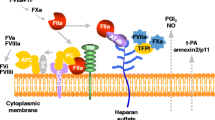
Sepsis is a clinical syndrome which symptoms are caused by a systemic host response to infection. Amongst the most prominent features of sepsis are inflammation-induced activation of coagulation with concurrent downregulation of anticoagulant systems and fibrinolysis, in its most severe form leading to disseminated intravascular coagulation, which contributes to multiple organ failure and mortality. Another important hallmark in sepsis is endothelial dysfunction. Under normal conditions the endothelium provides for an anticoagulant surface, a property which is lost in sepsis. Activation of coagulation in turn impacts on inflammation. In this chapter data on the interplay between inflammation and coagulation in sepsis are summarized with a special focus on the role of the endothelium. Tissue factor-induced activation of coagulation is discussed, together with the procoagulant role of microparticles, platelets and von Willebrand factor. Downregulation of the anticoagulants tissue factor pathway inhibitor, antithrombin and the protein C system is discussed, together with the roles of glycosaminoglycans, proteoglycans and heparin. Moreover, inhibition of fibrinolysis in sepsis is described. The influence of coagulation and fibrinolysis in turn on inflammation is discussed with a special focus on protease-activated receptors, a research area that could substantially contribute to our understanding of the interplay between coagulation and inflammation in sepsis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Cohen J (2002) The immunopathogenesis of sepsis. Nature 420:885–891
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, and Pinsky MR (2001) Epidemiology of severe sepsis in the United States: Analysis of incidence, outcome, and associated costs of care. Crit Care Med 29:1303–1310
Opal SM and Esmon CT (2003) Bench-to-bedside review: functional relationships between coagulation and the innate immune response and their respective roles in the pathogenesis of sepsis. Crit Care 7:23–38
Levi M and Ten Cate H (1999) Disseminated intravascular coagulation. N Engl J Med 341: 586–592
Dhainaut JF, Yan SB, Joyce DE, Pettila V, Basson B, Brandt JT, Sundin DP, and Levi M (2004) Treatment effects of drotrecogin alfa (activated) in patients with severe sepsis with or without overt disseminated intravascular coagulation. J Thromb Haemost 2:1924–1933
Aird WC (2003) The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome. Blood 101:3765–3777
Henneke P and Golenbock D T (2002) Innate immune recognition of lipopolysaccharide by endothelial cells. Crit Care Med 30:S207–S213
Zhang FX, Kirschning CJ, Mancinelli R, Xu XP, Jin YP, Faure E, Mantovani A, Rothe M, Muzio M, and Arditi M (1999) Bacterial lipopolysaccharide activates nuclear factor-kappa B through interleukin-1 signaling mediators in cultured human dermal endothelial cells and mononuclear phagocytes. J Biol Chem 274:7611–7614
Bombeli T, Müller M, and Häberli A (1997) Anticoagulant properties of the vascular endothe-lium. Thromb Haemost 77:408–423
Levi M, Van der Poll T, and Büller H R (2004) Bidirectional relation between infl ammation and coagulation. Circulation 109:2698–2704
Camerer E, Kolsto A B, and Prydz H (1996) Cell biology of tissue factor, the principal initiator of blood coagulation. Thromb Res 81:1–41
Osterud B and Flaegstad T (1983) Increased tissue thromboplastin activity in monocytes of patients with meningococcal infection: related to an unfavourable prognosis. Thromb Haemost 49:5–7
Osterud B (1998) Tissue factor expression by monocytes: regulation and pathophysiological roles. Blood Coagulat Fibrinol 9:S9–S14
Monroe DM and Key NS (2007) The tissue factor-factor VIIa complex: procoagulant activity, regulation, and multitasking. J Thromb Haemost 5:1097–1105
Bogdanov V, Pyo R T, Taubman M B, and Nemerson Y (2002) Identifi cation of alternatively spliced murine tissue factor mRNA and protein. Circulation 106:39–39
Szotowski B, Antoniak S, Poller W, Schultheiss H P, and Rauch U (2005) Procoagulant soluble tissue factor is released from endothelial cells in response to infl ammatory cytokines. Circulation Res 96:1233–1239
Bevilacqua MP, Pober JS, Majeau GR, Fiers W, Cotran RS, and Gimbrone MA, Jr. (1986) Recombinant tumor necrosis factor induces procoagulant activity in cultured human vascular endothelium: characterization and comparison with the actions of interleukin 1. Proc Natl Acad Sci U S A 83:4533–4537
Lupu C, Westmuckett AD, Peer G, Ivanciu L, Zhu H, Taylor FB, and Lupu F (2005) Tissue factor-dependent coagulation is preferentially up-regulated within arterial branching areas in a baboon model of Escherichia coli sepsis. Am J Pathol 167:1161–1172
Erlich J, Fearns C, Mathison J, Ulevitch R J, and Mackman N (1999) Lipopolysaccharide induction of tissue factor expression in rabbits. Infect Immun 67:2540–2546
De Jonge E, Dekkers PEP, Creasey AA, Hack CE, Paulson SK, Karim A, Kesecioglu J, Levi M, Van Deventer SJH, and Van der Poll T (2000) Tissue factor pathway inhibitor dose-dependently inhibits coagulation activation without infl uencing the fi brinolytic and cytokine response during human endotoxemia. Blood 95:1124–1129
De Pont ACJM, Moons AHM, de Jonge E, Meijers JCM, Vlasuk GP, Rote WE, Büller HR, Van der Poll T, and Levi M (2004) Recombinant nematode anticoagulant protein c2, an inhibitor of tissue factor/factor VIIa, attenuates coagulation and the interleukin-10 response in human endotoxemia. J Thromb Haemost 2:65–70
Levi M, Ten Cate H, Bauer KA, Van der Poll T, Edgington TS, Büller HR, Van Deventer SJH, Hack CE, Ten Cate JW, and Rosenberg RD (1994) Inhibition of endotoxin-induced activation of coagulation and fi brinolysis by pentoxifylline or by a monoclonal antitissue factor antibody in chimpanzees. J Clin Investig 93:114–120
Taylor FB, Chang A, Ruf W, Morrissey JH, Hinshaw L, Catlett R, Blick K, and Edgington TS (1991) Lethal Escherichia coli septic shock is prevented by blocking tissue factor with monoclonal-antibody. Circulatory Shock 33:127–134
Berckmans RJ, Nieuwland R, Boing AN, Romijn FPHT, Hack CE, and Sturk A (2001) Cell-derived microparticles circulate in healthy humans and support low grade thrombin generation. Thromb Haemost 85:639–646
Aras O, Shet A, Bach RR, Hysjulien JL, Slungaard A, Hebbel RP, Escolar G, Jilma B, and Key NS (2004) Induction of microparticle- and cell-associated intravascular tissue factor in human endotoxemia. Blood 103:4545–4553
Nieuwland R, Berckmans RJ, McGregor S, Boing AN, Romijn FPHT, Westendorp RGJ, Hack CE, and Sturk A (2000) Cellular origin and procoagulant properties of microparticles in meningococcal sepsis. Blood 95:930–935
Satta N, Toti F, Feugeas O, Bohbot A, Dacharyprigent J, Eschwege V, Hedman H, and Freyssinet JM (1994) Monocyte vesiculation is a possible mechanism for dissemination of membrane-associated procoagulant activities and adhesion molecules After stimulation by lipopolysaccharide. J Immunol 153:3245–3255
Del Conde I, Shrimpton CN, Thiagarajan P, and Lopez JA (2005) Tissue-factor-bearing microvesicles arise from lipid rafts and fuse with activated platelets to initiate coagulation. Blood 106:1604–1611
Combes V, Simon AC, Grau GE, Arnoux D, Camoin L, Sabatier F, Mutin M, Sanmarco M, Sampol J, and Dignat-George F (1999) In vitro generation of endothelial microparticles and possible prothrombotic activity in patients with lupus anticoagulant. J Clin Invest 104: 93–102
Jy W, Jimenez JJ, Cheng PY, Mauro LM, Horstman LL, Ahn ER, Bidot CJ, and Ahn YS (2004) Endothelial microparticles (EMP) bind and complex with unusually-large multimers of von Willebrand factor (ULvWf) and act as potent inducers of platelet adhesion and aggregation. Blood 104:997A–998A
Dignat-George F, Camoin-Jau L, Sabatier F, Arnoux D, Anfosso F, Bardin N, Veit V, Combes V, Gentile S, Moal V, Sanmarco M, and Sampol J (2004) Endothelial microparticles: a potential contribution to the thrombotic complications of the antiphospholipid syndrome. Thromb Haemost 91:667–673
Akca S, Haji-Michael P, De Mendonca A, Suter P, Levi M, and Vincent JL (2002) Time course of platelet counts in critically ill patients. Crit Care Med 30:753–756
Zielinski T, Wachowicz B, Saluk-Juszczak J, and Kaca W (2002) Polysaccharide part of Proteus mirabilis lipopolysaccharide may be responsible for the stimulation of platelet adhesion to collagen. Platelets 13:419–424
Zimmerman GA, McIntyre TM, Prescott SM, and Stafforini DM (2002) The platelet-activating factor signaling system and its regulators in syndromes of infl ammation and thrombosis. Crit Care Med 30:S294–S301
Reinhart K, Bayer O, Brunkhorst F, and Meisner M (2002) Markers of endothelial damage in organ dysfunction and sepsis. Crit Care Med 30:S302–S312
Bernardo A, Ball C, Nolasco L, Moake J F, and Dong J F (2004) Effects of infl ammatory cytokines on the release and cleavage of the endothelial cell-derived ultralarge von Willebrand factor multimers under fl ow. Blood 104:100–106
Martin K, Borgel D, Lerolle N, Feys H B, Trinquart L, Vanhoorelbeke K, Deckmyn H, Legendre P, Diehl J L, and Baruch D (2007) Decreased ADAMTS-13 (A disintegrin-like and metalloprotease with thrombospondin type 1 repeats) is associated with a poor prognosis in sepsis-induced organ failure. Crit Care Med 35:2375–2382
Levi M and Van der Poll T (2005) Two-way interactions between infl ammation and coagulation. Trends Cardiovasc Med 15:254–259
Ott I, Miyagi Y, Miyazaki K, Heeb MJ, Müller BM, Rao LVM, and Ruf W (2000) Reversible regulation of tissue factor-induced coagulation by glycosylphosphatidylinositol-anchored tissue factor pathway inhibitor. Arterioscler Thromb Vasc Biol 20:874–882
Brandtzaeg P, Sandset PM, Joo GB, Ovstebo R, Abildgaard U, and Kierulf P (1989) The quantitative association of plasma endotoxin, antithrombin, protein C, extrinsic pathway inhibitor and fi brinopeptide A in systemic meningococcal disease. Thromb Res 55:459–470
Sandset PM, Warncramer BJ, Maki SL, and Rapaport SI (1991) Immunodepletion of extrinsic pathway inhibitor sensitizes rabbits to endotoxin-induced intravascular coagulation and the generalized Shwartzman reaction. Blood 78:1496–1502
Creasey AA, Chang ACK, Feigen L, Wun TC, Taylor FB, and Hinshaw LB (1993) Tissue factor pathway inhibitor reduces mortality from Escherichia Coli septic shock. J Clin Invest 91:2850–2860
Abraham E, Reinhart K, Opal S, Demeyer I, Doig C, Rodriguez AL, Beale R, Svoboda P, Laterre PF, Simon S, Light B, Spapen H, Stone J, Seibert A, Peckelsen C, De Deyne C, Postier R, Pettila V, Artigas A, Percell SR, Shu V, Zwingelstein C, Tobias J, Poole L, Stolzenbach JC, and Creasey AA (2003) Effi cacy and safety of tifacogin (recombinant tissue factor pathway inhibitor) in severe sepsis: a randomized controlled trial. JAMA 290:238–247
Dickneite G (1998) Preclinical evaluation of antithrombin III in experimental sepsis and DIC. Blood 92:357A
Minnema MC, Chang AC, Jansen PM, Lubbers YT, Pratt BM, Whittaker BG, Taylor FB, Hack CE, and Friedman B (2000) Recombinant human antithrombin III improves survival and attenuates infl ammatory responses in baboons lethally challenged with Escherichia coli. Blood 95:1117–1123
Taylor FB Jr, Emerson TE Jr, Jordan R, Chang AK, and Blick KE (1988) Antithrombin-III prevents the lethal effects of Escherichia coli infusion in baboons. Circ Shock 26:227–235
Leitner JM, Firbas C, Mayr FB, Reiter RA, Steinlechner B, and Jilma B (2006) Recombinant human antithrombin inhibits thrombin formation and interleukin 6 release in human endotoxemia. Clin Pharmacol Therapeut 79:23–34
Harada N, Okajima K, Kushimoto S, Isobe H, and Tanaka K (1999) Antithrombin reduces ischemia/reperfusion injury of rat liver by increasing the hepatic level of prostacyclin. Blood 93:157–164
Ostrovsky L, Woodman RC, Payne D, Teoh D, and Kubes P (1997) Antithrombin III prevents and rapidly reverses leukocyte recruitment in ischemia/reperfusion. Circulation 96:2302–2310
Horie S, Ishii H, and Kazama M (1990) Heparin-like glycosaminoglycan is a receptor for antithrombin III-dependent but not for thrombin-dependent prostacyclin production in human endothelial cells. Thromb Res 59:895–904
Warren BL, Eid A, Singer P, Pillay SS, Carl P, Novak I, Chalupa P, Atherstone A, Penzes I, Kubler A, Knaub S, Keinecke HO, Heinrichs H, Schindel F, Juers M, Bone RC, and Opal SM (2001) High-dose antithrombin III in severe sepsis — a randomized controlled trial. J Am Med Assoc 286:1869–1878
Gotte M (2003) Syndecans in infl ammation. FASEB J 17:575–591
Parish CR (2006) The role of heparan sulphate in infl ammation. Nat Rev Immunol 6:633–643
Xie X, Rivier AS, Zakrzewicz A, Bernimoulin M, Zeng XL, Wessel HP, Schapira M, and Spertini O (2000) Inhibition of selectin-mediated cell adhesion and prevention of acute infl am-mation by nonanticoagulant sulfated saccharides — studies with carboxyl-reduced and sulfated heparin and with trestatin A sulfate. J Biol Chem 275:34818–34825
Esmon CT (2003) The protein C pathway. Chest 124:26S–32S
Conway EM and Rosenberg RD (1988) Tumor necrosis factor suppresses transcription of the thrombomodulin gene in endothelial cells. Mol Cell Biol 8:5588–5592
Liaw PC, Esmon CT, Kahnamoui K, Schmidt S, Kahnamoui S, Ferrell G, Beaudin S, Julian JA, Weitz JI, Crowther M, Loeb M, and Cook D (2004) Patients with severe sepsis vary markedly in their ability to generate activated protein C. Blood 104:3958–3964
Takano S, Kimura S, Ohdama S, and Aoki N (1990) Plasma thrombomodulin in health and diseases. Blood 76:2024–2029
Faust SN, Levin M, Harrison OB, Goldin RD, Lockhart MS, Kondaveeti S, Laszik Z, Esmon CT, and Heyderman RS (2001) Dysfunction of endothelial protein C activation in severe meningococcal sepsis. N Engl J Med 345:408–416
Xu J, Qu D, Esmon NL, and Esmon CT (2000) Metalloproteolytic release of endothelial cell protein C receptor. J Biol Chem 275:6038–6044
Taylor FB Jr, Chang A, Esmon CT, D'Angelo A, Vigano-D'Angelo S, and Blick KE (1987) Protein C prevents the coagulopathic and lethal effects of Escherichia coli infusion in the baboon. J Clin Invest 79:918–925
Taylor FB Jr, Stearns-Kurosawa DJ, Kurosawa S, Ferrell G, Chang AC, Laszik Z, Kosanke S, Peer G, and Esmon CT (2000) The endothelial cell protein C receptor aids in host defense against Escherichia coli sepsis. Blood 95:1680–1686
Taylor FB Jr, Chang AC, Peer GT, Mather T, Blick K, Catlett R, Lockhart MS, and Esmon CT (1991) DEGR-factor Xa blocks disseminated intravascular coagulation initiated by Escherichia coli without preventing shock or organ damage. Blood 78:364–368
Bernard GR, Vincent JL, Laterre PF, LaRosa SP, Dhainaut JF, Lopez-Rodriguez A, Steingrub JS, Garber GE, Helterbrand JD, Ely EW, and Fisher CJ Jr. (2001) Effi cacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med 344:699–709
Van der Poll T, Levi M, Büller HR, Van Deventer SJ, De Boer JP, Hack CE, and Ten Cate JW (1991) Fibrinolytic response to tumor necrosis factor in healthy subjects. J Exp Med 174:729–732
Carmeliet P, Schoonjans L, Kieckens L, Ream B, Degen J, Bronson R, Devos R, Vandenoord JJ, Collen D, and Mulligan RC (1994) Physiological consequences of loss of plasminogen-activator gene-function in mice. Nature 368:419–424
Levi M, Dörffler-Melly J, Reitsma P, Büller H, Florquin S, Van der Poll T, and Carmeliet P (2003) Aggravation of endotoxin-induced disseminated intravascular coagulation and cytokine activation in heterozygous protein-C-defi cient mice. Blood 101:4823–4827
Esmon CT (2005) The interactions between infl ammation and coagulation. Br J Haematol 131:417–430
Van de Wouwer M, Collen D, and Conway EM (2004) Thrombomodulin-protein C-EPCR system: integrated to regulate coagulation and infl ammation. Arterioscler Thromb Vasc Biol 24:1374–1383
Myles T, Nishimura T, Yun T H, Nagashima M, Morser J, Patterson AJ, Pearl RG, and Leung LL (2003) Thrombin activatable fi brinolysis inhibitor, a potential regulator of vascular infl am-mation. J Biol Chem 278:51059–51067
Conway EM, Van de Wouwer M, Pollefeyt S, Jurk K, Van Aken H, De Vriese A, Weitz JI, Weiler H, Hellings PW, Schaeffer P, Herbert JM, Collen D, and Theilmeier G (2002) The lectin-like domain of thrombomodulin confers protection from neutrophil-mediated tissue damage by suppressing adhesion molecule expression via nuclear factor kappaB and mitogen-activated protein kinase pathways. J Exp Med 196:565–577
Blasi F (1999) Proteolysis, cell adhesion, chemotaxis, and invasiveness are regulated by the u-PA-u-PAR-PAI-1 system. Thromb Haemost 82:298–304
Rijneveld AW, Levi M, Florquin S, Speelman P, Carmeliet P, and Van der Poll T (2002) Urokinase receptor is necessary for adequate host defense against pneumococcal pneumonia. J Immunol 168:3507–3511
Raaphorst J, Groeneveld ABJ, Bossink AW, and Hack CE (2001) Early inhibition of activated fi brinolysis predicts microbial infection, shock and mortality in febrile medical patients. Thromb Haemost 86:543–549
Hermans PW and Hazelzet JA (2005) Plasminogen activator inhibitor type 1 gene polymorphism and sepsis. Clin Infect Dis 41(Suppl 7):S453–S458
Renckens R, Roelofs JJTH, Bonta PI, Florquin S, de Vries CJM, Levi M, Carmeliet P, Van 't Veer C, and Van der Poll T (2007) Plasminogen activator inhibitor type 1 is protective during severe Gram-negative pneumonia. Blood 109:1593–1601
Ossovskaya VS and Bunnett NW (2004) Protease-activated receptors: Contribution to physiology and disease. Physiol Rev 84:579–621
Coughlin SR (2000) Thrombin signalling and protease-activated receptors. Nature 407:258–264
Bunnett NW (2006) Protease-activated receptors: how proteases signal to cells to cause infl am-mation and pain. Semin Thromb Hemost 32(Suppl 1):39–48
Ruf W, Dorfleutner A, and Riewald M (2003) Specifi city of coagulation factor signaling. J Thromb Haemost 1:1495–1503
Nakanishi-Matsui M, Zheng YW, Sulciner DJ, Weiss EJ, Ludeman MJ, and Coughlin SR (2000) PAR3 is a cofactor for PAR4 activation by thrombin. Nature 404:609–613
Riewald M, Petrovan RJ, Donner A, Müller BM, and Ruf W (2002) Activation of endothelial cell protease activated receptor 1 by the protein C pathway. Science 296:1880–1882
Feistritzer C and Riewald M (2005) Endothelial barrier protection by activated protein C through PAR1-dependent sphingosine 1-phosphate receptor-1 crossactivation. Blood 105:3178–3184
Ludeman MJ, Kataoka H, Srinivasan Y, Esmon NL, Esmon CT, and Coughlin SR (2005) PAR1 cleavage and signaling in response to activated protein C and thrombin. J Biol Chem 280:13122–13128
Esmon CT (2005) Is APC activation of endothelial cell PAR1 important in severe sepsis?: No. J Thromb Haemost 3:1910–1911
Ruf W (2005) Is APC activation of endothelial cell PAR1 important in severe sepsis?: Yes. J Thromb Haemost 3:1912–1914
Pawlinski R, Pedersen B, Schabbauer G, Tencati M, Holscher T, Boisvert W, Andrade-Gordon P, Frank RD, and Mackman N (2004) Role of tissue factor and protease-activated receptors in a mouse model of endotoxemia. Blood 103:1342–1347
Kaneider NC, Leger AJ, Agarwal A, Nguyen N, Perides G, Derian C, Covic L, and Kuliopulos A (2007) “Role reversal” for the receptor PAR1 in sepsis-induced vascular damage. Nat Immunol 8(12):1303–1312
Camerer E, Cornelissen I, Kataoka H, Duong DN, Zheng YW, and Coughlin SR (2006) Roles of protease-activated receptors in a mouse model of endotoxemia. Blood 107:3912–3921
Niessen F, Schaffner F, Furlan-Freguia C, Pawlinski R, Bhattacharjee G, Chun J, Derian CK, Andrade-Gordon P, Rosen H, and Ruf W (2008) Dendritic cell PAR1-S1P3 signalling couples coagulation and infl ammation. Nature 452:654–658
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Schouten, M., van der Poll, T. (2009). Coagulation Disorders in Sepsis. In: Rello, J., Díaz, E., Rodríguez, A. (eds) Management of Sepsis: The PIRO Approach. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-00479-7_10
Download citation
DOI: https://doi.org/10.1007/978-3-642-00479-7_10
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-00478-0
Online ISBN: 978-3-642-00479-7
eBook Packages: MedicineMedicine (R0)