Abstract
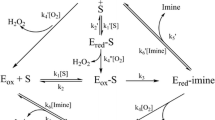
Through the study of mechanism-based inhibitors, it has been shown that the two-electron oxidation of biogenic amines carried out by the flavin moiety of monoamine oxidase occurs by stepwise electron transfer. This is illustrated by cyclopropylamine-based inhibitors. Mechanisms of propargylamine- and hydrazine-type inhibitors have also been clarified.
Enzymes react with substrates and inhibitors by forming either fleeting or stabilized adducts linked by bonds ranging from covalent to minimally attracting forces. Apoenzymes and/or cofactors may be involved in such reactions. Biochemists have approached the study of these interactions from an opportunistic point of view. An inorganic ion or a well-understood organic coenzyme will offer the most detailed chance of following the course of enzymic reactions. The macromolecular apoenzyme determines the course of interaction with substrate or inhibitor molecules of average size. Reactive portions of the participating amino acid side chains are thought to offer a handle in pinpointing reaction mechanisms while the backbone of the apoenzyme can be relegated to the background. Thiol groups of cysteine residues, serine and tyrosine phenolic hydroxyls, imino positions of indole and imidazole sections, the carboxyl of glutamic acid and amino and guanidino groups of lysine and arginine side chains have been called upon most frequently for this purpose.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
R. R. Rando: Annu. Repts. Med. Chem. 9, 234 (1974); Science 185, 320 (1974).
M. L. C. Hare: Biochem. J., 22, 968 (1928).
Monoamine Oxidase and its Inhibition. Ciba Foundation Symposium 39 (n.s.). Elsevier, New York 1976.
N. Yoshino, T. Obata, S. Sho and H. Kinemuchi: Jap. J. Pharmac. 35, 105 (1984).
Ref. 3, p. 173 seqq., 261 seqq., 328 seqq. and references contained therein.
K. N. Westlund, R. M. Denney, L. M. Kochersperger, R. M. Rose and C. W. Abell: Science 230, 181 (1985).
E. Uchida and G. B. Koelle: J. Histochem. Cytochem. 32, 667 (1984).
D. L. Murphy: Biochem. Pharmac. 27, 1889 (1978).
M. D. Houslay and K. F. Tipton: Biochem. J. 135, 173 (1973).
M. D. Houslay: J. Pharm. Pharmac. 29, 664 (1977).
M. D. Houslay: Biochem. Pharmac. 29, 3211 (1980).
D.M. Denney, N. T. Patel, R. R. Fritz and C. W. Abell: Molec. Pharmacol. 22, 500 (1982).
H.-Y. Yang and N. H. Neff: J. Pharmac. exp., Ther. 187, 365 (1973).
D. J. Dial and D. E. Clarke: Pharmac. Res. Commun. 11, 491 (1979).
O. Suzuki, H. Hattori, M. Oya, Y. Katsumata and T. Matsumoto: Life Sci. 25, 1843 (1979).
O. Suzuki, Y. Katsumata, M. Oya and T. Matsumoto: Biochem. Pharmac. 28, 953 (1979).
O. Suzuki, T. Matsumoto, Y. Katsumata and M. Oya: Experientia 36, 895 (1979).
C. J. Fowler and S. B. Ross: Med. Res. Rev. 4, 323 (1984).
D. R. Patek, H. Y. K. Chuang and L. Hellerman: Fed. Proc. 31, 420 (1972).
L. Hellerman, H. Y. K. Chuang and D. C. DeLuca: Adv. Biochem. Psycho- pharmacol. 5, 327 (1972).
L. A. Peterson, P. S. Caldera, A. Trevor, K. Chiba and N. Castagnoli, Jr.: J. Med. Chem. 28, 1432 (1985).
K. T. Yasunobu and B. Gomez: Meth, Enzym. 17B, 709 (1971).
K. T. Yasunobu: Biochem. biophys. Res. Commun. 52, 631 (1973).
K. T. Yasunobu and S. Oi: Adv. Biochem. Psychopharmacol. 5, 91 (1972).
M. D. Houslay and K. F. Tipton: Biochem. J. 135, 735 (1973).
R. B. Silverman, S. J. Hoffman and W. B. Catus (III): J. Am. Chem. Soc. 102, 7126 (1980).
T. P. Singer and E. S. G. Barron: J. Biol. Chem. 157, 241 (1945).
V. G. Erwin and L. Hellerman: J. Biol. Chem. 242, 4230 (1967).
Z. I. Akopyan, L. N. Stesina and V. Z. Gorkin: J. Biol. Chem. 246, 4610 (1971).
S. Oi, K. T. Yasunobu and J. Westley: Arch. Biochem. Biophys. 145, 557 (1971).
K. T. Yasunobu, K. Watanabe and H. Zeidan, in: Monoamine Oxidase: Structure, Functions, and Altered Functions, p.251-263. Eds. T. P. Singer, R. W. Von Korff. D. L. Murphy. Academic Press, New York 1979.
A. Tan, M. D. Glantz, L. H. Piette and K. T. Yasunobu: Biochem. biophys. Res. Commun. 117, 517 (1983).
J. P. Johnston: Biochem. Pharmac. 17, 1285 (1968).
J. Knoll and K. Magyar: Adv. Biochem. Psychopharmacol. 5, 393 (1972).
R. McCauley and E. Racker: Molec. Cell. Biochem. 1, 73 (1973).
M. Zrieka, I. A. McDonald, P. Bey and M. G. Palfreyman: J. Neurochem. 43, 448 (1984).
A.-L. Ask, K. Högberg, L. Schmidt, H. Kissling and S. B. Ross: Biochem. Pharmac. 31, 1401 (1982).
A.-L. Ask, W. Hellström, S. Norman, S.-O. Ögren and S. B. Ross: Neuropharmacology 21, 299 (1982).
C. Sahlberg, S. B. Ross, I. Fagervall, A.-L. Ask and A. Claesson: J. Med. Chem. 26, 1036 (1983).
V. Z. Gorkin and L. V. Tatyanenko: Life. Sei. 6, 791 (1967).
A. Burger and S. Nara: J. Med. Chem. 8, 859 (1965).
D. R. Patek and L. Hellerman: J. Biol. Chem. 249, 2373 (1974).
T. C. Bruice and Y. Yano: J. Am. Chem. Soc. 97, 5263 (1975).
A. L. Maycock, R. H. Abeles, J. I. Salach and T. P. Singer: Biochemistry 15, 114(1976).
E. A. Zeller, B. Gärtner and P. Hemmerich: Z. Naturforsch. (B) 27, 1050 (1972).
A. Krantz, B. Kokel, Y. P. Sachdeva, J. Salach, K. Detmer, A. Claesson and C. Sahlberg, in: Monoamine Oxidase: Structure, Function, and Altered Functions, p. 71. Eds. T. P. Singer, R. W. Von Korff, D. L. Murphy. Academic Press, New York, 1979.
J. Barsky, W. L. Pacha, S. Sarkar and E. A. Zeller: J. Biol. Chem. 234, 389 (1959).
E. A. Zeller and S. Sarkar: J. Biol. Chem. 237, 2333 (1962).
L. Hellerman and V. G. Erwin: J. Biol. Chem. 243, 5234 (1968).
B. M. Bloom: Ann. N.Y. Acad. Sei. 107, 878 (1963).
A. Burger and W. L. Yost: J. Am. Chem. Soc. 70, 2198 (1948).
C. Kaiser, A. Burger, L. Zirngibl, C. S. Davis and C. L. Zirkle: J. Org. Chem. 27, 768 (1962).
C. Kaiser, B. M. Lester, C. L. Zirkle, A. Burger, C. S. Davis, T. J. Delia and L. Zirngibl: J. Med. Pharm. Chem. 5, 1243 (1962).
J. Finkelstein, E. Chiang, F. M. Vane and J. Lee: J. Med. Chem. P, 319 (1966).
R. B. Silverman and P. A. Zieske: Biochemistry, 24, 2128 (1985).
C. Paech, J. L. Salach and T. P. Singer: J. Biol. Chem. 255, 2700 (1979).
R. B. Silverman: J. Biol. Chem. 258, 14766 (1983).
R. B. Silverman and S. J. Hoffman: Biochem. biophys. Res. Commun. 101, No. 4, 1396 (1981).
T. L. Macdonald: J. Am. Chem. Soc. 104, 2050 (1982).
R. P. Hanzlik and R. H. Tullman: J. Am. Chem. Soc. 104, 2048 (1982).
F. P. Guengerich, R. J. Willard, J. P. Shea, L. E. Richards and T. L. Macdonald: J. Am. Chem. Soc. 106, 6446 (1984).
C. L. Zirkle, C. Kaiser, D. H. Tedeschi, R. E. Tedeschi and A. Burger: J. Med. Pharm. Chem. 5, 1265 (1965).
R. B. Silverman and R. B. Yamasaki: Biochemistry 23, 1322 (1984).
R. B. Silverman, S. J. Hoffman and W. B. Catus (III): J. Am. Chem. Soc. 162, 7126 (1980).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1986 Birkhäuser Verlag Basel
About this chapter
Cite this chapter
Richards, L.E., Burger, A. (1986). Mechanism-based inhibitors of monoamine oxidase. In: Jucker, E. (eds) Progress in Drug Research / Fortschritte der Arzneimittelforschung / Progrès des recherches pharmaceutiques. Progress in Drug Research / Fortschritte der Arzneimittelforschung / Progrès des recherches pharmaceutiques, vol 30. Birkhäuser Basel. https://doi.org/10.1007/978-3-0348-9311-4_7
Download citation
DOI: https://doi.org/10.1007/978-3-0348-9311-4_7
Publisher Name: Birkhäuser Basel
Print ISBN: 978-3-0348-9990-1
Online ISBN: 978-3-0348-9311-4
eBook Packages: Springer Book Archive