Abstract
Carbon dioxide CO2 plays an important role in global warming because of the greenhouse effect.
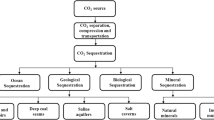
Different technologies have been studied for its capture and storage; an alternative pathway called “Mineral CO2 sequestration” is a process in which carbon dioxide CO2 reacts with materials having a high metal oxide composition to produce a chemically stable and insoluble metal carbonate. This work has the results of the reaction of mineral carbonation of natural JT material (CaSiO3). This last is synthesized starting from the calcination with 900 °C of the rough clay which is obtained from a tertiary formation located in Eastern Morocco. The carbonate of calcium (CaCO3) is formed after the submission of the sample to a flow of pure CO2 during 15 min under the normal conditions of the temperature and the pressure. It was assessed by diffraction of x-rays (DRX), electronic scan microscopy (MEB).
These techniques of analyzes physics-chemical validate the CO2 collection efficiency by our material.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
IPCC: Climate Change 2007: The Physical Science Basis. Summary for Policymakers. Contribution of working group, to the fourth assessment report (2007)
Ababou, A., Ajbary, M., Taleb, M., Kherbeche, A.: Direct mineral carbonation of new materials for CO2 sequestration. J. Mater. Environ. Sci. 8(9), 3106–3111 (2017)
Goff, F., Lackner, K.S.: Carbon dioxide sequestering using ultramafic rocks. Environ. Geosci. 5(3), 89–101 (1998)
Kerrick, D.M., Mckibben, M.A., Seward, T.M., Caldiera, K.: Convective hydrothermal CO2 emission from high heat flow regions. Chem. Geol. 121, 285–293 (1995)
Brady, P.V., Gislason, S.R.: Seafloor weathering controls on atmospheric CO2 and global climate. Geochimica et Cosmochimica Acta 61(5), 965–973 (1997)
Bryant, D.R., Zador, I., Landwehr, J.B., Wolfe, H.M.: Limited clinical utility of midtrimester fetal morphometric percentile rankings in screening for birth weight abnormalities. Am. J. Obstet. Gynecol. 177(4), 859–863 (1997)
Jepma, C.J., Munasinghe, M.: Climate Change Policy: Facts, Issues and Analyses. Cambridge University Press, Cambridge (1998)
White, C.M., Strazisar, B.R., Granite, E.J., Hoffman, J.S., Pennline, H.W.: Separation and capture of CO2 from large stationary sources and sequestration in geological formations - coalbeds and deep saline aquifers. J. Air Waste Manag. Assoc. 53, 645–715 (2003)
Santos, A., Ajbary, M., Toledo-Fernández, J.A., Morales-Flόrez, V., Kherbeche, A., Esquivias, L.: J. Sol. Gel. Sci. Technol. 48, 224 (2008)
Herzog, H.: Carbon Sequestration via Mineral Carbonation: Overview and Assessment 14 March 2002 (2002)
Santos, A., Ajbary, M., Kherbeche, A., Piñero, M., De la Rosa-Fox, N., Esquivias, L.: J. Sol-Gel Sci. Technol. (2008)
Bonenfant, D., Kharoune, L., Sauve, S., Hausler, R., Niquette, P., Mimeault, M., Kharoune, M.: Molecular analysis of carbon dioxide adsorption processes on steel slag oxides. Int. J. Greenhouse Gas Control 3, 20–28 (2008)
Baciocchi, R., Costa, G., Polettini, A., Pomi, R.: Influence of particle size on the carbonation of stainless steel slag for CO2 storage. Energy Proc. 1(1), 4859–4866 (2009)
Huijgen, W.J.J., Witkamp, G.-J., Comans, R.N.J.: CO2 mineral sequestration by steel slag carbonation. Environ. Sci. Technol. 39(24), 9676 (2005)
Huijgen, W.J.J., Comans, R.N.J.: Carbonation of steel slag for CO2 séquestration: leaching of products and réacfion mechanisms. Environ. Sci. Technol. 40(8), 2790 (2006)
Montes-Hemandez, G., Pérez-Lôpez, F., Renard, R., Nieto, J.M., Chariet, L.: Minerai séquestration of CO2 by aqueous carbonation of coal combustion fly-ash. J. Hazard. Mater. 161(2–3), 1347–1354 (2009). 30
Ababou, A., Ajbary, M., Taleb, M., Kherbeche, A.: CO2 sequestration on new materials. J. Mater. Environ. Sci. 6(9), 2367 (2015)
Jimoh, O.A., Otitoju, T.A., Hussin, H.: Understanding the precipitated calcium carbonate (PCC) production mechanism and its characteristics in the liquid-gas system using milk of line (MOL) suspension. South Afr. J. Chem. 70, 1–7 (2017)
Vaculikova, L., Plevova, E.: Identification of clay minerals and micas in sedimentary rocks. Acta Geodyn. Geomater. 2(2(138)), 167–175 (2005)
Feng, J., Guo, H., Wang, S., Zhao, Y., Ma, X.: Fabrication of multi-shelled hollow Mg- modified CaCO3 microspheres and their improved CO2 adsorption performance Chem. Eng. J. 321, 401–411 (2017)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this paper
Cite this paper
Ababou, A., Kharchafi, A., Taleb, M., Kherbeche, A. (2020). CO2 Carbonation on Alkaline Materials. In: Ezziyyani, M. (eds) Advanced Intelligent Systems for Sustainable Development (AI2SD’2019). AI2SD 2019. Advances in Intelligent Systems and Computing, vol 1104. Springer, Cham. https://doi.org/10.1007/978-3-030-36671-1_13
Download citation
DOI: https://doi.org/10.1007/978-3-030-36671-1_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-36670-4
Online ISBN: 978-3-030-36671-1
eBook Packages: Intelligent Technologies and RoboticsIntelligent Technologies and Robotics (R0)