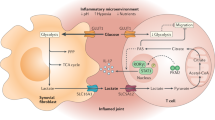
Abstract
The tumor microenvironment consists of complex and dynamic networks of cytokines, growth factors, and metabolic products. These contribute to significant alterations in tissue architecture, cell growth, immune cell phenotype, and function. Increased glycolytic flux is commonly observed in solid tumors and is associated with significant changes in metabolites, generating high levels of lactate. While elevated glycolytic flux is a characteristic metabolic adaption of tumor cells, glycolysis is also a key metabolic program utilized by a variety of inflammatory immune cells. As such lactate and the pH changes associated with lactate transport affect not only tumor cells but also immune cells. Here we provide an overview of lactate metabolic pathways and the effects lactate has on tumor growth and immune cell function. This knowledge provides opportunities for synergistic therapeutic approaches that combine metabolic drugs, which limit tumor growth and support immune cell function, together with immunotherapies to enhance tumor eradication.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Liotta LA, Kohn EC (2001) The microenvironment of the tumour–host interface. Nature 411:375–379
Balkwill FR, Capasso M, Hagemann T (2012) The tumor microenvironment at a glance. J Cell Sci 125:5591–5596
Hanahan D, Weinberg RA (2011) Hallmarks of cancer the next generation. Cell 144:646–674
Chen DS, Mellman I (2017) Elements of cancer immunity and the cancer–immune set point. Nature 541:321–330
Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 324:1029–1033
Pavlova NN, Thompson CB (2016) The emerging hallmarks of cancer metabolism. Cell Metab 23:27–47
Xie J et al (2015) Beyond Warburg effect – dual metabolic nature of cancer cells. Sci Rep 4:4927
Warburg O, Wind F, Negelein E (1927) The metabolism of tumors in the body. J Gen Physiol 8:519–530
Altman BJ, Stine ZE, Dang CV (2016) From Krebs to clinic: glutamine metabolism to cancer therapy. Nat Rev Cancer 16:619–634
Kamphorst JJ et al (2013) Hypoxic and Ras-transformed cells support growth by scavenging unsaturated fatty acids from lysophospholipids. Proc Natl Acad Sci 110:8882–8887
Boya P, Reggiori F, Codogno P (2013) Emerging regulation and functions of autophagy. Nat Cell Biol 15:713–720
Hanahan D, Coussens LM (2012) Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21:309–322
Goveia J et al (2016) Meta-analysis of clinical metabolic profiling studies in cancer: challenges and opportunities. EMBO Mol Med 8:1134–1142
Payen VL et al (2017) Monocarboxylate transporter MCT1 promotes tumor metastasis independently of its activity as a lactate transporter. Cancer Res 77:5591–5601
Carmona-Fontaine C et al (2017) Metabolic origins of spatial organization in the tumor microenvironment. Proc Natl Acad Sci 114:2934–2939
Brand A et al (2016) LDHA-associated lactic acid production blunts tumor immunosurveillance by T and NK cells. Cell Metab 24:657–671
Zhang J et al (2015) Prognostic value of pretreatment serum lactate dehydrogenase level in patients with solid tumors: a systematic review and meta-analysis. Sci Rep 5:9800
Walenta S et al (1997) Correlation of high lactate levels in head and neck tumors with incidence of metastasis. Am J Pathol 150:409–415
Schwickert G, Walenta S, Sundfør K, Rofstad EK, Mueller-Klieser W (1995) Correlation of high lactate levels in human cervical cancer with incidence of metastasis. Cancer Res 55:4757–4759
Sagman U et al (1991) The prognostic significance of pretreatment serum lactate dehydrogenase in patients with small-cell lung cancer. J Clin Oncol 9:954–961
Malhotra P, Sidhu LS, Singh SP (1986) Serum lactate dehydrogenase level in various malignancies. Neoplasma 33:641–647
Rizwan A et al (2013) Relationships between LDH-A, lactate, and metastases in 4T1 breast tumors. Clin Cancer Res 19:5158–5169
Dhup S, Dadhich RK, Porporato PE, Sonveaux P (2012) Multiple biological activities of lactic acid in cancer: influences on tumor growth, angiogenesis and metastasis. Curr Pharm Des 18:1319–1330
Hirschhaeuser F, Sattler UGA, Mueller-Klieser W (2011) Lactate: a metabolic key player in cancer. Cancer Res 71:6921–6925
Halestrap AP (2013) The SLC16 gene family - structure, role and regulation in health and disease. Mol Asp Med 34:337–349
Pinheiro C et al (2010) Expression of monocarboxylate transporters 1, 2, and 4 in human tumours and their association with CD147 and CD44. J Biomed Biotechnol 2010:1–7
Sonveaux P et al (2008) Targeting lactate-fueled respiration selectively kills hypoxic tumor cells in mice. J Clin Invest 118:3930–3942
Draoui N, Feron O (2011) Lactate shuttles at a glance: from physiological paradigms to anti-cancer treatments. Dis Model Mech 4:727–732
Roef MJ et al (2003) Gluconeogenesis in humans with induced hyperlactatemia during low-intensity exercise. Am J Physiol Endocrinol Metab 284:E1162–E1171
Gerich JE, Meyer C, Woerle HJ, Stumvoll M (2001) Renal gluconeogenesis: its importance in human glucose homeostasis. Diabetes Care 24:382–391
Cai T-Q et al (2008) Role of GPR81 in lactate-mediated reduction of adipose lipolysis. Biochem Biophys Res Commun 377:987–991
Ranganathan P et al (2018) GPR81, a cell-surface receptor for lactate, regulates intestinal homeostasis and protects mice from experimental colitis. J Immunol 200:1781–1789
Hoque R, Farooq A, Ghani A, Gorelick F, Mehal WZ (2014) Lactate reduces liver and pancreatic injury in toll-like receptor- and inflammasome-mediated inflammation via GPR81-mediated suppression of innate immunity. Gastroenterology 146:1763–1774
Seth RB, Sun L, Ea C-K, Chen ZJ (2005) Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-κB and IRF3. Cell 122:669–682
Zhang W et al (2019) Lactate is a natural suppressorof RLR signaling by targeting MAVS.Cell 178:176--189.e15
Hui S et al (2017) Glucose feeds the TCA cycle via circulating lactate. Nature 551:115–118
Whitaker-Menezes D et al (2011) Evidence for a stromal-epithelial “lactate shuttle” in human tumors. Cell Cycle 10:1772–1783
García-Cañaveras JC, Chen L, Rabinowitz JD (2019) The tumor metabolic microenvironment: lessons from lactate. Cancer Res 79:3155–3162
Romero-Garcia S, Moreno-Altamirano MMB, Prado-Garcia H, Sánchez-García FJ (2016) Lactate contribution to the tumor microenvironment: mechanisms, effects on immune cells and therapeutic relevance. Front Immunol 7:52
Wu H et al (2012) Central role of lactic acidosis in cancer cell resistance to glucose deprivation-induced cell death. J Pathol 227:189–199
Vegran F, Boidot R, Michiels C, Sonveaux P, Feron O (2011) Lactate influx through the endothelial cell monocarboxylate transporter MCT1 supports an NF- B/IL-8 pathway that drives tumor angiogenesis. Cancer Res 71:2550–2560
Sonveaux P et al (2012) Targeting the lactate transporter MCT1 in endothelial cells inhibits lactate-induced HIF-1 activation and tumor angiogenesis. PLoS One 7:e33418
Stern R, Shuster S, Neudecker BA, Formby B (2002) Lactate stimulates fibroblast expression of hyaluronan and CD44: the Warburg effect revisited. Exp Cell Res 276:24–31
Riedel A et al (2018) Tumor pre-conditioning ofdraining lymph node stroma by lactic acid. bioRxiv442137 https://doi.org/10.1101/442137
Newsholme P, Curi R, Gordon S, Newsholme EA (1986) Metabolism of glucose, glutamine, long-chain fatty acids and ketone bodies by murine macrophages. Biochem J 239:121–125
Alonso D, Nungester WJ (1956) Comparative study of host resistance of Guinea pigs and rats V. the effect of pneumococcal products on glycolysis and oxygen uptake by polymorphonuclear leucocytes J Infect Dis 99:174–181
Buck MD, O’Sullivan D, Pearce EL (2015) T cell metabolism drives immunity. J Exp Med 212:1345–1360
Assmann N et al (2017) Srebp-controlled glucose metabolism is essential for NK cell functional responses. Nat Immunol 18:1197–1206
MacIver NJ et al (2008) Glucose metabolism in lymphocytes is a regulated process with significant effects on immune cell function and survival. J Leukoc Biol 84:949–957
O’Neill LAJ, Kishton RJ, Rathmell J (2016) A guide to immunometabolism for immunologists. Nat Rev Immunol 16:553–565
Yaqoob P (2003) Fatty acids as gatekeepers of immune cell regulation. Trends Immunol 24:639–645
Pearce EL et al (2009) Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 460:103–107
Michalek RD et al (2011) Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4 + T cell subsets. J Immunol 186:3299–3303
Jha AK et al (2015) Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity 42:419–430
van der Windt GJW et al (2013) CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proc Natl Acad Sci USA 110:14336–14341
Mills EL et al (2018) Itaconate is an anti-inflammatory metabolite that activates Nrf2 via alkylation of KEAP1. Nature 556:113–117
Mills EL et al (2018) Accumulation of succinate controls activation of adipose tissue thermogenesis. Nature 560:102–106
Tannahill GM et al (2013) Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature 496:238–242
Ryan DG et al (2019) Coupling Krebs cycle metabolitesto signalling in immunity and cancer. NatMetab 11(1):16
Moon J-S et al (2015) mTORC1-induced HK1-dependent glycolysis regulates NLRP3 inflammasome activation. Cell Rep 12:102–115
Chang C-H et al (2013) Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153:1239–1251
Mills EL, Kelly B, O’Neill LAJ (2017) Mitochondria are the powerhouses of immunity. Nat Immunol 18:488–498
Murray PJ, Wynn TA (2011) Protective andpathogenic functions of macrophage subsets. NatRev Immunol 11:723--737
Colegio OR et al (2014) Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513:559–563
Goetze K, Walenta S, Ksiazkiewicz M, Kunz-Schughart LA, Mueller-Klieser W (2011) Lactate enhances motility of tumor cells and inhibits monocyte migration and cytokine release. Int J Oncol 39:453–463
Peter K, Rehli M, Singer K, Renner-Sattler K, Kreutz M (2015) Lactic acid delays the inflammatory response of human monocytes. Biochem Biophys Res Commun 457:412–418
Shime H et al (2008) Tumor-secreted lactic acid promotes IL-23/IL-17 proinflammatory pathway. J Immunol 180:7175–7183
Dietl K et al (2010) Lactic acid and acidification inhibit TNF secretion and glycolysis of human monocytes. J Immunol 184:1200–1209
Gottfried E et al (2006) Tumor-derived lactic acid modulates dendritic cell activation and antigen expression. Blood 107:2013–2021
Nasi A et al (2013) Dendritic cell reprogramming by endogenously produced lactic acid. J Immunol 191:3090–3099
Fischer K et al (2007) Inhibitory effect of tumor cell-derived lactic acid on human T cells. Blood 109:3812–3819
Pötzl J et al (2017) Reversal of tumor acidosis by systemic buffering reactivates NK cells to express IFN-γ and induces NK cell-dependent lymphoma control without other immunotherapies. Int J Cancer 140:2125–2133
Husain Z, Huang Y, Seth P, Sukhatme VP (2013) Tumor-derived lactate modifies antitumor immune response: effect on myeloid-derived suppressor cells and NK cells. J Immunol 191:1486–1495
Harmon C et al (2019) Lactate-mediated acidification of tumor microenvironment induces apoptosis of liver-resident NK cells in colorectal liver metastasis. Cancer Immunol Res 7:335–346
Chabner BA, Roberts TG (2005) Chemotherapy and the war on cancer. Nat Rev Cancer 5:65–72
Renner K et al (2017) Metabolic hallmarks of tumor and immune cells in the tumor microenvironment. Front Immunol 8:248
Tennant DA, Durán RV, Gottlieb E (2010) Targeting metabolic transformation for cancer therapy. Nat Rev Cancer 10:267–277
Galluzzi L, Kepp O, Vander Heiden MG, Kroemer G (2013) Metabolic targets for cancer therapy. Nat Rev Drug Discov 12:829–846
Cutolo M, Sulli A, Pizzorni C, Seriolo B, Straub RH (2001) Anti-inflammatory mechanisms of methotrexate in rheumatoid arthritis. Ann Rheum Dis 60:729–735
Doherty J, Cleveland J (2013) Targeting lactate metabolism for cancer therapeutics. J Clin Invest 123:3685–3692
Ganapathy-Kanniappan S, Geschwind J-FH (2013) Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer 12:152
Raez LE et al (2013) A phase I dose-escalation trial of 2-deoxy-d-glucose alone or combined with docetaxel in patients with advanced solid tumors. Cancer Chemother Pharmacol 71:523–530
Liberti MV et al (2017) A predictive model forselective targeting of the Warburg effect throughGAPDH inhibition with a natural product. CellMetab 26:648--659.e8
Noble RA et al (2017) Inhibition of monocarboxyate transporter 1 by AZD3965 as a novel therapeutic approach for diffuse large B-cell lymphoma and burkitt lymphoma. Haematologica 102:1247–1257
Manerba M et al (2012) Galloflavin (CAS 568-80-9): a novel inhibitor of lactate dehydrogenase. ChemMedChem 7:311–317
Le A et al (2010) Inhibition of lactate dehydrogenase a induces oxidative stress and inhibits tumor progression. Proc Natl Acad Sci 107:2037–2042
Critchlow SE et al (2019) Abstract 1207: Reversinglactate-driven immunosuppression using the novel,potent and selective MCT4 inhibitor AZD0095. In:Experimental and molecular therapeutics, pp 1207--1207
Ibrahim-Hashim A et al (2017) Tris-base buffer: a promising new inhibitor for cancer progression and metastasis. Cancer Med 6:1720–1729
Corbet C, Feron O (2017) Tumour acidosis: from the passenger to the driver’s seat. Nat Rev Cancer 17:577–593
Adeva-Andany MM, Fernández-Fernández C, Mouriño-Bayolo D, Castro-Quintela E, Domínguez-Montero A (2014) Sodium bicarbonate therapy in patients with metabolic acidosis. Sci World J 2014:627673
Chao M et al (2016) A nonrandomized cohort and a randomized study of local control of large hepatocarcinoma by targeting intratumoral lactic acidosis. Elife 5:e15691
Nazarewicz RR et al (2013) Does scavenging of mitochondrial superoxide attenuate cancer prosurvival signaling pathways? Antioxid Redox Signal 19:344–349
Porporato PE et al (2014) A mitochondrial switch promotes tumor metastasis. Cell Rep 8:754–766
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Harmon, C., O’Farrelly, C., Robinson, M.W. (2020). The Immune Consequences of Lactate in the Tumor Microenvironment. In: Birbrair, A. (eds) Tumor Microenvironment. Advances in Experimental Medicine and Biology, vol 1259. Springer, Cham. https://doi.org/10.1007/978-3-030-43093-1_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-43093-1_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-43092-4
Online ISBN: 978-3-030-43093-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)