Abstract
Purpose: 4′-[methyl-11C]-thiothymidine (4DST) has been developed as an in vivo cell proliferation marker based on the DNA incorporation method. We evaluated 4DST uptake on PET in patients with newly diagnosed and recurrent gliomas and correlated the results with proliferative activity.
Methods: 4DST PET was investigated in 32 patients, including 21 with newly diagnosed gliomas and 11 with recurrent gliomas. PET imaging was performed at 15 min after 4DST injection. The standardized uptake value (SUV) was determined by region-of-interest analysis. The maximal SUV for tumor (T) and the mean SUV for contralateral normal brain tissue (N) were calculated and T/N ratio was determined. Proliferative activity as indicated by the Ki-67 index was estimated in tissue specimens.
Results: The sensitivity of 4DST PET for the detection of newly diagnosed and recurrent gliomas was 86 % and 100 %, respectively. In newly diagnosed gliomas, there was a weak correlation between T/N ratio and Ki-67 index (r = 0.45; p < 0.05). In recurrent gliomas, there was no significant difference between T/N ratio and Ki-67 index.
Conclusion: In newly diagnosed gliomas, 4DST PET seems to be useful in the noninvasive assessment of proliferation.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Markers of proliferative activity are essential for individualized patient therapy and management of brain gliomas [1]. Because tissue sampling is often obtained by stereotactic biopsy and, therefore, represents only a small part of the primary tumor, there is a probability of true malignant potential being underestimated [2]. Thus, noninvasive imaging-based technology for the detection of malignant progression is required to select the best possible treatment regimen.
Positron emission tomography (PET) is now an indispensable modality for assessment of various tumors. The radiotracer 3′-deoxy-3′-[18F]fluorothymidine (FLT) has been investigated as a promising PET tracer for evaluating tumor proliferating activity in brain tumors [3–5]. A theoretic limitation of FLT as a radiotracer for the salvage pathway of DNA synthesis is that it is not incorporated into DNA because of the lack of a 3′-hydroxyl [6].
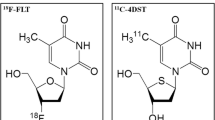
Toyohara et al. developed 4′-thiothymidine labeled with 11C at the methyl group (4′-[methyl-11C]-thiothymidine [4DST]), as a new candidate for cell proliferation imaging that is resistant to degradation by thymidine phosphorylase and is incorporated into DNA [7, 8]. A 11C-4DST PET pilot study of 6 patients with various brain tumors showed that 11C-4DST PET is feasible for brain tumor imaging and can be performed with acceptable dosimetry and pharmacologic safety at a suitable dose for adequate imaging [9]. In a mixed population of patients with newly diagnosed and recurrent gliomas, Toyota et al. has recently demonstrated that 4DST PET is feasible for evaluating cell proliferation [10]. These results indicate that 4DST has great potential for imaging cell proliferation.
The purpose of this study was to evaluate 4DST uptake in patients with newly diagnosed and recurrent gliomas and to correlate the results with proliferative activity as indicated by the Ki-67 index.
2 Materials and Methods
2.1 Patients
A total of 32 patients (15 men, 17 women) with brain gliomas who underwent 4DST PET examination were selected. Of the patients, 21 had newly diagnosed gliomas and 11 presented with recurrent gliomas that had been treated with surgery, chemotherapy and radiotherapy previously.
Pathologic diagnosis had been obtained by stereotactic biopsy or open surgery. Grading of the tumor was performed according to the World Health Organization (WHO) classification for neuroepithelial tumors [11]. Distribution of tumor grades according to WHO classification was as follows: grade II (n = 7), grade III (n = 9), and grade IV (n = 16).
2.2 4DST Synthesis and PET Imaging
The radiotracer 4DST was produced using an automated synthesis system with HM-18 cyclotron (QUPID; Sumitomo Heavy Industries Ltd, Tokyo, Japan). The 4DST was synthesized using the method described by Toyohara et al. [9].
All acquisitions were performed using a Biograph mCT 64-slice PET/CT scanner (Siemens Medical Solutions USA Inc., Knoxville, TN, USA). Data acquisition began with CT at the following settings: no contrast agent, 120 kV, 192 mA, 1.0-s tube rotation time, 3-mm slice thickness, 3-mm increments, and pitch 0.55. PET emission scanning of the head region with a 15-min acquisition of one bed position was performed 15 min after intravenous injection of 4DST (6 MBq/kg). The PET data were acquired in three-dimensional mode and were reconstructed by the baseline ordered-subset expectation maximization (OSEM) bases, with incorporating correction with point spread-function and time-of-flight model (5 iterations, 21 subsets).
2.3 Data Analysis
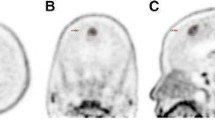
Visual image analysis was performed by an experienced nuclear physician. Tumor lesions were identified as areas of focally increased uptake, exceeding that of normal brain background.
Semiquantitative analysis was performed using the standardized uptake value (SUV). The region of interest (ROI) was placed over the entire tumor using the transverse PET image. For the reference tissue, a circular ROI of 15 × 15 mm was manually placed on the uninvolved contralateral hemisphere in the same plane that showed maximum 4DST tumor uptake. Radioactivity concentration values measured in the ROI were normalized to injected dose per patient’s body weight by calculation of SUV. The maximal SUV for tumor and the mean SUV for reference tissue were calculated. Tumor-to-contralateral normal brain tissue (T/N) ratio was determined by dividing the tumor SUV by that of the contralateral hemisphere.
2.4 Ki-67 Immunohistochemistry
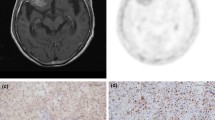
Formalin-fixed, paraffin-embedded sections of resected specimens from brain tumor were made for immunohistochemical staining. The Ki-67 index was estimated as the percentage of Ki-67-positive cell nuclei per 500–1,000 cells in the region of the tumor with the greatest density of staining.
2.5 Statistical Analysis
All semiquantitative data were expressed as mean ± SD. The Ki-67 index and the T/N ratio were compared using linear regression analysis. Differences were considered statistically significant at p < 0.05.
3 Results
3.1 4DST Uptake
In newly diagnosed gliomas, 4DST PET detected 3 of 7 grade II gliomas, all 6 grade III gliomas, and all 8 grade IV gliomas. In recurrent gliomas, 4DST PET detected all 3 grade III gliomas and all 8 grade IV gliomas. Although 4DST PET in newly diagnosed gliomas showed a slightly lower detection rate than that in recurrent gliomas (86 % vs. 100 %), the difference was not statistically significant.
3.2 4DST Uptake and Ki-67 Immunohistochemistry
In newly diagnosed gliomas, linear regression analysis indicated a weak correlation between T/N ratio and the Ki-67 index (r = 0.45, p < 0.05; Fig. 24.1a). In recurrent gliomas, there was no significant difference between T/N ratio and the Ki-67 index (r = 0.31, p = 0.36; Fig. 24.1b).
Linear regression analysis demonstrates a weak correlation between 4DST T/N ratio and proliferative activity (Ki-67 index) in newly diagnosed gliomas (r = 0.45, p < 0.05) (A). There was no significant correlation between 4DST T/N ratio and proliferative activity (Ki-67 index) in recurrent gliomas (r = 0.31, p = 0.36) (B)
4 Discussion
In the present study, we evaluated 4DST uptake in patients with newly diagnosed and recurrent gliomas. 4DST PET was feasible for imaging both newly diagnosed and recurrent gliomas. 4DST PET was found to be useful in the assessment of tumor proliferation in newly diagnosed gliomas.
Increased cell proliferation and DNA replication is a characteristic of malignant transformation [12]. The assessment of cellular proliferation rate by means of PET is useful as a noninvasive clinical approach. In a previous study, an initial clinical trial in only 6 patients with brain tumor was indicated that 4DST PET was feasible for imaging brain tumors [9]. Toyota et al. evaluated 4DST uptake and proliferative activity in 20 patients with gliomas, including 11 recurrent gliomas [10]. They showed a weak correlation between 4DST T/N ratio and Ki-67 index [10]. The present study also showed a weak correlation between 4DST T/N ratio and Ki-67 index in newly diagnosed gliomas. Minamimoto et al. evaluated 4DST uptake and Ki-67 index in patients with non-small cell lung cancer [13]. They showed a significant correlation between 4DST maximal SUV and Ki-67 index [13]. However, Ito et al. showed no significant correlation between 4DST maximal SUV and Ki-67 index in head and neck squamous cell carcinoma [14]. One possible reason for Ito et al.’s findings may be that the Ki-67 index was mainly obtained from biopsy specimens and not from resected specimens.
The present study showed no significant correlation between 4DST T/N ratio and Ki-67 index in recurrent gliomas. Eleven patients with recurrent gliomas in the present study had received chemoradiotherapy before the PET study. Radiation and chemoradiotherapy, used as an adjuvant therapy of gliomas, can cause loosening of endothelial tight junctions, vascular leakage, or endothelial cell death and increase vascular permeability [15]. Radiation could also act to increase vascular permeability not only in the blood-brain barrier (BBB) but also in the blood-tumor barrier (BTB) [15]. We suspect that in recurrent gliomas, breakdown of BBB and BTB contributes to the degree of 4DST uptake in addition to increased proliferation. Furthermore, there is biological difference between newly diagnosed and recurrent gliomas. In recurrent gliomas, recurrent tumor and treatment-induced necrosis frequently coexist.
Because the most proliferating part of the tumor is mainly responsible for tumor progression, 4DST analysis enables a more precise estimation of the malignancy. More important indication is the possibility that the cell proliferation imaging could be used for early evaluation of treatment effects. In the report by Toyohara et al., although 11C-methionine PET detected all the contrast-enhanced lesions visualized with MRI, a clinically stable tumor with contrast enhancement was not detected with 4DST [9]. The role of 4DST in therapy monitoring has not been evaluated so far. Further prospective studies involving a larger number of patients in a variety of tumor types are required to determine the clinical usefulness of 4DST PET for early evaluation of treatment response.
5 Conclusion
4DST PET was feasible for imaging both newly diagnosed and recurrent gliomas. 4DST PET seems to be useful in assessment of noninvasive tumor proliferation in newly diagnosed gliomas.
References
DeAngelis LM. Brain tumors. N Engl J Med. 2001;344:114–23.
Ceyssens S, Van Laere K, de Groot T, Goffin J, Bormans G, Mortelmans L. [11C]methionine PET, histopathology, and survival in primary brain tumors and recurrence. AJNR Am J Neuroradiol. 2006;27:1432–7.
Hatakeyama T, Kawai N, Nishiyama Y, et al. 11C-methionine (MET) and 18F-fluorothymidine (FLT) PET in patients with newly diagnosed glioma. Eur J Nucl Med Mol Imaging. 2008;35:2009–17.
Jacobs AH, Thomas A, Kracht LW, et al. 18F-fluoro-L-thymidine and 11C-methylmethionine as markers of increased transport and proliferation in brain tumors. J Nucl Med. 2005;46:1948–58.
Ullrich R, Backes H, Li H, et al. Glioma proliferation as assessed by 3′-fluoro-3′-deoxy-l-thymidine positron emission tomography in patients with newly diagnosed high-grade glioma. Clin Cancer Res. 2008;14:2049–55.
Rasey JS, Grierson JR, Wiens LW, et al. Validation of FLT uptake as a measure of thymidine kinase-1 activity in A549 carcinoma cells. J Nucl Med. 2002;43:1210–7.
Toyohara J, Kumata K, Fukushi K, Irie T, Suzuki K. Evaluation of [methyl-14C]4′-thiothymidine for in vivo DNA synthesis imaging. J Nucl Med. 2006;47:1717–22.
Toyohara J, Okada M, Toramatsu C, Suzuki K, Irie T. Feasibility studies of 4′-[methyl-11C]thiothymidine as a tumor proliferation imaging agent in mice. Nucl Med Biol. 2008;35:67–74.
Toyohara J, Nariai T, Sakata M, et al. Whole-body distribution and brain tumor imaging with 11C-4DST: a pilot study. J Nucl Med. 2011;52:1322–8.
Toyota Y, Miyake K, Kawai N, et al. Comparison of 4′-[methyl-11C]thiothymidine (11C-4DST) and 3′-deoxy-3′-[18F]fluorothymidine (18F-FLT) PET/CT in human brain glioma imaging. EJNMMI Res. 2015;5:7.
Louis DN, Ohgaki H, Wiestler OD, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114:97–109.
la Fougère C, Suchorska B, Bartenstein P, Kreth FW, Tonn JC. Molecular imaging of gliomas with PET: opportunities and limitations. Neuro Oncol. 2011;13:806–19.
Minamimoto R, Toyohara J, Seike A, et al. 4′-[Methyl-11C]-thiothymidine PET/CT for proliferation imaging in non-small cell lung cancer. J Nucl Med. 2012;53:199–206.
Ito K, Yokoyama J, Miyata Y, et al. Volumetric comparison of positron emission tomography/computed tomography using 4′-[methyl-11C]-thiothymidine with 2-deoxy-2-18F-fluoro-D-glucose in patients with advanced head and neck squamous cell carcinoma. Nucl Med Commun. 2015;36:219–25.
Cao Y, Tsien CI, Shen Z, et al. Use of magnetic resonance imaging to assess blood-brain/blood-gliomas barrier opening during conformal radiotherapy. J Clin Oncol. 2005;23:4127–36.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is distributed under the terms of the Creative Commons Attribution-Noncommercial 2.5 License (http://creativecommons.org/licenses/by-nc/2.5/) which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
The images or other third party material in this chapter are included in the work’s Creative Commons license, unless indicated otherwise in the credit line; if such material is not included in the work’s Creative Commons license and the respective action is not permitted by statutory regulation, users will need to obtain permission from the license holder to duplicate, adapt or reproduce the material.
Copyright information
© 2016 The Author(s)
About this paper
Cite this paper
Yamamoto, Y., Nishiyama, Y. (2016). Correlation of 4′-[methyl-11C]-Thiothymidine Uptake with Ki-67 Immunohistochemistry in Patients with Newly Diagnosed and Recurrent Gliomas. In: Kuge, Y., Shiga, T., Tamaki, N. (eds) Perspectives on Nuclear Medicine for Molecular Diagnosis and Integrated Therapy. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55894-1_24
Download citation
DOI: https://doi.org/10.1007/978-4-431-55894-1_24
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55892-7
Online ISBN: 978-4-431-55894-1
eBook Packages: MedicineMedicine (R0)