Abstract
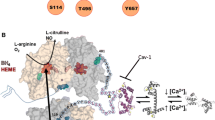
Numerous investigations have indicated that in both health and disease the shortlived radical nitric oxide (NO) is a key effector in the vascular system (Fig. 1). NO is generated by a five-electron oxidation of one of the terminal guanidino nitrogen atoms of l-arginine, catalyzed by NO synthases, and reaches the surrounding target cells by simple diffusion. The principal physiological source of NO in the vascular system is the endothelium, which constitutively expresses a NO synthase. The most important functions of endothelium-derived NO are the control of blood flow, and hence the supply of oxygen to organs, and the control of blood cell interaction with the vascular wall. Under certain pathophysiological conditions, endothelial cells are no longer the main source of NO. Indeed, in most types of vascular cells (e.g., vascular smooth muscle cells, macrophages, fibroblasts and endothelial cells) a NO producing pathway is induced following exposure to cytokines, such as interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α), the levels of which are elevated in response to infection and injury. NO generated by this pathway in large amounts may account, at least in part, for the cytotoxic effect of macrophages and thus play a crucial role in host defense. The finding that NO can have such contrasting effects, i.e., to be protective and yet also cytotoxic, may be explained by the involvement of two NO-generating systems regulated by distinctly different mechanisms.
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Abu-Soud HM, Stuehr DJ (1993) Nitric oxide synthases reveal a role for calmodulin in controlling electron transfer. Proc Natl Acad Sci USA 90: 10769–10772
Aisaka K, Gross SS, Griffith OW, Levi R (1989) NG-methylarginine, an inhibitor of endothelium-derived nitric oxide synthesis, is a potent pressor agent in the guinea pig — does nitric oxide regulate blood pressure in vivo. Biochem Biophys Res Commun 160: 881–886
Busconi L, Michel T (1993) Endothelial nitric oxide synthase: N-terminal myristoylation determines subcellular localization. J Biol Chem 268: 8410–8413
Busse R, Fleming I (1993) The endothelial organ. Curr Opin Cardiol 8: 719–727
Busse R, Mülsch A (1990a) Calcium-dependent nitric oxide synthesis in endothelial cytosol is mediated by calmodulin. FEBS Lett 265: 133–136
Busse R, Mülsch A (1990b) Induction of nitric oxide synthase by cytokines in vascular smooth muscle cells. FEBS Lett 275: 87–90
Busse R, Lückhoff A, Bassenge E (1987) Endothelium-derived relaxant factor inhibits platelet activation. Naunyn-Schmiedebergs Arch Pharmacol 336: 566–571
Busse R, Hecker M, Fleming I (1994) Control of nitric oxide and prostacyclin synthesis in endothelial cells. Arzneimittel Forschung/Drug Res 44 [Suppl]: 392–396
Cho HJ, Xie QW, Calaycay J, Mumford RA, Swiderek KM, Lee TD, Nathan C (1992) Calmodulin is a subunit of nitric oxide synthase from macrophages. J Exp Med 176: 599–604
Corbett JA, Sweetland MA, Lancaster JR, McDaniel ML (1993) A 1-hour pulse with IL-113 induces formation of nitric oxide and inhibits insulin secretion by rat islets of Langerhans: evidence for a tyrosine kinase signaling mechanism. FASEB J 7: 369–374
Craven PA, DeRubertis FR (1978) Restoration of the resposiveness of purified guanylyl cyclase to nitrosoguanidine, nitric oxide, and related activators ba heme and hemeproteins. J Biol Chem 253: 8433–8443
Dimmeler S, Lottspeich F, Brune B (1992) Nitric oxide causes ADP-ribosylation and inhibition of glyceraldehyde-3-phosphate dehydrogenase. J Biol Chem 267: 1 6771–16774
Drapier JC, Hibbs JB (1986) Murine cytotoxic activated macrophages inhibit aconitase in tumor cells. Inhibition involves the iron-sulfur prosthetic group and is reversible. J Clin Invest 78: 790–797
Durante W, Schini VB, Catovsky S, Kroll MH, Vanhoutte PM, Schafer Al (1993) Plasmin potentiates induction of nitric oxide synthesis by interleukin-1 3 in vascular smooth muscle cells. Am J Physiol 264: H617 — H624
Durante W, Schini VB, Kroll MH, Catovsky S, Scott-Burden T, White JG, Vanhoutte PM, Schafer Al (1994) Platelets inhibit the induction of nitric oxide synthesis by interleukin-1)3 in vascular smooth muscle cells. Blood 83: 1831–1838
Fleming I, Gray GA, Schott C, Stoclet JC (1991) Inducible but not constitutive production of nitric oxide by vascular smooth muscle cells. Eur J Pharmacol 200: 375–376
Fleming I, Hecker M, Busse R (1994) Intracellular alkalinization induced by bradykinin sustains activation of the constitutive nitric oxide synthase in endothelial cells. Circ Res 74: 1220–1226
Förstermann U, Pollock JS, Schmidt HHHW, Heller M, Murad F (1991) Calmodulin-dependent endothelium-derived relaxing factor/nitric oxide synthase activity is present in the particulate and cytosolic fractions of bovine aortic endothelial cells. Proc Natl Acad Sci USA 88: 1788–1792
Furchgott Rf, Vanhoutte PM (1989) Endothelium-derived relaxing and contracting factors. FASEB J 3: 2007–2018
Galle J, Bauersach J, Bassenge E, Busse R (1993) Arterial size determines the enhancement of contractile responses after supression of endothelium-derived relaxing factor formation. Pflugers Arch 422: 564–569
Gardiner SM, Compton AM, Bennett T, Palmer RMJ, Moncada S (1990) Control of regional blood flow by endothelium-derived nitric oxide. Hypertension 15: 486–492
Garg UC, Hassid A (1989) Nitric oxide-generating vasodilators and 8-bromo-cyclic guanosine mono-phosphate inihibit mitogenesis and proliferation of cultured rat vascular smooth muscle cells. J Clin Invest 83: 1774–1777
Griffith TM, Edwards DH, Lewis MJ, Newby AC, Henderson AH (1984) The nature of endothelium-derived vascular relaxant factor. Nature 308: 645–647
Gross SS, Levi R (1992) Tetrahydrobiopterin synthesis. J Biol Chem 267: 25722–25729
Gruetter CA, Barry BK, McNamara BD, Gruetter DY, Kadowitz PJ, Ignarro LJ (1979) Relaxation of bovine coronary artery and activation of coronary arterial guanylate cyclase by nitric oxide, nitoprusside and a carcinogenic nitrosoamine. J Cycl Nucl Res 5: 211–224
Hecker M, Mülsch A, Bassenge E, Busse R (1993) Vasoconstriction and increased flow: two principle mechanisms of shear stress-dependent endothelial autacoid relase. Am J Physiol 265: H828 — H833
Hecker M, Mülsch A, Bassenge E, Förstermann U, Busse R (1994) Subcellular localization and characterization of nitric oxide synthase(s) in endothelial cells–Physiologic implications. Biochem J 299: 247–252
Hibbs JB, Taintor RR, Vavrin Z, Rachlin EM (1988) Nitric oxide: a cytotoxic activated macrophage effector molecule. Biochem Biophys Res Commun 157: 87–94
lyengar R, Stuehr DJ, Marlette MA (1987) Macrophage synthesis of nitrite, nitrate, and N-nitrosamines: precursors and role of the respiratory burst. Proc Natl Acad Sci USA 84: 6369–6373
Julou-Schaeffer G, Gray GA, Fleming I, Parratt JR, Stoclet JC (1991) Activation of the L-arginine pathway is involved in vascular hyporeactivity induced by endotoxin. J Cardiovasc Pharmacol 17: S207 - S212
Karaki H, Sato K, Ozaki H, Murakami K (1988) Effects of sodium nitroprusside on cytosolic calcium level in vascular smooth muscle. Eur J Pharmacol 156: 259–266
Katsuki S, Arnold WP, Mittal CK, Murad F (1977) Stimulation of guanylate cyclase by sodium nitroprusside, nitroglycerin and nitric oxide in various tissue preparations and comparison to the effects of sodium azide and hydroxylamine. J Cycl Nucl Prot Phosphoryl Res 3: 23–35
Keaney JF, Simon DI, Stamler JS, Jaraki O, Scharfstein J, Vita JA, Loscalzo J (1993) NO forms an adduct with serum albumin that has endothelium-derived relaxing factor-like properties. J Clin Invest 91: 1582–1589
Kilbourn RG, Belloni P (1990) Endothelial cell production of nitrogen oxides in response to interferon-gamma in combination with tumor necrosis factor, interleukin-1, or endotoxin. J Natl Cancer I ns 82: 772–776
Koide M, Kawahara Y, Nakayama I, Tsuda T, Yokoyama M (1993) Cyclic AMP elevating agents induce an inducible type of nitric oxide synthase in cultured vascular smooth muscle cells. J Biol Chem 268: 24959–24966
Kourembanas S, McQuillan LP, Leung GK, Faller DV (1993) Nitric oxide regulates the expression of vasoconstrictors and growth factors by vascular endothelium under both normoxia and hypoxia. J Clin Invest 92: 99–104
Kunz D, Mühl H, Walker G, Pfeilschifter J (1994) Two distinct signalling pathways trigger the expression of inducible nitric oxide synthase in rat mesangial cells. Proc Natl Acad Sci USA 91: 5387–5391
Lamas S, Marsden PA, Li GK, Tempst P, Michel T (1992) Endothelial nitric oxide syntahse: molecular cloning and characterization of a distinct constitutive enzyme isoform. Proc Natl Acad Sci USA 89: 6348–6352
Lamontagne D, Pohl U, Busse R (1992) Mechanical deformation of vessel wall and shear stress determine the basal release of endothelium-derived relaxing factor in the intact rabbit coronary vascular bed. Circ Res 70: 123–130
Marczin N, Papapetropoulos A, Catravas JD (1993) Tyrosine kinase inhibitors suppress endotoxin-and IL-ill-induced NO synthesis in aortic smooth muscle cells. Am J Physiol 265: H1014 - H1018
Marsden PA, Scheppert KT, Chen HS, Flowers M, Sundell CI, Wilcox JN, Lamas S, Michel T (1992) Molecular cloning and characterization of human endothelial nitric oxide synthase. FEBS Lett 307: 287–293
Martin W, Furchgott RF, Villani GM, Jothianandan D (1986) Depression of contractile responses in rat aorta by spontaneously released endothelium-derived relaxing factor. J Pharmacol Exp Ther 237: 529–538
McIlion BT, Ignarro LJ, Ohlstein EH, Pontecorvo EG, Hyman AL, Kadowitz PJ (1981) Evidence for the inhibitory role of guanosine 3’,5’-monophosphate in ADP-induced human platelet aggregation in the presence of nitric oxide and related vasodilators. Blood 57: 946–955
Molina-y-Vedia L, McDonald B, Reep B, Brüne B, DiSilvio M, Billiar TR, Lapetina EG (1992) Nitric oxide-induced S-nitrosylation of glyceraldehyde-3-phosphate dehydrogenase inhibits enzymatic activity and increases endogenous ADP-ribosylation. J Biol Chem 267: 24929–24932
Moore PK, Alswayeh OA, Chong NWS, Evans RA, Gibson A (1990) L-N3-nitro arginine (L-NOARG), a novel L-arginine-reversible inhibitor of endothelium-dependent vasodilatation in vitro. Br J Pharmacol 99: 408–412
Mülsch A, Busse R (1990) NG-nitro-L-arginine (N5-[iminolnitroamino)methyll-L-ornithine) impairs endothelium-dependent dilations by inhibiting cytosolic nitric oxide synthesis from L-arginine. NaunynSchmiedebergs Arch Pharmacol 341: 143–147
Nathan C (1992) Nitric oxide as a secretory product of mammalian cells. FASEB J 6: 3051–3064
Nishida K, Harrison DG, Navas JP, Fisher AA, Dockery Sp, Uematsu M, Nerem RM, Alexander RW, Murphy TJ (1992) Molecular cloning and characterization of the constitutive bovine aortic endothelial cell nitric oxide synthase. J Clin Invest 90: 2092–2096
Pohl U, Lamontagne D, Bassenge E, Busse R (1994) Attenuation of coronary autoregulation in the isolated rabbit heart by endothelium derived nitric oxide. Cardiovasc Res 28: 414–419
Rees DD, Palmer RM, Moncada S (1989e) Role of endothelium-derived nitric oxide in the regulation of blood pressure. Proc Natl Acad Sci USA 86: 3375–3378
Rees DD, Palmer RMJ, Hodson HF, Moncada S (1989b) A specific inhibitor of nitric oxide formation from L-arginine attenuates endothelium-dependent relaxation. Br J Pharmacol 96: 418–424
Schini VB, Durante W, Elizondo E, Scott-Burden T, Junquero DC, Schafer Al, Vanhoutte PM (1992) The induction of nitric oxide synthase activity is inhibited by TGFj31, PDGFAB and PDGFBB in vascular smooth muscle cells. Eur J Pharmacol 216: 379–383
Schini VB, Catovsky S, Durante W, Scott-Burden T, Schafer Al, Vanhoutte PM (1993) Thrombin inhibits induction of nitric oxide synthase in vascular smooth muscle cells. Am J Physiol 264: H611–H616
Schray-Utz B, Zeiher AM, Busse R (1993) The expression of monocyte chemoattractant protein (MCP-1) mRNA in human endothelial cells is modulated by nitric oxide. FASEB J 7: A130
Scott-Burden T, Schini VB, Elizondo E, Junquero DC, Vanhoutte PM (1992) Platelet-derived growth factor suppresses and fibroblast growth factor enhances cytokine-induced production of nitric oxide by cultured smooth muscle cells; effects on cell proliferation. Circ Res 71: 1088–1100
Sessa WC, Harrison JK, Barber CM, Zeng D, Durieux ME, D’ Angelo DD, Lynch KE, Peach MJ (1992) Molecular cloning and expression of cDNA encoding endothelial cell nitric oxide synthase. J Biol Chem 267: 1 52 74–1 52 76
Sessa WC, Barber CM, Lynch KR (1993) Mutation of N-myristoylation site converts endothelial cell nitric oxide synthase from a membrane to a cytosolic protein. Circ Res 72: 921–924
Sessa WC, Pritchard K, Seyedi N, Wang J, Hintze TH (1994) Chronic exercise in dogs increases coronary vascular nitric oxide production and endothelial cell nitric oxide synthase gene expression. Circ Res 74: 249–353
Stamler JS, Jaraki O, Osborne J, Simon Dl, Keaney J, Vita J, Singel D, Valeri CR, Loscalzo J (1992a) Nitric oxide circulates in mammalian plasma primarily as an S-nitroso adduct of serum albumin. Proc Natl Acad Sci USA 89: 7674–7677
Stamler JS, Simon Dl, Osborne JA, Mullins ME, Jaraki O, Michel T, Singel DJ, Loscalzo J (1992b) Snitrosylation of proteins with nitric oxide—synthesis and characterization of biologically active compounds. Proc Natl Acad Sci USA 89: 444–448
Thiemermann C, Vane JR (1990) Inhibition of nitric oxide synthesis reduces the hypotension induced by bacterial lipopolysaccharides in the rat in vivo. Eur J Pharmacol 182: 591–595
Valiance P, Collier J, Moncada S (1989) Effects of endothelium-derived nitric oxide on peripheral arteriolar tone in man. Lancet 2: 997–1000
Werner ER, Werner-Felmayer G, Fuchs D, Hausen A, Reibnegger G, Wachter H (1989) Parallel induction of tetrahydrobiopterin biosynthesis and indoleamine 2,3-dioxygenase activity in human cells and cell lines by interferon-gamma. Biochem J 262: 861–868
Xie Qw, Whisnant R, Nathan C (1993) Promotor of the mouse gene encoding calcium-dependent nitric oxide synthase confers inducibility by interferon gamma and bacterial lipopolysaccharide. J Exp Med 177: 1779–1784
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1995 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Busse, R., Fleming, I., Schini, V.B. (1995). Nitric Oxide Formation in the Vascular Wall: Regulation and Functional Implications. In: Koprowski, H., Maeda, H. (eds) The Role of Nitric Oxide in Physiology and Pathophysiology. Current Topics in Microbiology and Immunology, vol 196. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-79130-7_3
Download citation
DOI: https://doi.org/10.1007/978-3-642-79130-7_3
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-79132-1
Online ISBN: 978-3-642-79130-7
eBook Packages: Springer Book Archive