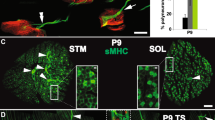
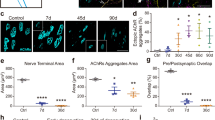
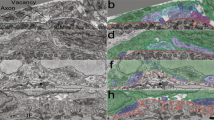
Abstract
A large amount of information has been accumulating over the last 30 years indicating that the adult motoneuron is capable of producing extensive peripheral sprouting under a variety of conditions. Sprouts can reoccupy abandoned synaptic sites, form new ones, or might remain or be withdrawn without forming synaptic contacts. Such abnormal outgrowth of branches, which was first described after partial denervation of a muscle (van Harreveld 1945; Weiss and Edds 1945; Edds 1950; Hoffman 1950), includes collateral sprouts which originate from the nodes of Ranvier, preterminal sprouts and sprouts from the endplate itself (‘ultraterminal’ sprouts). Consequently, when searching for sprouts most investigators have been looking for nerve branches which appear “abnormal” in the sense that they leave the vicinity of the parent endplate and do not regularly occur under normal conditions (see Sect. 2.1). This is demonstrated in Fig. 1, which shows a typical endplate from a normal mouse muscle (Fig. 1A) and an endplate after local application of tetanus toxin (Fig. 1B); in the latter unusual branches are present (arrows) which leave the endplate area, often to reach neighboring muscle fibers (cf. Duchen and Tonge 1973).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Barker D, Ip MC (1966) Sprouting and degeneration of mammalian motor axons in normal and deafferentated skeletal muscle. Proc R Soc London Ser B 163: 538–554
Bixby JL, van Essen DC (1979) Competition between foreign and original nerves in adult mammalian skeletal muscle. Nature (London) 282: 726–728
Blinzinger K, Kreutzberg G (1968) Displacement of synaptic terminals from regenerating moto-neurons by microglial cells. Z Zellforsch 85: 145–157
Brown MC, Holland RL (1979) A central role for denervated tissues in causing nerve sprouting. Nature (London) 282: 724–726
Brown MC, Ironton R (1977) Motor neuron sprouting induced by prolonged tetrodotoxin block of nerve action protentials. Nature (London) 265: 549–561
Brown MC, Ironton R (1978) Sprouting and regression of neuromuscular synapses in partially denervated mammalian muscles. J Physiol 278: 325–348
Brown MC, Holland RL, Ironton R (1978) Degnerating nerve products affect innervated muscle fibres. Nature (London) 275: 652–654
Burden SJ, Sargent PB, McMahan UJ (1979) Acetylcholine receptors in regenerating muscle ac- cumulate at original synaptic sites in the absence of the nerve. J Cell Biol 82: 412–425
Cangiano A, Lutzemberger L, Nicotra L (1977) Non-equivalence of impulse blockade and denerva- tion in the production of membrane changes in rat skeletal muscle. J Physiol 273: 325–348
Cangiano A, Lutzemberger L, Nicotra L (1977) Non-equivalence of impulse blockade and denerva- tion in the production of membrane changes in rat skeletal muscle. J Physiol 273: 325–348
Diamond J, Cooper E, Turner C, Macintyre L (1976) Trophic regulation of nerve sprouting. Science 193: 371–377
Dimpfel W, Habermann E (1973) Histoautoradiographic localisation of 125I-labelled tetanus toxin in rat spinal cord. Naunyn-Schmiedeberg’s Arch Pharmacol 280: 177–182
Duchen LW (1971) An electron microscopic study of the changes induced by botulinum toxin in the motor end-plates of slow and fast skeletal muscle fibres of the mouse. J Neurol Sci 14: 4760
Duchen LW (1973) The effects of tetanus toxin on the motor end-plates of the mouse — an electron microscopic study. J Neurol Sci 19: 153–167
Duchen LW, Strich SJ (1968) The ffects of botulinum toxin on the pattern of innervation of skeletal muscle in the mouse. Q J Exp Physiol 53: 84–89
Duchen LW, Tonge DA (1973) The effects of tetanus toxin of neuromuscular transmission and on the morphology of motor endplates in slow and fast skeletal muscle of the mouse. J Physiol 228: 157–172
Edds MV (1950) Collateral regeneration of residual motor axons in partially denervated muscles. J Exp Zool 113: 517–552
Edds MV (1953) Collateral nerve regeneration. Q Rev Biol 28: 260–276
Frank E, Jansen JKS, Lömo T, Westgaard RH (1975) The interaction between foreign and original motor nerves innervating the soleus muscle of rats..1 Physiol 247: 725–743
Goldowitz D, Cotman CW (1980) Do neurotrophic interactions control synapse formation in the adult rat brain. Brain Res 181: 325–344
Guth L (1968) “Trophic” influences of nerve on muscle. Physiol Rev 48: 645–687
Haimann C, Mallart A, Zilber-Gachelin NF (1976) Competition between motor nerves in the estab-lishment of neuromuscular junctions in striated muscles of Xenopus laevis. Neurosci Lett 2: 15–20
Hansson HA, Norström A (1971) Glial reactions induced by colchicine-treatment of the hypothalamic-neurohypophyseal system. Z Zellforsch 113: 294–310
Hoffman H (1950) Local re-innervation in partially denervated muscle: a histophysiological study. Aust J Exp Biol Med Sci 28: 383–397
Holland RL, Brown MC (1980) Postsynaptic transmission block can cause terminal sprouting of a motor nerve. Science 207: 649–651
Huizar P, Kuno M, Kudo N, Miyata Y (1977) Reaction of intact spinal motoneurones to partial denervation of the muscle. J Physiol 265: 175–191
Ip MC (1974) Some morphological features of the myoneural junction in certain normal muscles of the rat. Anat Rec 180: 605–616
Karnovsky MJ, Roots L (1964) A “direct-coloring” thiocholine method for cholinesterases. J Histochem Cytochem 12: 219–221
Koenig J (1973) Morphogenesis of motor end-plates “in vivo” and “in vitro”. Brain Res 62: 361–435
Kuffler D, Thompson W, Jansen JKS (1977) The elimination of synapses in multiply-innervated skeletal muscle fibres of the rat: dependence on distance between end-plates. Brain Res 138: 353–358
Letinsky MS, Fischbeck KH, McMahan UJ (1976) Precision of reinnervation of original postsynaptic sites in frog muscle after a nerve crush. J Neurocytol 5: 691–718
Lömo T (1980) The role of impulse activity in the formation of neuromuscular junctions. In: Taxi J (ed) Ontogenesis and functional mechanisms of peripheral synapses. Elsevier, Amsterdam pp 327–334
Lömo T, Slater CR (1978) Control of acetylcholine sensitivity and synapse formation by muscle activity. J Physiol 275: 391–402
Lömo T, Slater CR (1980a) Control of junctional acetylcholinesterase by neural and muscular influences in the rat. J Physiol 303: 191–202
Lömo T, Slater CR (1980b) Acetylcholine sensitivity of developing ectopic nerve-muscle junctions in adult rat soleus muscles. J Physiol 303: 173–189
Nyström B (1968) Postnatal development of motor nerve terminals in “slow-red” and “fast-white” cat muscles. Acta Neurol Scand 44: 363–383
Pecot-Dechavassine M, Wernig A, Stöver H (1979) A combined silver and cholinesterase method for studying exact relations between the pre-and the postsynaptic elements at the frog neuromuscular junction. Stain Technol 54: 25–28
Pestronk A, Drachman DB (1978) Motor nerve sprouting and acetylcholine receptors. Science 199: 1223–1225
Purves D, Lichtman JW (1978) Formation and maintenance of synaptic connections in autonomic ganglia. Physiol Rev 58: 821–862
Rotshenker S (1979) Synapse formation in intact innervated cutaneous pectoris muscles of the frog following denervation of the opposite muscle. J Physiol 292: 535–547
Sanes JR, Marshall LM, McMahan UJ (1978) Reinnervation of muscle fiber basal lamina after removal of myofibers. J Cell Biol 78: 176–198
Schubert P, Kreutzberg GW, Lux HD (1972) Neuroplasmic transport in dendrites: effect of colchi- cine on morphology and physiology of motoneurones in the cat. Brain Res 47: 331–343
Schwartz MS, Sargent MK, Swash M (1977) Neostigmin-induced end-plate proliferation in the rat. Neurology 27: 289–293
Sumner BEH (1977) Ultrastructural responses of the hypoglossal nucleus to the presence in the tongue of botulinum toxin, a quantitative study. Exp Brain Res 30: 313–321
Teräväinen H (1968) Development of the myoneural junction in the rat. Z Zellforsch 87: 249–265
Tonge DA (1977) Effect of implantation of an extra nerve on the recovery of neuromuscular transmission from botulinum toxin. J Physiol 265: 809–820
Tonge DA (1978) Prolonged effects of a post-synaptic blocking fraction of “naja siamensis” venom on skeletal muscle of the mouse. Q J Exp Physiol 63: 33–47
Tuffery RA (1971) Growth and degeneration of motor end-plates in normal cat hind limb muscles. J Anat 110: 221–247
Tweedle CD, Kabara JJ (1977) Lipophilic nerve sprouting factor(s) isolated form denervated muscle. Neurosci Lett 6: 41–46
Van Harreveld D (1945) Reinnervation of denervated muscle fibres by adjacent functioning motor units. Am J Physiol 144: 477–493
Watson WE (1968) Observations on the nucleolar and total cell body nucleic acid of injured nerve cells. J Physiol 196: 655–676
Watson WE (1969) The response of motor neurones to intramuscular injection of botulinum toxin. J Physiol 202: 611–630
Watson WE (1972) Some quantitative observations upon the responses of neuroglial cells which follow axotomy of adjacent neurones. J Physiol 225: 415–435
Weiss P, Edds MV (1945) Spontaneous recovery of muscle following partial denervation. Am J Physiol 145: 587–607
Wernig A, Pecot-Dechavassine M, Stöver H (1980a) Sprouting and regression of the nerve at the frog neuromuscular junction in normal conditions and after prolonged paralysis with curare. J Neurocytol 9: 277–303
Wernig A, Pecot-Dechavassine M, Stöver H (1980b) Signs of nerve regression and sprouting in the frog neuromuscular synapse. In: Taxi J (ed) Ontogenesis and functional mechanisms of peripheral synapses. Elsevier, Amsterdam, pp 225–238
Wernig A, Anzil AP, Bieser A (1981a) Light and electron microscopic identification of a nerve sprout in muscle of normal adult frog. Neurosci Lett 21: 261–266
Wernig A, Anzil AP, Bieser A (1981a) Light and electron microscopic identification of a nerve sprout in muscle of normal adult frog. Neurosci Lett 21: 261–266
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1981 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Wernig, A., Anzil, A.P., Bieser, A. (1981). Formation and Regression of Synaptic Contacts in the Adult Muscle. In: Flohr, H., Precht, W. (eds) Lesion-Induced Neuronal Plasticity in Sensorimotor Systems. Proceedings in Life Sciences. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-68074-8_4
Download citation
DOI: https://doi.org/10.1007/978-3-642-68074-8_4
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-68076-2
Online ISBN: 978-3-642-68074-8
eBook Packages: Springer Book Archive