Abstract
The pathways of oxidative metabolism of the aromatic A-ring of steroidal estrogens such as estradiol involve initial hydroxylation reactions at C-2 and C-4. Estrogen 2-/4-hydroxylase activities, which catalyze the formation of the resulting catechol estrogen products, are found in microsomes from a variety of mammalian tissues. The relatively high rate of catechol estrogen formation in the liver has generally been regarded as a deactivating reaction because the catechol products were considered to be biologically inert. Recently, however, there has been a resurgence of interest in 2- and 4-hydroxy-estradiol due to their potential biological activities when they are formed within tissues, since these catechols are further metabolized extremely rapidly when they are administered to animals or humans. It has also become evident from differences between some of the biological activities of 2- and 4-hydroxy-estrogens that it is no longer appropriate to consider catechol estrogens as a single class of compounds.
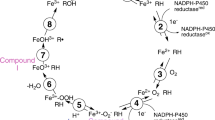
There is relatively little information on the mechanism of microsomal estrogen 244-hydroxylase activities. In this paper it is proposed that the active intermediates are stabilized tautomers of dienol epoxides, related in structure to some of the intermediates known to be involved in the conversion of carcinogenic polycyclic aromatic hydrocarbons into their reactive ultimate carcinogens, the dihydrodiol epoxides. The chemistry of estrogen-derived dienol epoxides will be discussed and syntheses described. Biochemical results will be provided which implicate one of these dienol epoxide taumomers as an intermediate in the conversion of estradiol to 2-hydroxyestradiol.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Fishman, J. (1983): Aromatic hydroxylation of estrogens. Ann. Rev. Physiol., 45, 61–72.
MacLusky, N.J., Naftolin, F., Krey, L.C. and Franks, S. (1981): The catechol estrogens. J. Steroid Biochem., 15, 111–124.
Merriam, G.R., MacLusky, N.J., Picard, M.K. and Naftolin, F. (1980): Comparative properties of the catechol estrogens, I: Methylation “by catechol-0-methyl-transferase and binding to cytosol estrogen receptors. Steroids, 36, 1–11.
Poth, M.A., Axelrod, J. and Hoffman, A.R. (1983): Estrogen-2-hydroxylase: developmental, hormonal, and pharmacological studies using a radioenzymatic method. In: Catechol Estrogens, edited by G.R. Merriam and M.B. Lipsett, pp. 19–30, Raven Press, New York.
Brown, H.S. Jeffrey, A.M. and Weinstein, I.B. (1979): Formation of DNA adducts in 10T1/2 mouse embryo fibroblasts incubated with benzo[a]pyrene or dihydrodiol oxide derivatives. Cancer Res., 39, l673-l677.
Tomatis, L., Agthe, C., Bartsch, H., Huff, J., Montesano, R., Saracci, R., Walker, E. and Wilbourn, J. (1978): Evaluation of the carcinogenicity of chemicals: A review of the monograph program of the International Agency for Research on Cancer. (1971 to 1977). Cancer Res., 38, 877–885.
Jull, J.W. (1976): Endocrine aspects of carinogenesis. ACS Monogr., 173, 52–82.
Lipsett, M.B. (1979): Interaction of drugs, hormones, and nutrition in the causes of cancer. Cancer, 43, 1967–1981.
Lipsett, M.B. (1983): Hormones, medications, and Cancer. Cancer, 51, 2426–2429
McCann, J. and Ames, B.N. (1976): Detection of carcinogens as mutagens in the Salmonella/microsome test: Assay of 300 chemicals: Discussion. Proc. Natl. Acad. Sci. U.S.A., 73, 950–954.
Glatt, H.R., Metzler, M., and Oesch, F. (1979): Diethylstilbestrol and 11 derivatives. A mutagenicity study with Salmonella typhimurium. Mutat. Res. 67, 113–121.
Mehta, R.D. and von Borstel, R.C. (1982): Genetic activity of diethylstilbestrol in Saccharomyces cerevisiae. Enhancement of mutagenicity by oxidizing agents. Mutat. Res., 92, 49–6l.
Creus, A., Xamena, N. and Marcos, R. (1983): Positive response of diethylstilbestrol in the sex-linked recessive lethal assay in Drosophila after larval feeding. Mutat. Res. 122, 309–313.
Clive, D., Johnson, K.O., Spector, J.F.S., Batson, A.G. and Brown M.M.M. (1979): Validation and characterization of the L5178Y/TK mouse lymphoma mutagen assay system. Mutat. Res., 59, 61-108.
Barrett, J.C., Wong, A. and McLachlan, J.A, (1981): Diethylstilbestrol induces neoplastic transformation without measurable gene mutation of two loci. Science, 212, 1402–1404.
Purdy, R.H., Goldzieher, J.W., Le Quesne, P.W., Abdel-Baky, S., Durocher, C.K., Moore, P.H., Jr. and Rhim, J.S. (1983): Active intermediates and carcinogenesis. In: Catechol Estrogens, edited by G.R. Merriam and M.B. Lipsett, pp. 123–140, Raven Press, New York.
Kakunaga, T., Lo, K.-Y., Leavitt, J. Ikenaga, M. (1980): Relationship between transformation and mutation in mammalian cells. Jerusalem Symp. Quantum Chem. Biochem., 13, 527–541.
Holder, G., Yagi, H., Dansett, P., Jerina, D.M., Levin, W., Lu, A.Y.H. and Conney, A.H. (1974): Effects of inducers and epoxide hydrase on the metabolism of benzo-[a]pyrene by liver microsomes and a reconstituted system: Analysis by high pressure liquid chromatography. Proc. Natl. Acad. Sci. U.S.A., 71, 4356–4360.
Soloway, A.H. and Le Quesne, P.W. (1980): Potential endogenous mutagens/carcinogens. J. Theoret. Biol., 85, 153–163.
Le Quesne, P.W., Durga, A.V., Subramanyam, V., Soloway, A.H., Hart, R.W. and Purdy, R.H. (1980): Biomimetic synthesis of catechol estrogens: Potentially mutagenic arene oxide intermediates in estrogen metabolism. J. Med. Chem., 23, 239–240.
Abdel-Baky, S. (1983): Ph.D. Thesis, Department of Chemistry, Northeastern Univesity, Boston, MA.
Paul, S.M., Hoffman, A.R., and Axelrod, J. (1980): Catechol estrogens: Synthesis and metabolism in brain and other endocrine tissues. Front. Neuroendocriniol., 6, 203–217.
Paul, S.M., Purdy, R.H., Hoffman, A.R. and Axelrod, J. (1983): Radioenz ymatic assays for catechol estrogens. In: Catechol Estrogens, edited “by G.R. Merriam and M.B. Lipsett, pp. 83–90, Raven Press, New York.
Kaufman, S. (1961): The enzymic conversion of 4-fluorophenylalanine to tyrosine. Biochim. Biophys. Acta, 51, 619–621.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1986 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Le Quesne, P.W., Purdy, R.H. (1986). The Oxidative Metabolism of the Aromatic A-Ring of Steroidal Estrogens. In: Rahman, Au. (eds) Natural Product Chemistry. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-71425-2_12
Download citation
DOI: https://doi.org/10.1007/978-3-642-71425-2_12
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-71427-6
Online ISBN: 978-3-642-71425-2
eBook Packages: Springer Book Archive