Abstract
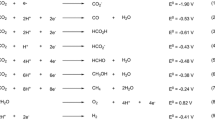
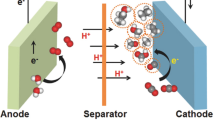
The dramatic increase in anthropogenic carbon dioxide emissions in recent decades has forced us to look for alternative carbon-neutral processes for the production of energy vectors and commodity chemicals. Photo- and electrochemical reduction of CO2 are appealing strategies for the storage of sustainable and intermittent energies in the form of chemical bonds of synthetic fuels and value-added molecules. In these approaches, carbon dioxide is converted to products such as CO, HCOOH and MeOH through proton-coupled electron transfer reactions. The use of earth-abundant elements as components of the catalytic materials is crucial for the large-scale applicability of this technology. This review summarizes the most recent advances related to this issue, with particular focus on studies where molecular metal complexes are used as catalysts. In addition, with the aim of aiding in the design of more robust and efficient non-noble metal-based catalysts, we discuss the lessons learned from the corresponding mechanistic studies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Rosas-Hernández, A., Steinlechner, C., Junge, H., Beller, M. (2017). Photo- and Electrochemical Valorization of Carbon Dioxide Using Earth-Abundant Molecular Catalysts. In: Wu, XF., Beller, M. (eds) Chemical Transformations of Carbon Dioxide . Topics in Current Chemistry Collections. Springer, Cham. https://doi.org/10.1007/978-3-319-77757-3_7
Download citation
DOI: https://doi.org/10.1007/978-3-319-77757-3_7
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-77756-6
Online ISBN: 978-3-319-77757-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)