Abstract
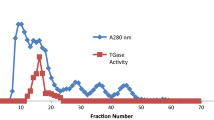
Homogeneous glycogen synthetase b has been obtained from both trout liver and rat liver. A molecular weight in the range of 260,000 was obtained for the enzyme from both sources. The trout enzyme appears to have less affinity for homologous glycogen than the rat enzyme as evidenced by the lesser fraction of the former which sediments with the glycogen pellet and by the dissociation of the glycogen-enzyme complex from the former but not the latter on DEAE-cellulose columns. A subunit molecular weight of 85,000 was determined for the rat enzyme, suggesting that it exists as a trimer. Further aggregation to a component of twice the original size occurs on storage in the cold. Determinations of alkali-labile phosphate and reactive sulfhydryl groups gave values of 12.4 and about 6, respectively. Activity decreased linearly to zero with the titration of the first 3 sulfhydryl groups (or the first 2 when glycogen was present), indicating that at least one of the most reactive groups is essential for activity. Incubation of the purified synthetase at 0° led to inactivation which was reversed on rewarming. Glycogen protected against this cold inactivation. Homogeneous rat liver synthetase b serves as a substrate for synthetase phosphatase which is currently being purified.
Supported by a grant (AM-08873) from the National Institutes of Health.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Abbreviations
- cAMP:
-
cyclic adenosine 3′, 5′-phosphate
- UDPG:
-
UDP-glucose
- G-6-P:
-
glucose 6-phosphate
- SDS:
-
sodium dodecyl sulfate
- DTNB:
-
5, 5′-dithiobis (2-nitrobenzoic acid)
References
Ames, B. N. (1966), Methods in Enzymology 8, 116.
Assaf, S. A., and Yunis, A. A. (1971), FEBS (Fed. Eur. Biochem. Soc.) Lett. 19, 22.
Brown, N. E., and Larner, J. (1971), Biochim. Biophys. Acta 242, 69.
Derechin, M., Rustum, Y. M., and Barnard, E. A. (1972), Biochemistry 11, 1793.
De Wulf, H., Stalmans, E., and Hers, H. G. (1970), Eur. L Biochem. 15, 1.
Ellman, G. L. (1959), Arch. Biochem. Biophys. 82, 70.
Gold, A. H. (1970), Biochemistry 9, 946.
Gruhner, K., and Segal, H. L. (1970), Biochim. Biophys. Acta 222, 508.
Hornbrook, K. R., Burch, H. B., and Lowry, O. H. (1965), Biochem. Biophys. Res. Commun. 18, 206.
Hornbrook, K. R., Burch, H. B., and Lowry, O. H. (1966), Mol. Pharmacol. 2, 106.
Issa, A. I., and Mendicino, J. (1973), L Biol. Chem. 248, 685.
Kato, K., and Bishop, J. S. (1972), L Biol. Chem. 247, 7420.
Lin, D. C., Segal, H. L., and Massaro, E. J. (1972), Biochemistry 11, 4466.
Lin, D. C., Segal, H. L., and Massaro, E. J. (1972), Biochemistry 11, 4466.
Mersmann, H. J., and Segal, H. L. (1967), Proc. Nat. Acad. Sci. U.S. 58, 1688.
Mersmann, H. J., and Segal, H. L. (1969), L Biol. Chem. 244, 1701.
Miller, T. B., Jr., and Larner, J. (1973), L. Biol. Chem. 248, 3483.
Rose11-Perez, M., and Larner, J. (1964), Biochemistry 3, 81.
Rosell-Pérez, M. (1972), Ital. L Biochem. 21, 34.
Rothman-Denes, L. B., and Cabib, E. (1971), Biochemistry 10, 1236.
Sanada, Y., and Segal, H. L. (1971), Biochem. Biophys. Res. Commun. 45, 1159.
Sevall, J. S., and Kim, K. H. (1970), Biochim. Biophys. Acta 206, 359.
Smith, C. H., Brown, N. E., and Larner, J. (1971), Biochim. Biophys. Acta 242, 81.
Soderling, T. R., Hickenbottom, J. P., Reimann, E. M., Hunkeler, F. L., Walsh, D. A., and Krebs, E. G. (1970), L Biol. Chem. 245, 6317.
Stalmans, W., De Wulf, H., and Hers, H. G. (1971), Eur. L Biochem. 18, 582.
Stalmans, W., De Wulf, H., Lederer, B., and Hers, H. G. (1970), Eur. L Biochem. 15, 9.
Staneloni, R. J., and Piras, R. (1971), Biochem. Biophys. Res. Commun. 42, 237.
Steiner, D. F., Younger, L., and King, J. (1965), Biochemistry 4, 740.
Tanford, C., Kawahara, K., and Lapanje, S. (1967), L Amer. Chem. Soc. 89, 729.
Thomas, J. A., Schiender, K. K., and Larner, J. (1968), Anal. Biochem. 25, 486.
Ullman, A., Goldberg, M. E., and Monod, J. (1968), Biochemistry 7, 261.
Weber, K., and Osborn, M. (1969), L Biol. Chem. 244, 4406.
Yphantis, D. A. (1964), Biochemistry 3, 297.
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 1974 Springer-Verlag Berlin · Heidelberg
About this paper
Cite this paper
Segal, H.L., Lin, D.C. (1974). Liver Glycogen Synthetase. In: Metabolic Interconversion of Enzymes 1973. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-80817-3_8
Download citation
DOI: https://doi.org/10.1007/978-3-642-80817-3_8
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-80819-7
Online ISBN: 978-3-642-80817-3
eBook Packages: Springer Book Archive