Abstract
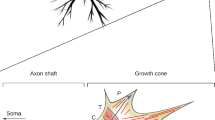
Neurite outgrowth is one of the early cellular events leading to the differentiation of the neuronal cell. The timing and rate of neurite outgrowth is of crucial importance since the first synaptical contact established appears to have certain advantages in the process of selection during the maturation of the nervous system (Changeux and Danchin, 1976). The rate of neurite outgrowth determines the success or failure in the regeneration phenomena. Indeed, neurons of the central nervous system have the ability to regenerate neurites if provided a proper environment (Benfey and Aguayo, 1982). Therefore the identification of the biochemical events which increase the rate of neurite outgrowth could as well increase our chances to influence the regeneration of functional connections.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Baron-van Evercooren A, Kleinman HK, Ohno S, Marangos P, Schwartz JP, Dubois-Dalcq ME (1982) Nerve growth factor, laminin and fibronectin promote neurite growth in human fetal sensory ganglia cultures. J. Neurosci. Res. 8: 179–193
Benfey M, Aguayo AJ (1982) Extensive elongation of axons from rat brain into peripheral nerve grafts. Nature 296: 150–152
Bray D, Chapman K (1985) Analysis of microspike movements on the neuronal growth cone. J. Neuroscience 5: 3204–3213
Changeux JP, Danchin A (1976) Selective stabilization in developing synapses as a mechanism for the specification of neuronal networks. Nature 264: 705–712
Edgar D, Timpl R, Thoenen H (1984) The heparin-binding domain of laminin is responsible for its effects on neurite outgrowth and neuronal survival. EMBO J. 3: 1463–1468
Ferrari G, Greene LA (1985) Does phospholipid methylation play a role in the primary mechanism of action of nerve growth factor. J. Neurochem. 45: 853–859
Ferrari G, Fabris M, Gorio A (1983) Gangliosides enhance neurite outgrowth in PC-12 cells. Dev.Brain Res. 8: 215–221
Gloor S, Odink K, Guenther J, Nick H, Monard D (1986) A glia- derived neurite promoting factor with protease inhibitory activity belongs to the protease nexins. Cell 47: 687–693
Gorio A, Carmignoto G, Facci L, Finesso M (1980) Motor nerve sprouting induced by ganglioside treatment. Possible implications for gangliosides on neuronal growth. Brain Res. 197: 236–241
Guenther J, Nick H, Monard D (1985) A glia-derived neurite- promoting factor with protease inhibitory activity. EMBO J. 4: 1963–1966
Ignatius MJ, Gebicke-Haerter PJ, Skene JHP, Schilling JW, Weisgraber KH, Mahley RW, Shooter EM (1986) Expression of apolipoprotein E during nerve degeneration and regeneration. Proc. Natl. Acad. Sci. USA 83: 1125–1129
Ignatius MJ, Shooter EM, Pitas RE, Mahley RW (1987) Lipoprotein uptake by neuronal growth cones in vitro. Science 236: 959–962
Krystosek A, Seeds NW (1981) Plasminogen activator release at the neuronal growth cone. Science 213: 1532–1534
Maltese WA, Reitz BA, Volpe JJ (1981) Effects of prior sterol depletion on neurite outgrowth in neuroblastoma cells. J. Cell. Physiol. 108: 475–482
Monard D (1985) Implications of proteases and protease inhibitors in neurite outgrowth. In Neurobiochemistry, Selected Topics, Colloquium-Mosbach 1985 Hamprecht B and Neuhoff V eds 36: 7–12
Monard D, Rentsch M, Schuerch-Rathgeb Y, Lindsay R (1977) Morphological differentiation of neuroblastoma cells in medium supplemented with delipidated serum. Proc. Natl. Acad. Sci. (USA) 74: 3893–3897
Monard D, Niday E, Limat A, Solomon F (1983) Inhibition of protease activity can lead to neurite extension in neuroblastoma cells. Prog. Brain Res. 58: 359–364
Murato K, Monard D (1982) Inhibition of S-adenosylmethionine- linked methylation can lead to neurite extension in neuroblastoma cells. FEBS Letters 144: 321–325
Nakajima J, Tsuji S, Nagai Y (1986) Bioactive gangliosides: analysis of functional structures of the tetrasialoganglioside Go1b which promotes neurite outgrowth. Biochim. biophys. Acta 876: 65–71
Pittman RN (1985) Release of plasminogen activator and a calcium- dependent metalloprotease from cultured sympathetic and sensory neurons. Dev. Biol. 110: 91–101
Quigley JP, Gold LI, Schwimmer R, Sullivan LM (1987) Limited cleavage of cellular fibronectin by plasminogen activator purified from transformed cells. Proc. Natl. Acad. Sci. USA 84: 2776–2780
Roisen FJ, Bartfeld H, Nagele R, Yorke G (1981) Ganglioside stimulation of axonal sprouting in vitro. Science 214: 577–578
Scott RW, Bergman BL, Bajpai A, Hersh RT, Rodriguez H, Jones BN, Barreda C, Watts S, Baker JB (1985) Protease nexin. J. Biol. Chem. 260: 7029–7034
Skaper SD, Katoh-Semba R, Varon S (1985) GMl ganglioside accelerates neurite outgrowth from primary peripheral and central neurons under selected culture conditions. Dev. Brain Res. 23: 19–26
Snipes GJ, McGuire CB, Norden JJ, Freeman JA (1986) Nerve injury stimulates the secretion of apolipoprotein E by nonneuronal cells. Proc. Natl. Acad. Sci. USA 83: 1130–1134
Sommer J, Gloor SM, Rovelli GF, Hofsteenge J, Nick H, Meier R, Monard D (in press) cDNA sequence coding for a rat glia-derived nexin and its homology to members of the serpin superfamily. Biochemistry
Stone S, Nick H, Hofsteenge J, Monard D (1987) Glia-derived neurite promoting factor is a slow-binding inhibitor of trypsin, thrombin and urokinase Arch. Biochem. Biophys. 252: 237–244
Traynor AE (1984) The relationship between neurite extension and phospholipid metabolism in PC12 cells. Dev. Brain Res. 14: 205–210
Zurn A, Nick H, Monard D (submitted) A glia-derived nexin promotes neurite outgrowth in cultured chick sympathetic neurons.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1988 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Monard, D. (1988). Rate Limiting Events in Neurite Outgrowth. In: Gorio, A., Perez-Polo, J.R., de Vellis, J., Haber, B. (eds) Neural Development and Regeneration. NATO ASI Series, vol 22. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-73148-8_11
Download citation
DOI: https://doi.org/10.1007/978-3-642-73148-8_11
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-73150-1
Online ISBN: 978-3-642-73148-8
eBook Packages: Springer Book Archive