Abstract
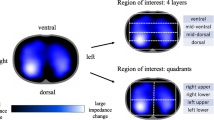
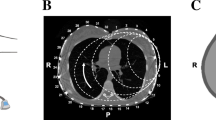
Each year 15 million babies are born prematurely and many suffer from respiratory failure due to immaturity of the lung and lack of control of breathing. Although respiratory support, especially mechanical ventilation, can improve their survival, it may also cause injury to the vulnerable lung resulting in severe and chronic pulmonary morbidity, lasting into adulthood. Heterogeneity of lung aeration, with areas of over inflation and collapse, plays a crucial part in the mortality and morbidity due to respiratory failure. This regionally different distribution of lung aeration cannot be detected by currently available bedside monitoring tools and imaging methods. Thus, an imaging technique for continuous non-invasive bedside monitoring of infants´ lung function is urgently needed. In order to address this, CRADL will use EIT technology to establish a tool for monitoring regional lung function and to guide interventions in the paediatric population.
Electrical impedance tomography (EIT) is a non-radiative, inexpensive technique that can facilitate real time monitoring of lung aeration, and recent studies have shown that it is effective in monitoring aeration in preterm babies. CRADL will show how EIT can provide new cost effective, easy to use, respiratory management tools and clinical protocols that can be universally adopted to reduce deaths and disability in preterm babies by delivering a clinical device that provides continuous, non-invasive, radiation free, bedside information on regional lung aeration and ventilation during daily clinical care of (preterm) infants and children with respiratory failure. CRADL will also assess the effectiveness, efficacy and safety of such a system in guiding respiratory management and supportive care of the most common causes of paediatric respiratory failure (respiratory distress syndrome, bronchiolitis and acute respiratory distress syndrome), with the final goal of reducing short and long term adverse effects of disease and side effects of its treatment.
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 668259. This reflects only the author’s views and that the Commission is not responsible for any use that may be made of the information it contains.
Chapter PDF
Similar content being viewed by others
Keywords
- Respiratory Failure
- Acute Respiratory Distress Syndrome
- Electrical Impedance Tomography
- Real Time Monitoring
- Regional Lung
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this paper
Cite this paper
Bayford, R. et al. (2016). Continuous Regional Analysis Device for Neonate Lung (CRADL). In: Kyriacou, E., Christofides, S., Pattichis, C. (eds) XIV Mediterranean Conference on Medical and Biological Engineering and Computing 2016. IFMBE Proceedings, vol 57. Springer, Cham. https://doi.org/10.1007/978-3-319-32703-7_35
Download citation
DOI: https://doi.org/10.1007/978-3-319-32703-7_35
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-32701-3
Online ISBN: 978-3-319-32703-7
eBook Packages: EngineeringEngineering (R0)