Abstract
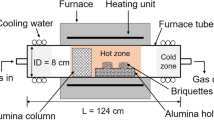
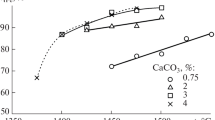
The Nkana Cu-Co mineral sulphide concentrates containing 40 wt.% SiO2 were carbothermically reduced in the presence of CaO by following the equilibrium: MS + CaO + C = M + CaS + CO(g), where M represents the metallic Cu, Co and Fe, over a temperature range of 1073 K – 1323 K. We investigated the magnetic separation of alloy phases into copper-cobalt rich and iron-cobalt rich fractions. The proposed approach appears more energy efficient than melting a mixture which yields a ternary alloy composed of Cu, Co and Fe, and a non-magnetic fraction (CaS, excess CaO and SiO2). The extent of magnetic separation was affected by the temperature and mole ratios of MS/CaO and C/CaO, used for the reduction of mineral concentrate. The reduced and magnetically separated samples were characterised by XRD, SEM-EDX and XRF techniques. The alloy phase contained ~92 wt% of metallic constituents, with remaining 8 wt.% as mixture of CaS and SiO2. The utilisation of this gangue mixture in the process is briefly discussed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Davenport, W.G.L., et al., Extractive Metallurgy of Copper (4th Edition) . Chemical, Petrochemical & Process. 2002: Elsevier. 1–452.
Banda, W., High Temperature Phase Equilibria in the Fe-Co-Cu-Si system Pertaining to slag cleaning, in Department of Process Engineering2006, Stellenbosch: Stellenbosch.
Jones, R.T., et al., Recovery of cobalt from slag in a DC arc furnace at Chambishi, Zambia. Journal of The South African Institute of Mining and Metallurgy, 2002. 102(Compendex): p. 5–9.
Banda, W., N.T. Beukes, and J.J. Eksteen, Factors influencing base metal recovery from waste reverberatory furnace slags in a 50 kVA laboratory DC plasma arc furnace. Journal of The South African Institute of Mining and Metallurgy, 2004. 104(3): p. 201–207.
Hara, Y.R.S. Reduction Mechanisms of Copper, Cobalt and Iron during Low Temperature Recovery from Mineral Sulphide Concentrates. in Characterization of Minerals, Metals, and Materials 2013 2013. John Wiley & Sons.
Hara, Y. and A. Jha, Carbothermic reduction of Zambian sulphide concentrates in presence of lime. Mineral Processing and Extractive Metallurgy, 2013. 122(3): p. 146–156.
Jha, A., S.H. Tang, and A. Chrysanthou, Phase equilibria in the metal-sulfur-oxygen system and selective reduction of metal oxides and sulfides 1. The carbothermic reduction and calcination of complex mineral sulfides. Metallurgical and Materials Transactions B-Process Metallurgy and Materials Processing Science, 1996. 27(5): p. 829–840.
Bale, C.W., et al., FactSage thermochemical software and databases. Calphad: Computer Coupling of Phase Diagrams and Thermochemistry, 2002. 26(2): p. 189–228.
West, E.G., Copper and its alloys. 1982, Chichester: Ellis Horwood.
Cullity, B.D., Introduction to Magnetic Materials. 1972, London Addison-Wesley.
Jones, R.T. and A.C. Deneys, Using a direct-current arc furnace to recover cobalt from slags. JOM, 1998. 50(10): p. 53–56.
Hara, Y.R.S. and A. Jha. A Novel Low Energy Route for the Extraction of Copper and Cobalt Metals/Alloys from the Zambian Sulphide Concentrates. in Characterization of Minerals, Metals, and Materials. 2012. John Wiley & Sons.
Hara, Y.S.R., Mineral sulphide-lime reactions and effect of CaO/C mole ratio during carbothermic reduction of complex mineral sulphides. International Journal of Minerals, Metallurgy, and Materials, 2014. 21(1): p. 1–11.
Hara, Y.S.R., Study of Reaction Mechanisms for Copper-Cobalt-Iron Sulfide Concentrates in the Presence of Lime and Carbon. JOM, 2013: p. 1–11.
Roine, A., HSC Chemistry 5.1, 2002, Outokumpu Research Oy: Finland.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 TMS (The Minerals, Metals & Materials Society)
About this chapter
Cite this chapter
Hara, Y.R.S., Jha, A. (2015). Energy Efficient Separation of Magnetic Alloy fron the Carbothermic Reduction of NKANA Cu-Co Concentrates. In: Jha, A., et al. Energy Technology 2015. Springer, Cham. https://doi.org/10.1007/978-3-319-48220-0_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-48220-0_10
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48602-4
Online ISBN: 978-3-319-48220-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)