Abstract
Osteoporosis is a global public health problem that imposes a large economic and medical burden on individuals and societies. Early identification of adults with osteoporosis may allow for interventions to reduce future fractures. Osteoporosis assessment involves evaluation of clinical risk factors and, in many cases, measurement of bone mineral density (BMD) by dual-energy X-ray absorptiometry (DXA). Screening recommendations vary by country, but North American guidelines call for routine bone density testing of all women aged 65 years and older, as well as selected younger men and women with clinical risk factors. Other countries recommend a case-finding strategy among high-risk adults. There is controversy over the optimal BMD screening interval. Estimation of future fracture risk using fracture risk calculators (with or without BMD) can aid in determining who should be screened and/or treated for osteoporosis. Treatment is recommended on the basis of history of fragility fracture, presence of low BMD T-score, or prediction of a high future risk of fracture. Despite guidelines, rates of osteoporosis screening and treatment remain low.
Similar content being viewed by others
Keywords
- Dual-energy X-ray absorptiometry (DXA)
- Bone mineral density (BMD)
- T-score
- Clinical risk factors
- Secondary osteoporosis
- Osteoporosis screening guidelines
- Fracture risk calculators
- FRAX
-
Osteoporosis is defined by low bone mass and skeletal fragility and is highly predictive of future fractures.
-
Fracture risk assessment takes into account clinical risk factors for osteoporosis and measurement of bone mineral density by dual-energy X-ray absorptiometry, if available.
-
In North America, routine bone density screening is recommended for all women aged 65 years and older, as well as for postmenopausal women and men aged 50 years and older with clinical risk factors for osteoporosis.
-
Pharmacologic osteoporosis treatment is recommended for adults with a history of fragility fracture, or bone density T-score ≤−2.5. In the United States, treatment is also advised for osteopenic adults who have a ≥3% risk of hip fracture or ≥20% risk of major osteoporotic fracture over the next 10 years.
Introduction
Osteoporosis is a disease defined by low bone mass and skeletal fragility that leads to an increased risk of fractures. Osteoporosis affects an estimated 200 million women worldwide [1] and disproportionately affects older adults. It is expected that the prevalence of osteoporosis may further increase as the population of adults aged 65 years and older is expected to double between 2010 and 2040 [2]. “Osteoporotic” fractures, also known as “low-trauma” or “fragility” fractures, are interchangeable terms that refer to fractures resulting from an impact equivalent to a fall of less than or equal to standing height. Approximately nine million osteoporotic fractures occur every year worldwide [3]. One out of every two women and one out of five men over the age of 50 years will have a low-trauma fracture in their lifetime [4, 5]. Osteoporotic fractures often lead to rapid deterioration of health status and decreased quality of life [6, 7]. These fragility fractures are also associated with high rates of mortality compared to age- and sex-matched controls [8, 9], and hip fractures in particular lead to 37% and 17% excess mortality in men and women, respectively [10, 11]. There is also a significant economic burden from osteoporotic fractures. As of 2005, fractures cost $20 billion a year in the United States and €36 billion a year in Europe [2].
Given these sobering statistics, screening for osteoporosis has been recommended to identify individuals at high risk of fracture who would benefit from treatments to minimize that risk. Osteoporosis screening typically involves assessments of clinical risk factors and measurement of bone mineral density (BMD).
Clinical Risk Factors
Numerous clinical factors have been identified that can aid in stratifying fracture risk for individual patients. Advancing age has been found to be an even stronger determinant of fracture risk than bone mass [12]. History of prior fragility fracture is also a robust risk factor for fracture, increasing future fracture probability by two to four times even after adjustment for age and BMD [13, 14]. Other validated risk factors that are independent of BMD include chronic glucocorticoid therapy, parental history of hip fracture, low body weight, current cigarette smoking, and excessive alcohol consumption [15].
Race and ethnicity are also clinical factors that influence fracture risk. In the United States, black and Asian-American women have lower rates of fracture than white, Hispanic, or Native American women [16, 17]. This relationship between race/ethnicity and fracture is not entirely mediated by bone density. For example, higher BMD among black women does not fully account for lower fracture rates [18], and Asian-American women experience fewer fractures despite having lower BMD than white women [19]. Worldwide rates of fracture also vary significantly by country, with the highest incidence of fractures occurring in Scandinavia [20].
As part of a thorough clinical assessment, it is important to evaluate for secondary causes of osteoporosis to inform prognosis and treatment decisions (Table 7.1). These disorders are associated with low bone density and increased fracture risk through varied mechanisms such as estrogen deficiency, vitamin D or calcium malabsorption, systemic inflammation, osteoblast/osteoclast toxicity, and/or high bone turnover states. Some of these secondary causes of osteoporosis are addressed in detail in other chapters of this volume.
Bone Mineral Density
The most commonly used technique to assess BMD in clinical practice is dual-energy X-ray absorptiometry (DXA). DXA provides precise and accurate assessments of the density of bone mineral (i.e., calcium hydroxyapatite) at clinically relevant sites such as the lumbar spine, proximal femur, and distal radius [21]. This bone-imaging technique involves low doses of ionizing radiation that are equivalent to daily background radiation and are ten times lower than the radiation exposure of a chest X-ray film [22]. There is a robust correlation between skeletal biomechanical strength and BMD measured by DXA [23].
Multiple studies have verified that low bone density at axial and appendicular sites predicts future osteoporotic fractures [24,25,26,27,28]. BMD T-scores quantify the standard deviation difference between a patient and a reference population of healthy young adults. For every standard deviation decrease in age-adjusted BMD, there is a roughly twofold increase in the risk of fracture [28]. This leads to an exponential increase in fracture risk, such that a patient with a T-score of −3.0 will have an eightfold higher risk of fracture than a person of the same age with a T-score of 0. Although low BMD at any site can predict osteoporotic fractures at all sites, it has better predictive ability at the site of measurement. For example, hip BMD is superior to BMD measured at other skeletal sites for predicting hip fracture [24, 25].
A World Health Organization (WHO) working group developed a categorization of BMD based on T-scores, with T-scores of −1.0 or above classified as normal, T-scores between −1.0 and −2.5 classified as osteopenia, and T-scores of −2.5 or below classified as osteoporosis [29]. These classifications apply to postmenopausal women and men of age 50 years and older. Despite these discrete T-score categorizations, it is important to recognize that there is no clear threshold below which fracture risk is suddenly increased. On the contrary, the relationship between BMD and fracture risk is continuous [29]. Thus, while fracture rates are highest among women with osteoporotic T-scores of −2.5 or below, the majority of women who fracture have osteopenic or normal BMD. Studies estimate that between 55% and 80% of women who fracture have nonosteoporotic T-scores [30,31,32]. Using a threshold T-score of ≤−2.5 has a sensitivity of 46% and specificity of 84% for identifying adults who will sustain osteoporotic fractures [32].
In summary, DXA provides BMD measurements that are highly predictive of future fracture risk, but risk stratification based on T-score alone is insufficient to identify the majority of individuals who will fracture. Thus, an effective screening strategy should additionally incorporate other factors that are independent of BMD to identify those who are at high risk of fracture and would benefit from intervention.
Osteoporosis Evaluation: Putting It All Together
A comprehensive evaluation for osteoporosis involves assessment of both clinical risk factors and bone mineral density by DXA. Suggested components of a clinical osteoporosis evaluation are presented in Table 7.2. A complete medical history, basic laboratory evaluation, and DXA scan will provide useful information to risk stratify patients. Selected patients with medical history suggestive of secondary causes may further benefit from additional targeted laboratory testing.
Fracture Risk Calculators
One commonly proposed osteoporosis screening strategy involves identification of high-risk individuals based on assessment of their absolute fracture risk after taking into account clinical risk factors and/or BMD. Fracture risk calculators have been developed to integrate clinical variables with or without BMD into a model that will predict an individual’s future fracture risk.
FRAX Calculator
The most studied calculator is the fracture risk assessment tool (FRAX) algorithm [33], which provides estimates of an individual’s 10-year probability of hip fracture or major osteoporotic (combined clinical spine, hip, shoulder, and wrist) fracture. Fracture risk assessment tool (FRAX) was derived using data from 9 international cohorts and has been validated in more than 26 external cohorts [34]. This algorithm incorporates 11 patient factors (i.e., age, sex, height, weight, prior fracture, parental hip fracture, smoking, alcohol, glucocorticoids, rheumatoid arthritis, and either secondary osteoporosis or BMD) to calculate an individual’s fracture risk.
Unique strengths of the FRAX algorithm are that it takes into account country-specific epidemiology and also incorporates competing mortality risk, thus potentially providing more accurate assessments among elderly patients [35]. An important caveat is that FRAX has only been evaluated in treatment-naive populations, and therefore should not be used to predict fractures in patients who are currently taking or have previously received pharmacologic osteoporosis treatments. In addition, FRAX has been shown to systematically underestimate fracture risk among adults with type 2 diabetes [36, 37]. Correction factors have been created to modify FRAX estimates based on discordant spine/hip T-scores [38], glucocorticoid dose [39], and vertebral textural assessment by trabecular bone score (TBS) [40].
Other Fracture Calculators
Many other fracture risk calculators have been developed that vary in scope and complexity. The most complex of these, the updated QFracture algorithm, encompasses 31 risk factors [41]. On the other end of the spectrum, Garvan involves five risk factors and takes into account dose–response relationships with prior fractures and falls to predict 5- or 10-year fracture risk [42, 43]. These calculators have been externally validated and have roughly similar discriminative power as FRAX to predict fractures [34, 44]. To date, however, only FRAX has been incorporated into national osteoporosis guidelines for screening and intervention (Table 7.3).
Screening Guidelines
North American Recommendations
In the United States and Canada, routine BMD screening with DXA is recommended in all women aged 65 years and older. This recommendation is endorsed by the US Preventative Services Task Force (USPSTF, grade B recommendation) [46] as well as multiple expert groups (Canadian Osteoporosis Society [47], American Association of Clinical Endocrinology (AACE) [48], American Association of Family Practice (AAFP) [49], National Osteoporosis Foundation (NOF) [50], International Society for Clinical Densitometry (ISCD) [51], North American Menopause Society (NAMS) [52], American College of Preventative Medicine (ACPM) [53], and American College of Obstetrics and Gynecology (ACOG) [54]).
There is less consensus about the utility of routine osteoporosis screening in older men. The USPSTF did not find sufficient evidence to recommend routine screening for men [46]. The Canadian Osteoporosis Society recommends BMD testing for all men older than 65 years [47], while the Endocrine Society, NOF, ACPM, and ISCD suggest that all men aged 70 years and older should be screened for low BMD [50, 51, 53, 55]. The American College of Physicians (ACP) recommends DXA tests in men who are at increased risk for osteoporosis (including men aged >70 years) and are candidates for drug therapy (strong recommendation, moderate quality evidence [56]).
North American guidelines also call for BMD screening of selected younger postmenopausal women and men who have clinical risk factors for osteoporosis. For example, the USPSTF suggests measuring BMD in women aged between 50 and 64 years whose FRAX-calculated 10-year risk of major osteoporotic fracture is ≥8.4%, which is the equivalent 10-year fracture risk of a 65-year-old white woman with no other fracture risk factors (grade B recommendation) [46]. While this approach to BMD screening has practical merit, the proposed FRAX threshold has neither undergone cost-effectiveness analysis nor has it been validated in any patient population. Other expert groups recommend DXA tests in men and women older than 50 years with clinical risk factors for fracture [47, 48, 50,51,52,53, 55].
Screening Recommendations from Around the World
Screening guidelines differ in different regions of the world where different cost–benefit models are employed [57]. In Japan, routine BMD screening in women starts as early as age 40 years [58], whereas Australian guidelines recommend DXA tests in all men and women aged 70 years and older [59].
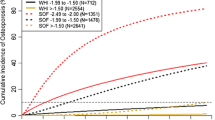
In most of Europe, a case-finding approach is taken for osteoporosis screening, with recommendations for BMD testing based on risk stratification [60]. In particular, the decision to obtain a DXA test for an individual is based upon age-specific fracture probability thresholds calculated using FRAX (without BMD information). Low-risk individuals are recommended not to have a DXA test, given the lower likelihood of finding a low BMD that would necessitate intervention. Importantly, the fracture probability thresholds vary considerably by age, with older women needing to surpass a higher fracture threshold before BMD testing is recommended (see Fig. 7.1). Certain European countries, such as the United Kingdom, take a further parsimonious approach by applying an upper threshold to BMD testing, whereby adults with the highest probability of fracture are recommended to start osteoporosis treatment without requiring a screening BMD test, although BMD measurements might be obtained to monitor treatment. In this scenario, only adults with an intermediate fracture probability, in whom the addition of BMD results might change the decision for intervention, are referred for BMD testing [61].
Osteoporosis assessment guidelines based on the 10-year probability of a major fracture. The dotted line signifies the treatment intervention threshold. A bone mineral density test is recommended for individuals where the probability assessment lies in the orange region [60]. (With permission from Springer Nature)
Effectiveness of Osteoporosis Screening Approaches
Several cohort studies have suggested that BMD screening can improve osteoporosis treatment rates and potentially decrease fractures [62, 63]. However, confounding in these observational studies may be difficult to characterize and may hamper broad interpretation. A US-based modeling study found that screening strategies that initiate BMD screening in women as young as 55 years old were more effective and less expensive than not implementing any screening strategy [64].
Only three randomized controlled trials have been performed to study the effectiveness of osteoporosis screening. In one study of women aged 45–54 years who were randomized to receive screening BMD or usual care, the screened group had greater utilization of osteoporosis medications over a 9-year follow-up period [65]. As this study was initiated prior to the widespread introduction of T-scores, women were recommended to start on treatment if their bone density was in the lowest quartile of the population. In intention-to-treat analyses, no significant difference in fractures rates was found in the screened and control groups, although per-protocol analyses restricted to women who received DXA scans demonstrated a reduction in risk of fractures as compared to controls (hazard ratio (HR) = 0.741, 95% confidence interval [CI]: 0.551–0.998).
Another large randomized screening trial evaluated the effectiveness of a FRAX-based community screening program on 12,495 women aged 70–85 years in the United Kingdom screening in the community to reduce fractures in older women (SCOOP) trial [66]. Women were randomized to usual care versus risk stratification based on the FRAX algorithm, with the selected use of BMD testing in women who exceeded an age-specific FRAX threshold. Among the 14% of the study population that was deemed to be at high fracture risk, 78% initiated treatment. Over 5 years of follow-up, screening did not reduce the primary endpoint of all osteoporosis-related fractures but did lead to a 28% reduction in hip fractures (HR = 0.72, 95% CI: 0.59–0.89), a prespecified secondary outcome. Furthermore, this systematic community-based screening program was highly cost-effective [67].
The largest randomized trial of osteoporosis screening involved 34,229 Danish women aged 65–80 years (ROSE trial) [68]. Similar to the design of SCOOP, this study also involved a case-finding strategy using FRAX-based thresholds to guide DXA scan recommendations. Unlike SCOOP, treatment recommendations were based solely on BMD results and did not take into account FRAX intervention thresholds. After a median follow-up of 5 years, there was a small increase in osteoporosis medication use in the screening group (23% versus 18%, p < 0.001) but no significant differences in fracture rates were observed compared to controls in intention-to-treat analyses. Interpretation of this study is limited given high drop-out rates of high-risk women before DXA scan. Per-protocol analyses among women who received DXA scans did demonstrate a reduction in major osteoporotic fractures (HR = 0.87, 95% CI: 0.77–0.99) and hip fractures (HR = 0.74, 95% CI: 0.58–0.95) in comparison with controls.
Steps After Screening
Intervention Thresholds
Individuals are diagnosed with osteoporosis if they have sustained a fragility fracture, or if their T-score is ≤−2.5 at the posterior–anterior lumbar spine, total hip, femoral neck, or 1/3 radius [51]. Postmenopausal women and men of age 50 years and older who have osteoporosis by this definition are recommended to start on pharmacologic therapy to reduce their fracture risk. Numerous randomized controlled trials have demonstrated antifracture efficacy of osteoporosis medications among individuals with osteoporotic T-scores [46].
Given the poor sensitivity of using osteoporotic T-score thresholds as a case-finding strategy, US guidelines also recommend providing pharmacologic intervention for high-risk osteopenic patients, as defined as adults whose 10-year fracture risk exceeds 3% at the hip or 20% for major osteoporotic fracture, as calculated by FRAX [48, 50, 52, 54, 69]. These criteria were developed from US-based cost-effectiveness analyses assuming osteoporosis pharmacotherapy costs of $600/year [70], and may not apply to other countries. The subsequent availability of both cheaper generic drugs (<$100/year) as well as newer more expensive therapies could conceivably alter the US cost-effectiveness thresholds. It is important to note that there have been no trials that have studied fracture prevention within the specific high-risk osteopenic population identified by these FRAX criteria. Nevertheless, a 6-year randomized placebo-controlled trial of 2000 women with osteopenic bone density found significant reductions in vertebral and nonvertebral fractures with zoledronic acid treatment [71], demonstrating proof of concept that pharmacotherapy in an osteopenic population can be beneficial. Furthermore, many Food and Drug Administration (FDA)-approved osteoporosis therapies have demonstrated no statistical interaction between fracture efficacy and baseline fracture risk as assessed by FRAX, which suggests that these medications have similar ability to reduce fractures across many baseline probabilities of fracture [72,73,74,75].
In Europe, osteoporosis treatment is recommended for women with a history of prior fragility fracture, and among men and women who surpass age-dependent intervention thresholds based on FRAX-calculated 10-year risk of major osteoporotic fracture (see Fig. 7.1) [61].Many other countries have instituted a tailored combination of fixed and/or age-dependent intervention thresholds based on FRAX [76].
Screening Intervals
The optimal BMD screening interval remains unclear for individuals who do not meet initial intervention thresholds. Several screening guidelines suggest a minimum of 2 years between repeated BMD tests based on limitations in the precision of DXA testing [46, 50]. However, there are conflicting data as to whether follow-up testing and/or rate of bone loss enhances population-based fracture risk prediction [77,78,79,80,81,82].
Two studies have investigated the length of time for individuals with normal or osteopenic bone densities to develop osteoporosis [83, 84]. As might be expected, both studies reported that the time intervals were highly dependent on baseline T-scores, with osteopenic baseline BMD predicting a shorter time to osteoporosis transition as compared to normal baseline BMD. One study in men and women over the age of 60 years found that this timing interval was also highly dependent on age, such that older adults with normal BMD transition to osteoporosis more quickly than younger adults [84]. Of note, osteoporosis diagnostic thresholds (based on T-score of −2.5 or lower) differ from treatment thresholds (based partly on absolute fracture risk), and therefore individuals with moderate or high absolute fracture risk may benefit from more frequent bone density testing to identify the proper timing for intervention [85]. Thus, while younger adults with normal BMD may not require repeat testing for 10–15 years, older patients and those with moderate or advanced osteopenia might benefit from testing in 2–5 years. Ultimately, repeat screening BMD testing is most likely to be valuable for individuals with risk factors or comorbidities that might lead to accelerated bone loss.
Screening and Diagnosis in Current Clinical Practice
Despite the significant burden of osteoporosis and the availability of screening tools and treatments, osteoporosis continues to be underdiagnosed. In the United States, less than half of women recommended for bone density screening received a bone density test [86]. There has been a decline in BMD testing for younger postmenopausal women since 2006 [87]. Only 34–42% of adults who have sustained fragility fractures get evaluation or osteoporosis treatment [88]. Given the large public health impact of osteoporosis, there is more work to be done to improve rates of osteoporosis screening to address these important diagnostic and treatment gaps.
References
Kanis JA, World Health Organization Scientific Group. Assessment of osteoporosis at the primary health care level Technical Report Sheffield. United Kingdom: World Health Organization Collaborating Centre for Metabolic Bone Diseases; 2008. p. 1–339.
Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res. 2007;22(3):465–75.
Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int. 2006;17(12):1726–33.
Lippuner K, Johansson H, Kanis JA, Rizzoli R. Remaining lifetime and absolute 10-year probabilities of osteoporotic fracture in swiss men and women. Osteoporos Int. 2009;20(7):1131–40.
Park C, Ha YC, Jang S, Jang S, Yoon HK, Lee YK. The incidence and residual lifetime risk of osteoporosis-related fractures in Korea. J Bone Miner Metab. 2011;29(6):744–51.
Pisani P, Renna MD, Conversano F, Casciaro E, Di Paola M, Quarta E, et al. Major osteoporotic fragility fractures: risk factor updates and societal impact. World J Orthop. 2016;7(3):171–81.
Roux C, Wyman A, Hooven FH, Gehlbach SH, Adachi JD, Chapurlat RD, et al. Burden of non-hip, non-vertebral fractures on quality of life in postmenopausal women: the global longitudinal study of osteoporosis in women (GLOW). Osteoporos Int. 2012;23(12):2863–71.
Bliuc D, Nguyen ND, Milch VE, Nguyen TV, Eisman JA, Center JR. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301(5):513–21.
Center JR, Nguyen TV, Schneider D, Sambrook PN, Eisman JA. Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet. 1999;353(9156):878–82.
Forsén L, Sogaard AJ, Meyer HE, Edna T, Kopjar B. Survival after hip fracture: short and long-term excess mortality according to age and gender. Osteoporosis Int. 1999;10(1):73–8.
Kanis JA, Johnell O, Oden A, Borgstrom F, Johansson H, De Laet C, et al. Intervention thresholds for osteoporosis in men and women: a study based on data from Sweden. Osteoporos Int. 2005;16(1):6–14.
Kanis JA, Johnell O, Oden A, Dawson A, De Laet C, Jonsson B. Ten year probabilities of osteoporotic fractures according to BMD and diagnostic thresholds. Osteoporos Int. 2001;12(12):989–95.
Black DM, Arden NK, Palermo L, Pearson J, Cummings SR. Prevalent vertebral deformities predict hip fractures and new vertebral deformities but not wrist fractures. J Bone Miner Res. 1999;14(5):821–8.
Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA, Berger M. Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res. 2010;15(4):721–39.
Kanis JA, Borgstrom F, De Laet C, Johansson H, Johnell O, Jonsson B, et al. Assessment of fracture risk. Osteoporos Int. 2005;16(6):581–9.
Leslie WD. Clinical review: ethnic differences in bone mass--clinical implications. J Clin Endocrinol Metab. 2012;97(12):4329–40.
Wright NC, Saag KG, Curtis JR, Smith WK, Kilgore ML, Morrisey MA, et al. Recent trends in hip fracture rates by race/ethnicity among older US adults. J Bone Miner Res. 2012;27(11):2325–32.
Cauley JA, Lui LY, Ensrud KE, Zmuda JM, Stone KL, Hochberg MC, et al. Bone mineral density and the risk of incident nonspinal fractures in black and white women. JAMA. 2005;293(17):2102–8.
Barrett-Connor E, Siris ES, Wehren LE, Miller PD, Abbott TA, Berger ML, et al. Osteoporosis and fracture risk in women of different ethnic groups. J Bone Miner Res. 2005;20(2):185–94.
Svedbom A, Hernlund E, Ivergård M, Compston J, Cooper C, Stenmark J, et al. Osteoporosis in the European Union: a compendium of country-specific reports. Arch Osteoporos. 2013;8:137.
Mazess R, Chesnut CH 3rd, McClung M, Genant H. Enhanced precision with dual-energy X-ray absorptiometry. Calcif Tissue Int. 1992;51(1):14–7.
Njeh CF, Fuerst T, Hans D, Blake GM, Genant HK. Radiation exposure in bone mineral density assessment. Appl Radiat Isot. 1999;50(1):215–36.
Lotz JC, Cheal EJ, Hayes WC. Fracture prediction for the proximal femur using finite element models: part I – linear analysis. J Biomech Eng. 1991;113(4):353–60.
Leslie WD, Tsang JF, Caetano PA, Lix LM. Manitoba bone density P. Effectiveness of bone density measurement for predicting osteoporotic fractures in clinical practice. J Clin Endocrinol Metab. 2007;92(1):77–81.
Stone KL, Seeley DG, Lui LY, Cauley JA, Ensrud K, Browner WS, et al. BMD at multiple sites and risk of fracture of multiple types: long-term results from the study of osteoporotic fractures. J Bone Miner Res. 2003;18(11):1947–54.
Johnell O, Kanis JA, Oden A, Johansson H, De Laet C, Delmas P, et al. Predictive value of BMD for hip and other fractures. J Bone Miner Res. 2005;20(7):1185–94.
Black DM, Cummings SR, Genant HK, Nevitt MC, Palermo L, Browner W. Axial and appendicular bone density predict fractures in older women. J Bone Miner Res. 1992;7(6):633–8.
Marshall D, Johnell O, Wedel H. Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ. 1996;312(7041):1254–9.
Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO study group. World Health Organ Tech Rep Ser. 1994;843:1–129.
Schuit SCE, van der Klift M, Weel AEAM, de Laet CEDH, Burger H, Seeman E, et al. Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam study. Bone. 2004;34(1):195–202.
Siris ES, Chen Y-T, Abbott TA, Barrett-Connor E, Miller PD, Wehren LE, et al. 128 bone mineral density thresholds for pharmacological intervention to prevent fractures. Arch Intern Med. 2004;164(10):1108–12.
Wainwright SA, Marshall LM, Ensrud KE, Cauley JA, Black DM, Hillier TA, et al. Hip fracture in women without osteoporosis. J Clin Endocrinol Metab. 2005;90(5):2787–93.
FRAX. Fracture Risk Assessment Tool. Centre for Metabolic Bone Diseases UoS, UK. Available from: https://www.sheffield.ac.uk/FRAX/index.aspx.
Dagan N, Cohen-Stavi C, Leventer-Roberts M, Balicer RD. External validation and comparison of three prediction tools for risk of osteoporotic fractures using data from population based electronic health records: retrospective cohort study. BMJ. 2017;356:i6755.
Leslie WD, Lix LM, Wu X, Manitoba Bone Density P. Competing mortality and fracture risk assessment. Osteoporos Int. 2013;24(2):681–8.
Giangregorio LM, Leslie WD, Lix LM, Johansson H, Oden A, McCloskey E, et al. FRAX underestimates fracture risk in patients with diabetes. J Bone Miner Res. 2012;27(2):301–8.
Schwartz AV, Vittinghoff E, Bauer DC, Hillier TA, Strotmeyer ES, Ensrud KE, et al. Association of BMD and FRAX score with risk of fracture in older adults with type 2 diabetes. JAMA. 2011;305(21):2184–92.
Leslie WD, Lix LM, Johansson H, Oden A, McCloskey E, Kanis JA. Spine-hip discordance and fracture risk assessment: a physician-friendly FRAX enhancement. Osteoporos Int. 2011;22(3):839–47.
Kanis JA, Johansson H, Oden A, McCloskey EV. Guidance for the adjustment of FRAX according to the dose of glucocorticoids. Osteoporos Int. 2011;22(3):809–16.
McCloskey EV, Oden A, Harvey NC, Leslie WD, Hans D, Johansson H, et al. A meta-analysis of trabecular bone score in fracture risk prediction and its relationship to FRAX. J Bone Miner Res. 2016;31(5):940–8.
Hippisley-Cox J, Coupland C. Derivation and validation of updated QFracture algorithm to predict risk of osteoporotic fracture in primary care in the United Kingdom: prospective open cohort study. BMJ. 2012;344:e3427.
Nguyen ND, Frost SA, Center JR, Eisman JA, Nguyen TV. Development of prognostic nomograms for individualizing 5-year and 10-year fracture risks. Osteoporos Int. 2008;19(10):1431–44.
Garvin-website. Fracture risk calculator. Garvin Institute. Available from: www.garvan.org.au/promotions/bone-fracture-risk/calculator/.
Rubin KH, Abrahamsen B, Friis-Holmberg T, Hjelmborg JV, Bech M, Hermann AP, et al. Comparison of different screening tools (FRAX(R), OST, ORAI, OSIRIS, SCORE and age alone) to identify women with increased risk of fracture. A population-based prospective study. Bone. 2013;56(1):16–22.
Leslie WD, Lix LM. Comparison between various fracture risk assessment tools. Osteoporos Int. 2014;25(1):1–21.
Viswanathan M, Reddy S, Berkman N, Cullen K, Middleton JC, Nicholson WK, et al. Screening to prevent osteoporotic fractures: updated evidence report and systematic review for the US preventive services task force. JAMA. 2018;319(24):2532–51.
Papaioannou A, Morin S, Cheung AM, Atkinson S, Brown JP, Feldman S, et al. Clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ. 2010;182(17):1864–73.
Camacho PM, Petak SM, Binkley N, Clarke BL, Harris ST, Hurley DL, et al. American Association of Clinical Endocrinologists and American College of endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis – 2016. Endocr Pract. 2016;22(Suppl 4):1–42.
Jeremiah MP, Unwin BK, Greenawald MH, Diagnosis CVE. Management of osteoporosis. Am Fam Physician. 2015;92(4):261–8.
Cosman F, de Beur SJ, LeBoff MS, Lewiecki EM, Tanner B, Randall S, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10):2359–81.
Densitometry ISfC. 2015 ISCD official positions – adult. Middletown: International Society for Clinical Densitometry, Inc; 2015. Available from: http://www.iscd.org/official-positions/2015-iscd-official-positions-adult/.
NAMS. Management of osteoporosis in postmenopausal women: 2010 position statement of the North American Menopause Society. Menopause. 2010;17(1):25–54.. quiz 5-6
Lim LS, Hoeksema LJ, Sherin K, Committee APP. Screening for osteoporosis in the adult U.S. population: ACPM position statement on preventive practice. Am J Prev Med. 2009;36(4):366–75.
Committee on Practice Bulletins-Gynecology TACoO, Gynecologists. ACOG Practice Bulletin N. 129. Osteoporosis. Obstet Gynecol. 2012;120(3):718–34.
Watts NB, Adler RA, Bilezikian JP, Drake MT, Eastell R, Orwoll ES, et al. Osteoporosis in men: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2012;97(6):1802–22.
Qaseem A, Snow V, Shekelle P, Hopkins R Jr, Forciea MA, Owens DK, et al. Screening for osteoporosis in men: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2008;148(9):680–4.
Leslie WD, Schousboe JT. A review of osteoporosis diagnosis and treatment options in new and recently updated guidelines on case finding around the world. Curr Osteoporos Rep. 2011;9(3):129–40.
Orimo H, Nakamura T, Hosoi T, Iki M, Uenishi K, Endo N, et al. Japanese 2011 guidelines for prevention and treatment of osteoporosis--executive summary. Arch Osteoporos. 2012;7:3–20.
Australia TRACoGPaO. Osteoporosis prevention diagnosis and management in postmenopausal women and men over 50 years of age. 2nd ed. East Melbourne: RACGP; 2017.
Kanis JA, McCloskey EV, Johansson H, Cooper C, Rizzoli R, Reginster JY, et al. European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 2013;24(1):23–57.
Kanis JA, McCloskey EV, Johansson H, Strom O, Borgstrom F, Oden A, et al. Case finding for the management of osteoporosis with FRAX--assessment and intervention thresholds for the UK. Osteoporos Int. 2008;19(10):1395–408.
Kern LM, Powe NR, Levine MA, Fitzpatrick AL, Harris TB, Robbins J, et al. Association between screening for osteoporosis and the incidence of hip fracture. Ann Intern Med. 2005;142(3):173–81.
Buist DS, LaCroix AZ, Brenneman SK, Abbott T 3rd. A population-based osteoporosis screening program: who does not participate, and what are the consequences? J Am Geriatr Soc. 2004;52(7):1130–7.
Nayak S, Roberts MS, Greenspan SL. Cost-effectiveness of different screening strategies for osteoporosis in postmenopausal women. Ann Intern Med. 2011;155(11):751–61.
Barr RJ, Stewart A, Torgerson DJ, Reid DM. Population screening for osteoporosis risk: a randomised control trial of medication use and fracture risk. Osteoporos Int. 2010;21(4):561–8.
Shepstone L, Lenaghan E, Cooper C, Clarke S, Fong-Soe-Khioe R, Fordham R, et al. Screening in the community to reduce fractures in older women (SCOOP): a randomised controlled trial. Lancet. 2018:391(10122):2017:741–7.
Turner DA, Khioe RFS, Shepstone L, Lenaghan E, Cooper C, Gittoes N, et al. The cost-effectiveness of screening in the community to reduce osteoporotic fractures in older women in the UK: economic evaluation of the SCOOP study. J Bone Miner Res. 2018;33(5):845–51.
Rubin KH, Rothmann MJ, Holmberg T, Hoiberg M, Moller S, Barkmann R, et al. Effectiveness of a two-step population-based osteoporosis screening program using FRAX: the randomized risk-stratified osteoporosis strategy evaluation (ROSE) study. Osteoporos Int. 2018;29(3):567–78.
NBHA AMoNBH, Alliance. World Health Organization – WHO criteria for diagnosis of osteoporosis 2008. Available from: http://www.4bonehealth.org/education/world-health-organization-criteria-diagnosis-osteoporosis/.
Tosteson AN, Melton LJ 3rd, Dawson-Hughes B, Baim S, Favus MJ, Khosla S, et al. Cost-effective osteoporosis treatment thresholds: the United States perspective. Osteoporos Int. 2008;19(4):437–47.
Reid I, Horne AM, Mihov B, et al. Fracture prevention with zoledronate in older women with osteopenia. NEJM. 2018; https://doi.org/10.1056/NEJMoa1808082. [Epub ahead of print].
Donaldson MG, Palermo L, Ensrud KE, Hochberg MC, Schousboe JT, Cummings SR. Effect of alendronate for reducing fracture by FRAX score and femoral neck bone mineral density: the fracture intervention trial. J Bone Miner Res. 2012;27(8):1804–10.
Kanis JA, Johansson H, Oden A, McCloskey EV. A meta-analysis of the efficacy of raloxifene on all clinical and vertebral fractures and its dependency on FRAX. Bone. 2010;47(4):729–35.
Harvey NC, Kanis JA, Oden A, Nakamura T, Shiraki M, Sugimoto T, et al. Efficacy of weekly teriparatide does not vary by baseline fracture probability calculated using FRAX. Osteoporos Int. 2015;26(9):2347–53.
McCloskey EV, Johansson H, Oden A, Harvey NC, Jiang H, Modin S, et al. The effect of Abaloparatide-SC on fracture risk is independent of baseline FRAX fracture probability: a post hoc analysis of the ACTIVE study. J Bone Miner Res. 2017;32(8):1625–31.
Kanis JA, Harvey NC, Cooper C, Johansson H, Oden A, McCloskey EV, et al. A systematic review of intervention thresholds based on FRAX : a report prepared for the National Osteoporosis Guideline Group and the international osteoporosis foundation. Arch Osteoporos. 2016;11(1):25.
Hillier TA, Stone KL, Bauer DC, Rizzo JH, Pedula KL, Cauley JA, et al. Evaluating the value of repeat bone mineral density measurement and prediction of fractures in older women: the study of osteoporotic fractures. Arch Intern Med. 2007;167(2):155–60.
Leslie WD, Morin SN, Lix LM. Manitoba bone density P. Rate of bone density change does not enhance fracture prediction in routine clinical practice. J Clin Endocrinol Metab. 2012;97(4):1211–8.
Berger C, Langsetmo L, Joseph L, Hanley DA, Davison KS, Josse RG, et al. Association between change in BMD and fragility fracture in women and men. J Bone Miner Res. 2009;24(2):361–70.
Nguyen TV, Center JR, Eisman JA. Femoral neck bone loss predicts fracture risk independent of baseline BMD. J Bone Miner Res. 2005;20(7):1195–201.
Ahmed LA, Emaus N, Berntsen GK, Bjornerem A, Fonnebo V, Jorgensen L, et al. Bone loss and the risk of non-vertebral fractures in women and men: the Tromso study. Osteoporos Int. 2010;21(9):1503–11.
Berry SD, Samelson EJ, Pencina MJ, McLean RR, Cupples LA, Broe KE, et al. Repeat bone mineral density screening and prediction of hip and major osteoporotic fracture. JAMA. 2013;310(12):1256–62.
Gourlay ML, Fine JP, Preisser JS, May RC, Li C, Lui LY, et al. Bone-density testing interval and transition to osteoporosis in older women. N Engl J Med. 2012;366(3):225–33.
Frost SA, Nguyen ND, Center JR, Eisman JA, Nguyen TV. Timing of repeat BMD measurements: development of an absolute risk-based prognostic model. J Bone Miner Res. 2009;24(11):1800–7.
Yu EW, Finkelstein JS. Bone density screening intervals for osteoporosis: one size does not fit all. JAMA. 2012;307(24):2591–2.
King AB, Fiorentino DM. Medicare payment cuts for osteoporosis testing reduced use despite tests' benefit in reducing fractures. Health Aff. 2011;30(12):2362–70.
Overman RA, Farley JF, Curtis JR, Zhang J, Gourlay ML, Deal CL. DXA utilization between 2006 and 2012 in commercially insured younger postmenopausal women. J Clin Densitom. 2015;18(2):145–9.
NCQA. NCQA – osteoporosis testing and management in older women (OTO, OMW). Available from: http://www.ncqa.org/report-cards/health-plans/state-of-health-care-quality/2017-table-of-contents/osteoporosis.
Acknowledgments
I am grateful to Michael Cheney, NP, for his editing and thoughtful suggestions for this chapter.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Yu, E.W. (2020). Osteoporosis Screening and Diagnosis. In: Leder, B., Wein, M. (eds) Osteoporosis. Contemporary Endocrinology. Humana, Cham. https://doi.org/10.1007/978-3-319-69287-6_7
Download citation
DOI: https://doi.org/10.1007/978-3-319-69287-6_7
Published:
Publisher Name: Humana, Cham
Print ISBN: 978-3-319-69286-9
Online ISBN: 978-3-319-69287-6
eBook Packages: MedicineMedicine (R0)