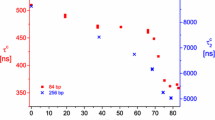
Abstract
In this paper we give an outlook on the problem pertinent to the structural stability of hydrated double-stranded (duplex) M +1 -DNA salts (M +1 stands for a monovalent counterion like Na+, K+, Rb+) in fibers and possibly in solutions. In particular we call attention to the processes which may occur at the interface between the polyion and its aqueous surroundings and to their relations with external control parameters like water activity aw and ionic strength I.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Pleasant reading is Watson’s book: Molecular Biology of the Gene, W.A. Benjamin Inc. (1977)
See, for instance, the masterpiece by Hess’s group at the Max Planck Institut of Dortmund (Germany); for a recent review, see: G. Gerisch, B. Hess: Proc. Nat. Acad. Sci. USA 71, 2118 (1974)
See, for instance: M.G. Rutten: In The Origin of Life by Natural Causes (Elsevier, Amsterdam 1971);
A.I. Oparin: In Genesis and Evolutionary Development of Life (Academic, New York 1968)
See, for instance: M. Eigen, P. Schuster: In The Hypercycle (Springer, Berlin, Heidelberg, New York 1979)
G. Nicolis, I- Prigogine: Self-Organization in Non-Equilibrium Systems (Wiley, New York, London, Sydney, Toronto 1977)
H. Haken: In Synergetics: An Introduction (Springer, Berlin, Heidelberg, New York 1978) and in books of his Synergetics Series
E. Del Giudice et al.: Phys. Lett. 85A, 402 (1982);
see also: Coherent Excitations of Biological Systems, ed. by H. Fröhlich, F. Kremer (Springer, Berlin, Heidelberg, New York 1983)
M. Levitt: Proc. Natl. Acad. Sci. USA 73, 640 (1978)
See also M. Sundaralingam, E. Westhoff: In Biomolecular Stereodynamics (Adenine Press 1981)
Henry M. Sobell: In Nucleic Acid Geometry and Dynamics (Pergamon Press, New York 1980)
Struther Arnott, Rengaswami Chandrasekaran: In Biomolecular Stereodynamics (Adenine Press, New York 1981, and references therein)
Richard E. Dickerson et al.: Science 216, 475 (1982) and references therein
P. De Santis et al.: Biopolymers 20, 1707 (1981) and references therein
M. Falk et al.: J. Am. Chem. Soc. 84, 3843 (1962)
E. Clementi, G. Corogiu: In Biomolecular Stereodynamics (Adenine Press New York 1981);
see also: Structure and Dynamics: Nucleic Acids and Proteins (Adenine Press, New York 1983)
B.N. Conner et al.: Nature (London) 295, 294 (1982)
See, for instance: Madeleine Lüscher-Mattle: Biopolymers 21, 419 (1982)
A.G.W. Leslie et al.: J. Mol. Biol. 143, 49 (1980)
See, for instance: A. Katchalsky: In Macromolecules. IUPAC Symposium (Butterworths, London 1971) and references therein;
see also: M. Guéron, G. Weisbuch: Biopolymers 19, 353 (1980);
and F. Oosawa: In Polyelectrolytes (Dekker Inc., New York 1971)
E. Clementi: In Structure and Dynamics: Nucleic Acids and Proteins (Adenine Press, New York 1983)
K. Zakrzewska et al.: In Biomolecular Stereodynamics (Adenine Press, New York 1981)
The shell-mode coupling involving asymmetric stretching and symmetric bending certainly affects the P-01, P-02 double-bond character, but it is expected not to affect directly the backbone conformation as far as the negative charge delocalization concerns only the two “pendent” oxygens. If, however, such delocalization would involve ester oxygens, a change in dihedral angles α and ζ will then affect the resonance integrals in the π-like system of orbitals of the whole phosphate group, so that an (α,ζ)-dependent energy term would appear. Of course, the actual situation will depend on the strength and symmetry of the ligand fields (absorbed water molecules and possibly coordinated counterions). This state of affairs may be tentatively approached in terms of a mean field theory following the same method as in ferroelectrics. (See, for instance, M. Tokunaga, M. Matsubara: Progr. Theor. Phys. 35, 581 (1966)
N.C. Seeman: In Nucleic Acid Geometry and Dynamics (Pergamon, New York 1979) Sarma ed. Furthermore, the A conformation should favour at low aw the onset of salt bridges between neighbouring phosphate groups on opposite strands
J. Wittenburg: In Dynamics of Systems of Rigid Bodies (Teubner, Stuttgart 1977)
During the elaboration of the present analysis we became aware of the work of De Santis (see Ref. [10]) where the same type of argument is employed
In the rough approximation in which β is kept constant at “trans” (t) position (β~180°), Eq. (14) would also imply cosγ = 0, or cosγ = -1. The former solution may be ascribed to both g+ or g- ranges; if ascribed to g+, this result appears to be consistent with the experimental observation (see Sect.2). Furthermore, the constancy of both β and γ implying two more equations (contraints) among α,ε,ζ allows one to choose ζ as relevant conformational parameter. Of course, for fixed δ, the other parameters become functions of the free ζ.
It appears that the stability of the water spine in the m.g. depends on base sequence, it is then a problem which should be treated in the framework of DNA genetic-dependent properties (see the introductory section)
Alternatively, by remembering the strict relation existing between sugar puckering and pseudorotation, one may think of a situation in which the energy barrier for (C2′-endo)→(C3′-endo) transition of sugar is controlled by the fluctuations in phosphate-group rotation ζ and, vice versa, the energy barrier for (t)→(g+) transition of phosphate group rotation ζ is controlled by fluctuations in sugar pseudorotation. Such a situation should be formally similar to that of dynamic barriers occurring in proteins, as suggested by H. Frauenfelder: Biochemistry 23, 5147 (1980)
See, for instance: H.E. Stanley: J. Phys. A12, 1329 (1979);
see also: E. Stoll, C. Domb: J. Phys. A12, 1843 (1979)
E. Clementi: In Lecture Notes in Chemistry, Vol.19 (Springer, Berlin, Heidelberg, New York 1980)
Fumio Oosawa: In Polyelectrolytes (Dekker, New York 1971)
See, for instance: T.L. Hill: Introduction to Statistical Thermodynamics (Addison-Wesley 1960)
See, for instance: H. Teitelbaum, S.W. Englander: J. Mol. Biol. 92, 79 (1985)
Note that the relation of Eq. (25) with second-order differential entropy ‘(see, for instance: Nicolis and Prigogine, Ref. [4] is trivial: \( {({\delta^2}S)_0} \equiv - (1/T)\sum\limits_{{ij}} {{{({\partial^2}G/\partial {\xi_i}\partial {\xi_j})}_0}\partial {\xi_i}\partial {\xi_j}} \) where the subscript (0) denotes “reference state” and {∂ξj} stands for the order parameters ζ,σ,ξ,θA,θB1,θB2; further considerations on kinetic properties of fluctuations, namely terms involving ∇ξj(Z,r,φ;t), would lead us to the wanted relaxation equations, at least for regimes near to equilibrium
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1983 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Nardelli, G.F., Bracale, M., Signorini, C., Zucchelli, G. (1983). Stability of Hydrated M +1 -DNAs: A Challenge in the Theory of Nonlinear Systems. In: Benedek, G., Bilz, H., Zeyher, R. (eds) Statics and Dynamics of Nonlinear Systems. Springer Series in Solid-State Sciences, vol 47. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-82135-6_6
Download citation
DOI: https://doi.org/10.1007/978-3-642-82135-6_6
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-82137-0
Online ISBN: 978-3-642-82135-6
eBook Packages: Springer Book Archive