and drought resistant strains of BCAs will prove beneficial (Koberl et al. 2011). Khan et al. (2012) reported an endophyte, Paraconiothyrium sp. as a potential producer of phytotoxin. So, it can be exploited for weed control strategies.
Among the most important microbes beneficial for agriculture are the different species of genus Trichoderma, being used worldwide as active ingredients of biopesticides and biofertilizers (Harman et al. 2004). Specific species of this genus can act endophytically in the vascular system by colonizing the root tissues thereby, establishing a physiological interaction with the host plant based on molecular crosstalk. This results in the promotion of plant growth and development along with resistance to diseases, degradation of toxic compounds, promotion of water and nutrient uptake and finally brings about the production of bioactive Trichoderma metabolites.
9 Endophytes as Nanoparticle Biosynthesizers
Endophytes can be used as potential biosynthesizers for production of various economically and industrially important compounds. Various endophytes can be exploited for the extracellular or intracellular synthesis of different indigenous or nonindigenous compounds. Pestalotia sp. isolated from the healthy leaves of Syzygium has been used for the extracellular synthesis of silver nanoparticles which exhibited good antimicrobial potential (Raheman et al. 2011). Similarly, Aspergillus clavatus, isolated from the sterilized stem tissue of Azadirachta indica, was also reported to be capable of synthesizing extracellular silver nanoparticles (Verma et al. 2010). The same endophyte was found to produce diverse range of gold nanoparticles as well, especially nanotriangles in the size range from 20 to 35 nm. These nanotriangles possess special optical and physical properties which can be exploited for vital applications such as biomedicine, optics, electronics, catalysis and many more (Verma et al. 2010). There is a big thrust in the area of synthesizing different metal nanoparticles using biological systems these days because of their potential application in nanomedicines (Panneerselvam et al. 2011). There are wide applications of nanoparticles in diverse fields as catalysts, sensors, medicines, etc. that largely depend on the different physical and optical properties of the particles. The synthesis of nanoparticles using biological systems provides new routes to develop nanoparticles with desired properties for making their exploitation possible in diverse fields (Pugazhenthiran et al. 2009). The nonpathogenic and ecofriendly behaviours of endophytes make them as good candidates for biosynthesizers.
10 Endophytes as Host Plant Growth Promoters
Endophytes might prove to be important biological agents in near future and revolutionize the agriculture industry without harming the nature. By artificially inoculating specific endophytes in non-host plants, the property of microbes to improve host plant growth potential can be transferred to new and economically important crops. It has been observed that seed bacterization of bhendi plant with some selected endophytic isolates promote plant growth (Vetrivelkalai et al. 2010). Another endophyte Piriformospora indica has great potential as plant growth-promoting agent and is sometimes termed as plant probiotic. Phosphorous (P) is one of the most essential mineral nutrients for proper plant growth and development. P. indica has important role in P transfer to the plant particularly under P-deprived conditions (Kumar et al. 2011). Some endophytes are reported to have nitrogen fixation ability that can also promote the host plant growth (Hurek et al. 2003). Enterobacter sp. 638, isolated from poplar plant, has been reported as a plant growth-promoting gamma-proteobacterium (Taghavi et al. 2010). Poplar plant is one of the potentially important biofuel feed stock plant. Combined genome, transcriptome and metabolome analysis revealed synergistic interaction between poplar and its growth-promoting endophyte. Nitrogen fixation, P solubilization, indole acetic acid (IAA) production and siderophore secretion are some of the commonly followed mechanisms through which endophytes are able to promote the host plant growth. Improved plant growth, higher nutrient content, resistance to insect pests and herbivores, resistance or tolerance to diseases, increased competitiveness, enhanced tolerance to stressful factors such as heavy metals, low pH, high salinity, etc., are the other benefits from the endophytic interaction (Kumar et al. 2011). Endophytes can be used as biofertilizers for plant growth promotion (Pandya et al. 2010). The plant growth-promoting potential of endophytes can also be exploited for the improved and sustainable production of important crops on marginal and nonagricultural soils (Table 4.2).
11 Endophytes as Host Plant Stress Relievers
Different kinds of abiotic factors such as drought, flood, high soil salinity, heat, cold, oxidative stress, heavy metal toxicity and nutrient deficiency, etc. lead to stress in the plant resulting in lower or no yield. Plant growth-promoting potential of endophytes can be exploited to overcome the limitations of crop productivity caused by different abiotic stress factors and it may prove to be a promising alternative strategy (Table 4.3). For the sustainable existence of plants, endophytes have done and are still doing a good job as symbionts (Singh et al. 2011). P. indica and Sebacina vermifera are common root endophytes found in wide variety of plant species. Both organisms have the potential to promote their host plant growth, and to induce resistance against soil-borne fungal pathogens (Ghahfarokhi et al. 2010). Agricultural productivity under saline conditions can be improved by exploiting the potential of endophytic fungi as they have been reported to produce important stress-relieving phytohormones (Khan et al. 2012). The ability of fungal endophytes to confer stress tolerance to plants may provide a novel strategy for mitigating the impacts of global climate change on agricultural and native plant communities (Rodriguez et al. 2008).
12 Endophytes as a Source of Biofuel
Constantly increasing energy consumption, energy demand and environmental concerns are constantly heralding the need of an alternative biofuel resource. Many endophytes possess enzymes of interest that have the ability to degrade available carbon sources into compounds that can be used as biofuels (Suryanarayanan et al. 2012). Studies say that endophytes can be exploited for the synthesis of fuel-based compounds utilizing cellulose-based medium from host plant. Solid-phase micro-extraction (SPME)-gas chromatography/mass spectrometry (GC/MS) analysis of an endophytic fungus, Gliocladium roseum revealed the production of a series of volatile hydrocarbons and their derivatives under controlled conditions (Strobel et al. 2008). The hydrocarbons characterized were named as myco-diesel as some of them had properties similar to diesel fuel. Some of the endophytes isolated from different species of tropical plants may prove to be potential biofuel sources as they contain high concentrations of methyl esters which are effective biofuel precursors (Santos et al. 2011).
13 Endophytes as Biocontrol Agents
Many plant endophytes are reported as potential biocontrol agents against plant pathogens. The biocontrol activity imposed by endophytes may be due to competition for space and nutrients, secretion of chitinolytic enzymes, myco-parasitism and production of inhibitory compounds. Some root endophytic fungi like Piroformospora indica, Sebacina vermifera and Trichoderma species have been reported as potential biocontrol agents against plant pathogens such as Gaeumannomyces graminis, causative agent of Take-all disease of wheat; Fusarium oxysporum, causative agent of fusarium wilt of lentil and many more (Ghahfarokhi et al. 2010; Dolatabadi et al. 2012). Shalini and Kotasthane (2007) reported strains of Trichoderma including Trichoderma harzianum, Trichoderma viride and Trichoderma aureoviride as growth inhibitors of Rhizoctonia solani, a potent plant pathogen. Some endophytes, if not completely eliminate, reduce the severity of the host plant disease to an efficient extent. P. indica-colonized host plants were reported to be affected less severely by Pseudocercosporella herpotrichoides disease (Pandya et al. 2010). It has been reported that P. indica has the ability to improve the production of tomato by acting antagonistically against Verticillium dahliae (Fakhro et al. 2010). The host plant growth promotion by P. indica was not only observed in soil but also in hydroponic cultures. Muscodor albus, an endophytic fungal isolate from the plant Cinnamomum zeylanicum was reported to produce bioactive volatile organic compounds (VOCs). The mixture of VOCs produced by the isolate was found to be effective against various human and plant pathogenic fungi and bacteria (Strobel et al. 2006). Some of the fungal endophytes isolated from different vegetable crops were reported to exhibit anti-oomycete property (Kim et al. 2007). In dual culture tests, the fungal isolates were shown to inhibit the growth of various oomycete plant pathogens, viz. Pythium ultimum, Phytophthora infestans and Phytophthora capsici. So, endophytes can be considered as potent anti-oomycete agents. More intensive research is required to optimize the application of endophytes as biocontrol agents so that the quality of crops in terms of taste and health should not be negatively affected.
13.1 Concluding Statement and Future Perspectives
In today’s world, man has become almost inadequate to cope up with the medical problems due to the emergence of new diseases, increased incidences of fungal infections and drug resistance by microorganisms. All this speaks about the need for new and useful bioactive compounds to provide relief and assistance to human sufferings. Endophytes are considered as a mine for bioprospecting and thereby, help in various aspects to overcome many problems. It is proposed that many interesting cases are yet to be discovered even after the exploitation of number of new endophytes and the metabolites thereof reported since last century. Endophytes have not only benefited animals, but have been exploited to protect various plants from a wide range of pathogens and insect pests, etc. Some among them function by antagonism, inducing host defenses and also help in improving plants health and nutrition.
Among the various endophytic microbes, fungi have particularly proved to be a promising and largely untapped reservoir of bioactive metabolites. The natural products have been optimized by evolutionary, environmental and ecological factors yielding useful and effective bioactive secondary metabolites.
Majority of the drugs that are now being used against infective organisms, agronomic pests, parasites of livestock and humans and diseases such as tuberculosis, cancer, malaria, diabetes, etc., are mainly composed of a diverse range of bioactive plant secondary metabolites.
To achieve a range of applications associated with plant–endophyte relationship, there is a need for understanding the mechanisms behind the interaction of endophytes with their host plant so that the biotechnological potential of such a relationship can be fully elucidated. A deeper understanding at the molecular and genetic levels of this relationship may prove beneficial for inducing and optimizing secondary metabolite production in vitro to yield potential bioactive metabolites. As a result of genetic engineering and metabolic regulation, the content and the yield of active ingredients in certain known strains can be accordingly increased for large-scale production. The present review has unlocked the myriad benefits of endophytes which can be utilized in different ways for the improvement of plant growth, development and productivity. Endophytes, hence is an important and interesting complex occupying a unique niche worth for exploration.
References
Agusta A, Maehara S, Ohashi K, Simanjuntak P, Shibuya H (2005) Stereoselective oxidation at C-4 of flavans by the endophytic fungus Diaporthe sp. isolated from a tea plant. Chem Pharm Bull 53:1565–1569
Aken VB, Yoon JM, Schnoor JL (2004) Biodegradation of nitro-substituted explosives 2, 4, 6-trinitrotoluene, hexahydro-1, 3, 5-trinitro-1, 3, 5- triazine, an octahydro-1, 3, 5, 7-tetranitro-1, 3, 5-tetrazocine by a phytosymbiotic Methylobacterium sp. associated with poplar tissues (Populus deltoides x nigra DN34). Appl Environ Microbiol 70:508–517
Aly AH, Debbab A, Proksch P (2011) Fungal endophytes: unique plant inhabitants with great promises. Appl Microbiol Biotechnol 90:1829–1845
Bae H, Sicher RC, Kim MS, Kim SH, Strem MD, Rachel L, Melnick RL, Bailey BA (2009) The beneficial endophyte Trichoderma hamatum isolate DIS 219b promotes growth and delays the onset of the drought response in Theobroma cacao. J Exp Bot 60(11):3279–3295
Bastos DZL, Idac P, de Jesus DA, de Oliveira BH (2007) Biotransformation of betulinic and betulonic acids by fungi. Phytochemisty 68:834–839
Bischoff KM, Wicklow DT, Jordan DB, de Rezende ST, Liu SQ, Hughes SR, Rich JO (2009) Extracellular hemicellulolytic enzymes from the maize endophyte Acremonium zeae. Current Microbiology 58:499–503
Borges KB, Borges WDS, Pupo MT, Bonato PS (2008) Stereoselective analysis of thioridazine-2-sulfoxide and thioridazine-5- sulfoxide: an investigation of rac-thioridazine biotransformation by some endophytic fungi. J Pharmaceut Biomed 46:945–952
Brooks DS, Gonzalez CF, Appel DN, Filer TH (1994) Evaluation of endophytic bacteria as potential biological control agents for oak wilt. Biol Contr 4:373–381
Bunkers GF, Kenfield D, Strobel GA (1991) Production of petasol by Drechslera gigantia in liquid culture. Mycological Research 95:347–351
Bunyapaiboonsri T, Yoiprommarat S, Srikitikulchai P, Srichomthong K, Lumyong S (2010) Oblongolides from the endophytic fungus Phomopsis sp. BCC 9789. J Nat Prod 73(1):55–59
Campos FF, Rosa LH, Cota BB, Caligiorne RB, Rabello ALT, Alves TMA, Rosa CA, Zani CL (2008) Leishmanicidal metabolites from Cochliobolus sp., an endophytic fungus isolated from Piptadenia adiantoides (Fabaceae). PLoS Negl Trop Dis 2(12):e348
Chauve M, Mathias H, Huc D, Casanave D, Monot F, Ferreira (2010) Comparative kinetic analysis of two fungal β-glucosidases. Biotechnol Biofuels. doi:10.1186/1754–6834-3–3
Cheplick GP, Faeth SH (2009) Ecology and evolution of the grass-endophyte symbiosis. Oxford University Press, Oxford. doi:10.1093/acprof:oso/9780195308082.001.0001
Cook GW (1992) Fertilizing for maximum yield, 3rd edn. MacMillan, New York
Cordell D et al (2009) The story of phosphorus: global food security and food for thought. Global Environ Change 19:292–305
Costa LSR, Azevedo JL, Pereira JO, Carneiro ML, Labate CA (2000) Symptomless infection of banana and maize by endophytic fungi impairs photosynthetic efficiency. New Phytol 147:609–615
Dai CC, Tian LS, Zhao YT, Chen Y, Xie H (2010) Degradation of phenanthrene by the endophytic fungus Ceratobasidum stevensii found in Bischofia polycarpa. Biodegradation 21:244–255
Debbab A, Aly AH, Edrada-Ebel RA, Muller WEG, Mosaddak M, Hakiki A, Ebel R, Proksch P (2009) Bioactive secondary metabolites from the endophytic fungus Chaetomium sp. isolated from Salvia officinalis growing in Morocco. Biotechnol Agron Soc Environ 13(2):229–234
Dolatabadi HK, Goltapeh EM, Mohammadi N, Rabiey M, Rohani N, Varma A (2012) Biocontrol Potential of Root Endophytic Fungi and Trichoderma Species Against Fusarium Wilt of Lentil Under In vitro and Greenhouse Conditions. J Agr Sci Tech 14:407–420
Dompeipen EJ, Srikandance Y, Suharso WP, cahyana H, Simanjuntak P (2011) Potential endophytic microbes selection for antidiabetic bioactive compounds production. Asian J Biochem 6(6):465–471
El-Shanshoury AR, El-Sououd SMA, Awadalla OA, El-Bandy NB (1996) Effects of Streptomyces corchorusii, Streptomyces mutabilis, pendimethalin and metribuzin on the control of bacterial and Fusarium wilt of tomato. Can J Bot Rev 74:1016–1022
Fakhro A, Linares DRA, Bargen SV, Bandte M, Buttner C, Grosch R, Schwarz D, Franken P (2010) Impact of Piriformospora indica on tomato growth and on interaction with fungal and viral pathogens. Mycorrhiza 20:191–200
Feng Y, Shen D, Song W (2006) Rice endophyte Pantoea agglomerans YS19 promotes host plant growth and affects allocations of host photosynthates. J Appl Microbiol 100(5):938–945
Fisher PJ, Petrini O, Lazpin SHM (1992) The distribution of some fungal and bacterial endophytes in maize (Zea mays L.). New Phytol 122:299–305
Gangadevi V, Muthumary J (2007) Endophytic fungal diversity from young, mature and senescent leaves of Ocimum basilicum L. with special reference to Taxol production. Ind J Sci Tech 1(1):1–12
Gao LW, Li WY, Zhao YL, Wang JW (2009) The cultivation, bioactive components and pharmacological effects of Armillaria mellea. Afr J Biotech 8(25):7383–7390
Ghahfarokhi RM, Goltapeh ME (2010) Potential of the root endophytic fungus Piriformospora indica; Sebacina vermifera and Trichoderma species in biocontrol of take-all disease of wheat Gaeumannomyces graminis var. tritici in vitro. J Agr Tech 6(1):11–18
Giridharan P, Verekar SA, Khanna A, Mishra PD, Deshmukh SK (2012) Anticancer activity of Sclerotiorin isolated from an endophytic fungus Cephalotheca faveoleta Yaguchi, Nishim & Udagawa. Ind J Exp Biol 50:464–468
Gupta R, Beg QK, Lorenz P (2002) Bacterial alkaline proteases: molecular approaches and industrial applications. Appl Microbiol Biotechnol 59:15–32
Haque MA, Hossain MS, Rahman MZ, Rahman MR, Hossain MS, Mosihuzzaman M, Nahar N, Khan SI (2005) Isolation of bioactive secondary metabolites from the endophytic fungus of Ocimum basilicum. J Pharm Sci 4(2):127–130
Harman GE, Howell CR et al (2004) Trichoderm a species-Opportunistic, avirulent plant symbionts. Nat Rev Microbiol 2(1):43–56
Hartl L, Zach S, Seidl-Seiboth V (2012) Fungal chitinases: diversity, mechanistic properties and biotechnological potential. Appl Microbiol Biotechnol 93:533–543
Hawksworth DL (1991) The fungal dimension of biodiversity: magnitude, significance, and conservation. Mycological Research 95:641–655
Hemtasin C, Kanokmedhakul S, Kanokmedhakul K, Hahnvajanawong C, Soytong K, Prabpai S, Kongsaeree P (2011) Cytotoxic Pentacyclic and tetracyclic aromatic sesquiterpenes from Phomopsis archeri. J Nat Prod 74(4):609–613
Hirel B et al (2007) The challenge of improving nitrogen use efficiency in crop plants: towards a more central role for genetic variability and quantitative genetics within integrated approaches. J Exp. Bot 58:2369–2401
Huang XZ, Zhu Y, Guan XL, Tian K, Guo JM, Wang HB, Fu GM (2012) A novel Antioxidant Isobenzofuranone Derivative from fungus Cephalosporium sp. AL031. Molecules 17:4219–4224
Hurek T, Hurek BR (2003) Azoarcus sp. strain BH72 as a model for nitrogen-fixing grass endophytes. J Biotech 106:169–178
Hussain H, Krohn K, Draeger S, Meier K, Schulz B (2009) Bioactive chemical constituents of a sterile endophytic Fungus from Meliotus dentatus. Rec Nat Prod 3(2):114–117
Idris A, Al-tahir I, Idris E (2013) Antibacterial activity of endophytic fungi extracts from the medicinal plant Kigelia africana. Egypt Acad J Biolog Sci 5(1):1–9
Isaka M, Chinthanom P, Boonruangprapa T, Rungjindamai N, Pinruan U (2010) Eremophilanetype sesquiterpenes from the fungus Xylaria sp. BCC 21097. J Nat Prod 73:683–687
Jordaan A, Taylor JE, Rossenkhan R (2006) Occurrence and possible role of endophytic fungi associated with seed pods of Colophospermum mopane (Fabaceae) in Botswana. South Afr J Bot 72:245–255
Joseph B, Priya RM (2011) Bioactive compounds from endophytes and their potential in pharmaceutical effect: a review. Am J Biochem Mol Biol 1:291–309
Khan SA, Hamayun M, Yoon H, Kim HY, Suh SJ, Hwang SK et al (2008) Plant growth promotion and Penicillium citrinum. BMC Microbiology. doi:10.1186/1471–2180-8–231
Khan AL, Hamayun M, Kang SM, Kim YH, Jung HY, Lee JH, Lee IJ (2012) Endophytic fungal association via gibberellins and indole acetic acid can improve plant growth under abiotic stress: an example of Paecilomyces formosus LHL10. BMC Microbiology 12(3):1–14
Kharwar RN, Verma VC, Kumar A, Gond SK, Harper JK, Hess WM, Lobkovosky E, Ma C, Ren Y, Strobel GA (2009) Javanicin, an antibacterial Naphthaquinone from an endophytic fungus of Neem, Chloridium sp. Current Microbiology 58:233–238
Kharwar RN, Mishra A, Gond SK, Stierle A, Stierle D (2011) Anticancer compounds derived from fungal endophytes: their importance and future challenges. Nat Prod Rep 28:1208–1228
Kim HY, Choi GJ, Lee HB, Lee SW (2007) Some fungal endophytes from vegetable crops and their anti-oomycete activities against tomato late blight. Lett Appl Microbiol 44:332–337
Koberl M, Muller H, Ramadan EM, Berg G (2011) Desert farming benefits from microbial potential in arid soils and promotes diversity and plant health. PLOS ONE 6(9)
Kumar M, Yadav V, Kumar H, Sharma R, Singh A, Tuteja N, Johri AK (2011) Piriformospora indica enhances plant growth by transferring phosphate. Plant Signal Behav 6(5):723–725
Kumaresan V, Suryanarayanan TS (2002) Endophyte assemblages in young, mature and senescent leaves of Rhizophora apiculata: evidence for the role of endophytes in mangrove litter degradation. Fungal Diversity 9:81–91
Kunamneni A, Camarero S, Garcia-Burgos C, Plou FJ, Ballesteros A, Alcalde M (2008) Engineering and applications of fungal laccases for organic synthesis. Microb Cell Factories. doi:10.1186/1475–2859-7–32
Li HY, Wei DQ, Shen M, Zhou ZP (2012) Endophytes and their role in phytoremediation. Fungal Diversity 54:11–18
Li XJ, Zhang Q, Zhang AL, Gao JM (2012) Metabolites from Aspergillus fumigatus, an endophytic fungus associated with Melia azedarach, and their antifungal, antifeedant, and toxic activities. J Agric Food Chem 60:3424–3431
Liu SF, Tang WH (1996) The study on endophytic streptomyces of cotton. In: Tang WH, Cook RJ, Rovira A (eds) Advances in biological control of plant diseases. China Agricultural University, China, pp 212–213
Lodewyckx C, Taghavi S, Mergeay M et al (2001) The effect of recombinant heavy metal resistant endophytic bacteria in heavy metal uptake by their host plant. Int J Phytoremediation 3:173–187
Ma Y, Prasad MNV, Rajkumar M, Freitas H (2011b) Plant growth promoting rhizobacteria and endophytes accelerate phytoremediation of metalliferous soils. Biotechnol Advances 29:248–258
Ma Y, Rajkumar M, Luo Y, Freitas H (2011a) Inoculation of endophytic bacteria on host and non-host plants-effects on plant growth and Ni uptake. J Hazard Mater 195:230–237
Meagher RB (2000) Phytoremediation of toxic elemental and organic pollutants. Curr Opin Plant Biol 3:153–162
Morse LJ, Day TA, Faeth SH (2002) Effect of Neotyphodium endophyte infection on growth and leaf gas exchange of Arizona fescue under contrasting water availability regimes. Environ Exp Bot 48:257–268
Nithya K, Muthumary J (2010) Secondary metabolite from Phomopsis sp. isolated from Plumeria acutifolia Poiret. Recent Res Sci Tech 2(4):99–103
Oses R, Valenzuela S, Freer J, Baeza J, Rodriguez J (2006) Evaluation of fungal endophytes for lignocellulolytic enzyme production and wood biodegradation. Int Biodeterior Biodegrad 57:129–135
Ostergaard LH, Olsen HS (2010) Industrial applications of fungal enzymes. In: Hofrichter XM (ed) The mycota. Springer, Berlin, pp 269–290
Pandya U, Saraf M (2010) Application of fungi as a biocontrol agent and their biofertilizer potential in agriculture. J Adv Dev Res 1(1):90–99
Panneerselvam C, Ponarulselvam S, Murugan K (2011) Potential anti-plasmodial activity of synthesized silver nanoparticle using Andrographis paniculata Nees (Acanthaceae). Arch Appl Sci Res 3(6):208–217
Peters S, Dammeyer B, Schulz B (1998) Endophyte-host interactions. I. Plant defense reactions to endophytic and pathogenic fungi. Symbiosis 25:193–211
Peterson R, Grinyer J, Nevalainen H (2011) Extracellular hydrolase profiles of fungi isolated from koala faeces invite biotechnological interest. Mycol Prog 10:207–218
Petrini O (1991) Fungal endophytes of tree leaves. In: Andrews JH, Hirano SS (eds) Microbial ecology of leaves. Springer Verlag, New York, pp 179–197
Pimentel MR, Molina G, Isio APD, Marostica Junior MR, Pastore GM (2010) The use of endophytes to obtain bioactive compounds and their application in biotransformation Process. Biotechnol Res Int. doi:10.4061/2011/576286
Premjanu N, Jayanthy C (2012) Endophytic fungi a repository of bioactive compounds—a review. Int J Inst Pharm Life Sci 2(1):135–162
Promputtha I, Hyde KD, McKenzie EHC, Peberdy JF, Lumyong S (2010) Can leaf degrading enzymes provide evidence that endophytic fungi becoming saprobes? Fungal Diversity 41:89–99
Pugazhenthiran N, Anandan S, Kathiravan G, Prakash NKU, Crawford S, Kumar MA (2009) Microbial synthesis of silver nanoparticles by Bacillus sp. J Nanopart Res 11:1811–1815
Puri SC, Nazir A, Chawla R, Arora R, Hasan SR, Amna T, Ahmed B, Verma V, Singh S, et al (2006) The endophytic fungus Trametes hirsuta as a novel alternative source of podophyllotoxin and related aryl tetralin lignans. J Biotechnol 122:494–510
Pyke N, Rolston P, Chynoweth R, Kelly M, Pennell (2010) Seed production of bird deterrent grass for use at airports. 29th Meeting of the International Bird Strike Committee, Cairns (Australia) 2010
Qadri M, Johri S, Shah BA, Khajuria A, Sidiq T, Lattoo SK et al (2013) Identification and bioactive potential of endophytic fungi isolated from selected plants of the Western Himalayas. SpringerPlus 2:8
Raheman F, Deshmukh S, Ingle A, Gade A, Rai M (2011) Silver nanoparticles: novel antimicrobial agent synthesized from a endophytic fungus Pestalotia sp. isolated from leaves of Syzygium cumini (L.). Nano Biomed Eng 1(3):174–178
Rajkumar M, Ae N, Prasad MNV, Freitas H (2010) Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends Biotech 28:142–149
Ramirez-Coronel MA, Viniegra-González G, Darvil A, Augur C (2003) A novel tannase from Aspergillus niger with β-glucosidase activity. Microbiology 149:2941–2946
Rao MB, Tanksale AM, Ghatge MS, Deshpande V (1998) Molecular and biotechnology aspects of microbial proteases. Microbiol Mol Biol Rev 62:597–635
Redko F, Clavin M, Weber D, Anke T, Martino V (2006) Search for active metabolites of Erythrina crista-galli and its endophyte Phomopsis sp. Mol Medi Chem 10:24–26
Redman RS, Kim YO, Claire JDA, Woodward CJDA, Greer C, Espino L, Doty SL, Rodriguez RJ (2011) Increased fitness of rice plants to abiotic stress via habitat adapted symbiosis: a strategy for mitigating impacts of climate change. PLOS ONE 6(7):1–10
Rodriguez RJ, Henson J, Volkenburgh EV, Hoy M, Wright L, Beckwith F, Yong-Ok Kim YO, Redman RS (2008) Stress tolerance in plants via habitat-adapted symbiosis. The ISME Journal 2:404–416
Saari S, Helander M, Faeth SH, Saikkonen K (2010) The effects of endophytes on seed production and seed predation of tall fescue and meadow fescue. Microb. Ecol 60:928–934
Saikkonen K, Wali P, Helander M, Faeth SH (2004) Evolution of endophyte-plant symbioses. Trends Plant Sci 9:275–280
Saikkonen K, Lehtonen P, Helander M, Koricheva J, Faeth SH (2006) Model systems in ecology: dissecting the endophyte-grass literature. Trends Plant Sci 11:428–433
Saikkonen K, Saari S, Helander M (2010) Defensive mutualism between plants and endophytic fungi? Fungal Diversity 41:101–113
Santiago C, Fitchett C, Murno MHG, Jalil J, Santhanam J (2012) Cytotoxic and antifungal activities of 5-Hydroxyramulosin, a compound produced by an endophytic fungus isolated from Cinnamomum mollisimum. Hindawi publishing corporation. doi:10.1155/2012/689310
Santos F, Florisvaldo C, Taicia PF, Joanita N, Marcos RM, Edson RF (2011) Endophytic fungi as a source of biofuel precursors. J Microbiol Biotechnol 21(7):728–733
Schulz B, Boyle C, Draeger S, Rommert AK, Kronh K (2002) Endophytic fungi: a source of novel biologically active secondary metabolites. Mycological Research 106:996–1004
Shalini S, Kotasthane AS (2007) Parasitism of Rhizoctonia solani by strain of Trichoderma spp. EJEAFChe 6(8):2272–2281
Shende S, Bhagwat K, Wadegaonkar P, Rai M, Varma A (2006) Piriformospora indica as a new and emerging mycofertilizer and biotizer: potentials and prospects in sustainable agriculture. In: Rai M (ed) Handbook of microbial biofertilizers. Food products press, New York, pp 477–496
Singh R, Adholeya A, Mukerji KG (2000) Mycorrhiza in control of soil-borne pathogens. In: Mukerji KG, Chamola BP, Singh J (eds) Mycorrhizal biology. Kluwer, New York, pp 173–196
Singh LP, Gill SS, Tuteja N (2011) Unraveling the role of fungal symbionts in plant abiotic stress tolerance. Plant Signaling Behav 6(2):175–191
Singh S, Bukkambudhi RG, Gudasalamani R, Uma Shaanker R, Manchanahally BS (2013) Endophytic fungi from Miquelia dentata Bedd., produce the anti-cancer alkaloid, Camptothecine. Phytomedicine 20:337–342
Song YC, Huang WY, Sun C, Wang FW, Tan RX (2005) Characterization of graphislactone a as the antioxidant and free radical-scavenging substance from the culture of Cephalosporium sp. IFB-E001, an endophytic fungus in Trachelospermum jasminoides. Biol Pharm Bull 28(3):506–509
Strobel G (2006) Harnessing endophytes for industrial microbiology. Curr Opin Microbiol 9:240–244
Strobel G, Daisy B (2003) Bioprospecting for microbial endophytes and their natural products. Microbial Mol Biol Rev 67(4):491–502
Strobel GA, Daisy B, Castillo U, Harper J (2004) Natural products from endophytic microorganisms. J Nat Prod 67(2):257–268
Strobel GA, Knighton B, Kluck K, Ren Y, Livinghouse T, Griffin M, Spakowich D, Sears J (2008) The production of myco-diesel hydrocarbons and their derivatives by the endophytic fungus Gliocladium roseum (NRRL 50072). Microbiology 154:3319–3328
Sumarah MW, Miller JD (2009) Anti-insect secondarys metabolites from fungal endophytes of conifer trees. Nat Prod Commun 4(11):1497–1504
Sun C, Johnsona JM, Caib D, Sherametia I, Oelmüllera R, Louc B (2010) Piriformospora indica confers drought tolerance in Chinese cabbage leaves by stimulating antioxidant enzymes, the expression of drought-related genes and the plastid-localized CAS protein. J Plant Physiol 167:1009–1017
Suryanarayanan TS, Thirunavukkarasu N, Govindarajulu MB, Gopalan V (2012) Fungal endophytes: an untapped source of biocatalysts. Fungal Diversity 54:19–30
Taghavi S, Lelie DV, Hoffman A, Zhang YB, Walla MD, Vangronsveld J, Newman L, Monchy S (2010) Genome sequence of the plant growth promoting endophytic bacterium Enterobacter sp. 638. PLOS Genet 6(5):1–15
Taghavi S, Weyens N, Vangronsveld J, van der Lelie D (2011) Improved phytoremediation of organic contaminants through engineering of bacterial endophytes of trees. endophytes of forest trees: biology and applications. Forestry Sci 80(4):205–216
Tan RX, Zou WX (2001) Endophytes: a rich source of functional metabolites. Nat Prod Rep 18:448–459
Tilman D et al (2002) Agricultural sustainability and intensive production practices. Nature 418:671–677
Vance CP (2001) Symbiotic nitrogen fixation and phosphorus acquisition. Plant nutrition in a world of declining renewable resources. Plant Physiology 127:390–397
Vendan RT, Yu YJ, Lee SH, Rhee YH (2010) Diversity of endophytic bacteria in Ginseng and their potential for plant growth promotion. J Microbiol 48(5):559–565
Verma VC, Kharwar RN, Gange AC (2010) Biosynthesis of antimicrobial silver nanoparticles by the endophytic fungus Aspergillus clavatus. Nanomedicine 5(1):33–40
Verza M, Arakawa NS, Lope NP, Kato MJ, Pupo MT, Said S, Carvalho I (2009) Biotransformation of a tetrahydrofuran lignin by the endophytic fungus Phomopsis sp. J Braz Chem Soc 20:195–200
Vetrivelkalai P, Sivakumar M, Jonathan EI (2010) Biocontrol potential of endophytic bacteria on Meloidogyne incognita and its effect on plant growth in bhendi. J Biopest 3(2):452–457
Waller F, Achatz B, Baltruschat H, Fodor J, Becker K, Fischer M, Heier T, Huckelhoven R, Neumann C, von Wettstein D, Franken P, Kogel KH (2005) The endophytic fungus Piriformospora indica reprograms barley to salt stress tolerance, disease resistance and higher yield. PNAS 102:13386–13391
Wang Y, Dai CC (2011) Endophytes: a potential resource for biosynthesis, biotransformation, and biodegradation. Ann Microbiol 61:207–215
Wang LW, Xu BG, Wang JY, Su ZZ, Lin FC, Zhang CL, Kubicek CP (2012) Bioactive metabolites from Phoma species, an endophytic fungus from the Chinese medicinal plant Arisaema erubescens. Appl Microbiol Biotechnol 93:1231–1239
Wu H, Yang H, You X, Li Y (2012) Isolation and characterization of Saponin-producing fungal endophytes from Aralia elata in Northeast China. Int J Mol Sci 13:16255–16266
Yang XL, Li ZZ (2013) New Spiral γ-Lactone Enantiomers from the plant endophytic fungus Pestalotiopsis foedan. Molecules 18:2236–2242
Zhang JY, Tao LY, Liang YJ, Chen LM, Mi YJ, Zheng LS, Wang F, She ZG, Lin YC, To KKW, Fu LW (2010) Anthracenedione derivatives as anticancer agents isolated from secondary metabolites of the mangrove endophytic fungi. Drugs 8(4):1469–1481
Zhao J, Zhou L, Wang J, Shan T, Zhong L, Liu X, Gao X (2010) Endophytic fungi for producing bioactive compounds originally from their host plants. Current research, technology and education topics in applied microbiology microbial biotechnology 567–576
Zhao Y, Park RD, Muzzarelli RAA (2010) Chitin deacetylases: properties and applications. Marine Drugs 8:24–46
Zhao JT, Fu YJ, Luo M, Zu YG, Wang W, Zhao CJ, Gu CB (2012) Endophytic fungi from Pigeon Pea [Cajanus cajan (L.) Mill sp.] produce Antioxidant Cajaninstilbene Acid. J Agric Food Chem 60:4314–4319
Zikmundova M, Drandarov K, Bigler L, Hesse M, Werner C (2002) Biotransformation of 2-Benzoxazolinone and 2-Hydroxy-1, 4- Benzoxazin-3-one by endophytic fungi isolated from Aphelandra tetragona. Appl Environ Microbiol 10:4863–4870
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer India
About this chapter
Cite this chapter
Kaul, S., Ahmed, M., Sharma, T., Dhar, M. (2014). Unlocking the Myriad Benefits of Endophytes: An Overview. In: Kharwar, R., Upadhyay, R., Dubey, N., Raghuwanshi, R. (eds) Microbial Diversity and Biotechnology in Food Security. Springer, New Delhi. https://doi.org/10.1007/978-81-322-1801-2_4
Download citation
DOI: https://doi.org/10.1007/978-81-322-1801-2_4
Published:
Publisher Name: Springer, New Delhi
Print ISBN: 978-81-322-1800-5
Online ISBN: 978-81-322-1801-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)