Abstract
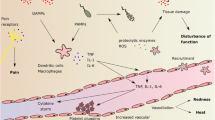
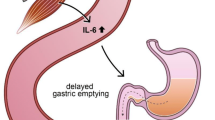
The metabolic impact of infectious, neoplastic and certain other disease states have long been known to clinicians. Those diseases in general induce, aside specific symptoms associated with the disease itself, general signs and symptoms [1]. These alterations include fever, hypermetabolism, cachexia, leukocytosis, hypo-ferremia and marked changes in carbohydrate-, protein- and lipid metabolism, resulting in hyperglycemia, sometimes followed by hypoglycemia, increased glucose production, protein catabolism, hypertriglyceridemia, increased free fatty acid (FFA) turnover and decreased ketonemia [2, 3]. It was once believed that these metabolic derangements were induced by the disease itself and that differences in the metabolic response were due to differences between disease states. In recent years however, there has been a growing awareness that the metabolic responses to disease tend to occur in a consistent and predictable manner and that endogenous mediators could be essential elements in the pathogenesis of above-mentioned responses [1, 3, 4]. Some of these mediators, the “classical” hormones are well-known for a longtime: corticosteroids, catecholamines, glucagon, growth-hormone and insulin. The accepted explanation for the changes in glucose-, protein-, and fat metabolism were marked increases in the plasma concentrations of the counter-regulatory hormones together with decreased tissue sensitivity to insulin [2]. These explanations have been substantiated by recent studies [3]. In healthy volunteers was shown that hypermetabolism, hyperglycemia, but not hypoglycemia, protein loss and increased lipolysis can be induced by the infusion of the counter-regulatory hormones. Simultaneous infusion of these hormones was necessary, as separate infusion of each of them provided lesser and sometimes only transient metabolic changes. These metabolic changes however are sometimes also seen despite seemingly normal plasma concentrations of the counter-regulatory hormones [5] whereas other metabolic responses like fever, acute phase protein synthesis, hypoferremia and leukocytosis could be induced only by injection of an inflammatory agent like etiocholanon [3, 4].
Chapter PDF
Similar content being viewed by others
Keywords
- Rest Energy Expenditure
- Human Immune Deficiency Virus
- Insulin Clearance
- Recombinant Human Tumor Necrosis Factor
- Human Immune Deficiency Virus Infection
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Beutler B, Cerami A (1987) Cachectin: more than a tumor necrosis factor. N Engl J Med 316:379–385
Evans RD, Argilés JM, Williamson DH (1989) Metabolic effects of tumor necrosis factoralfa (cachectin) and interleukin-1. Clin Sci 77:357–364
Watters JM, Wilmore DW (1989) The metabolic responses to trauma and sepsis. In: DeGroot LJ (ed) Endocrinology. Philadelphia, pp 2367–2393
Watters JM, Bessey PQ, Dinarello EA, Wolff SM, Wilmore DW (1986) Both inflammatory and endocrine mediators stimulate most responses to sepsis. Arch Surg 121:179–190
Romijn JA (1990) On metabolic modulation by starvation and disease. Thesis, University of Amsterdam
Warren RS, Donner DB, Stames HF, Brennan MF (1987) Modulation of endogenous hormone action by recombinant human tumor necrosis factor. Proc Natl Acad Sci 84:8619–8622
Fong Y, Moldawer LL, Shires T, Lowry SF (1990) The biologic characteristics of cytokines and their implication in surgical injury. Surg Gyn Obstetr 170:363–378
Bendtzen K (1988) Interleukin-1, interleukin-6 and tumor necrosis factor in infection, inflammation and immunity. Immunol Lett 19:183–192
van Deventer SJH, Buller HR, ten Cate JW, Aarden LA, Hach CE, Sturk A (1990) Experimental endotoxemia in humans: analysis of cytokine release and coagulation, fibrinolytic and complement pathways. Blood (in press)
Fong Y, Marano MA, Moldawer LL, et al. (1990) The acute splanchnic and peripheral tissue metabolic response to endotoxin in humans. J Clin Invest 85:1896–1904
Tracey KJ, Vlassara H, Cerami A (1989) Cachectin/tumor necrosis factor. Lancet 1:1122–1127
Beutler B, Mahoney J, LeTrang N, Pehala P, Cerami A (1985) Purification of cachectin, a lipoprotein lipase suppressing hormone secreted by endotoxin-induced RAW 264.7 cells. J Exp Med 161:984–995
Semb H, Peterson J, Tavernier J, Olivecrona T (1987) Multiple effects of tumor necrosis factor on lipoprotein lipase. J Biol Chem 262:8390–8394
Feingold KR, Soued M, Staprans I, et al. (1989) Effect of tumor necrosis factor (TNF) on lipid metabolism in the diabetic rat. Evidence that inhibition of adipose tissue lipoprotein lipase activity is not required for TNF-induced hyperlipidemia. J Clin Invest 83:1116–1121
Kern PA (1988) Recombinant human tumor necrosis factor does not inhibit lipoprotein lipase in primary cultures of isolated human adipocytes. J Exp Res 29:909–914
Vaantjes WJ, de Haas CGM, Houweling M (1990) Acute effects of interleukin 1-alfa and interleukin-6 on intermediary metabolism in freshly isolated vat hepatocytes. Bioch Bioph Res Comm 169:623–628
Hill MR, Stith RD, McCallum RE (1986) Interleukin-1 : a regulatory role in glucocorticoid-regulated hepatic metabolism. J Immunol 137:858–862
Tredget EE, Yu YM, Zhong S, et al. (1988) Role of interleukin-1 and tumor necrosis factor on energy metabolism in rabbits. Am J Phys 255:E760–E768
Fong Y, Tracey KJ, Moldawer LL, et al. (1989) Antibodies to cachectin/tumor necrosis factor reduce interleukin-1 bèta and interleukin-6 appearance during lethal bacteremia. J Exp Med 170:1627–1683
Michie HR, Spriggs DR, Manogue KR, et al. (1988) Tumor necrosis factor and endotoxin induce similar metabolic responses in human beings. Surgery 104:280–286
Starnes HF Jr, Warren RJ, Jeevanandan M, et al. (1988) Tumor necrosis factor and the acute metabolic reponse to tissue injury in man. J Clin Invest 82:1321–1325
Balkwill F, Osborne R, Burke F, et al. (1987) Evidence for tumor necrosis factor/cachectin production in cancer. Lancet 2:1229–1232
Tracey KJ, Wei H, Manogue KR, et al. (1988) Cachectin/tumor necrosis factor induces cachexia anemia and inflammation. J Exp Med 167:1211–1227
v. d. Poll T, Romijn JA, Endert E, Borm JJJ, Büller HR, Sauerwein HP (1990) Tumor necrosis factor mimics the metabolic response to acute infections in healthy humans. Clin Nutrition 9:5 (abstract)
Hommes MJT (1990) Host metabolism in HIV infection. Thesis, University of Amsterdam
Hommes MJT, Romijn JA, Godfried MH, et al. (1991) Increased resting energy expenditure in HIV-infected men. Metabolism (in press)
Hommes MJT, Romijn JA, Endert E, Sauerwein HP (1991) Resting energy expenditure and substrate oxidation in human immunodeficiency virus (HIV)- infected asymptomatic men. Am J Clin Nutr (in press)
Hommes MJT, Romijn JA, Endert E, Eeftinck-Schattenkerk JKM, Sauerwein HP (1991) Insulin sensitivity and insulin clearance in HIV infected men. Metabolism (in press)
Hommes MJT, Romijn JA, Endert E, Eeftinck-Schattenkerk JKM, Sauerwein HP (1991) Basal fuel hemostasis in symptomatic human immunodeficiency virus infection. Clin Science (in press)
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1991 Springer-Verlag Berlin, Heidelberg
About this paper
Cite this paper
Sauerwein, H.P., van der Poll, T., Romijn, J.A. (1991). Cytokines: Role in Human Metabolism. In: Vincent, J.L. (eds) Update 1991. Update in Intensive Care and Emergency Medicine, vol 14. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-84423-2_29
Download citation
DOI: https://doi.org/10.1007/978-3-642-84423-2_29
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-53672-7
Online ISBN: 978-3-642-84423-2
eBook Packages: Springer Book Archive