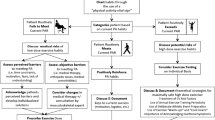
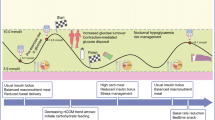
Abstract
Individuals with any type of diabetes can gain health benefits from regularly participating in physical activity that extend well beyond increasing insulin sensitivity and lowering blood glucose levels, and only minimal risks are present in some individuals. As various types of training can benefit cardiometabolic health, the exercise prescription for patients with diabetes should be tailored according to the timing of their medications, presence of diabetes complications, and individual fitness and glycemic management goals. A comprehensive exercise program performed consistently and progressively will help persons with diabetes better manage their disease, limit the impact of cardiovascular disease risk factors, improve their overall quality of life, and potentially limit premature mortality. In some cases, pre-exercise assessment and exercise testing may be warranted to ensure safe and effective participation. Athletes with diabetes may very well engage in competitive sports, although certain precautions are important. In athletes using insulin, fluid and food intake during exercise are important to consider, along with following an insulin regimen compensatory for the type, order, timing, and duration of physical activities to avoid hypoglycemia and hyperglycemia and negative long-term consequences.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Colberg SR SR, Yardley JE, Riddell MC, Dunstan DW, Dempsey PC, Horton ES, Castorino K, Tate DF. Physical activity/exercise and diabetes: A position statement of the American Diabetes Association. Diabetes Care. 2016;39(11).
Colberg SR, Sigal RJ, Fernhall B, Regensteiner JG, Blissmer BJ, Rubin RR, et al. Exercise and type 2 diabetes: the American College of Sports Medicine and the American Diabetes Association: joint position statement. Diabetes Care. 2010;33(12):e147–67.
Jakicic JM, Egan CM, Fabricatore AN, Gaussoin SA, Glasser SP, Hesson LA, et al. Four-year change in cardiorespiratory fitness and influence on glycemic control in adults with type 2 diabetes in a randomized trial: the look AHEAD trial. Diabetes Care. 2013;36(5):1297–303. https://doi.org/10.2337/dc12-0712. Epub 2012 Dec 6
Kokkinos P, Myers J, Nylen E, Panagiotakos DB, Manolis A, Pittaras A, et al. Exercise capacity and all-cause mortality in African American and Caucasian men with type 2 diabetes. Diabetes Care. 2009;32(4):623–8.
Pi-Sunyer X. The look AHEAD trial: a review and discussion of its outcomes. Curr Nutr Rep. 2014;3(4):387–91.
Newsom SA, Everett AC, Hinko A, Horowitz JF. A single session of low-intensity exercise is sufficient to enhance insulin sensitivity into the next day in obese adults. Diabetes Care. 2013;36(9):2516–22.
Yardley JE, Hay J, Abou-Setta AM, Marks SD, McGavock J. A systematic review and meta-analysis of exercise interventions in adults with type 1 diabetes. Diabetes Res Clin Pract. 2014;106(3):393–400.
Ratjen I, Weber KS, Roden M, Herrmann ME, Mussig K. Type 1 diabetes mellitus and exercise in competitive athletes. Exp Clin Endocrinol Diabetes. 2015;123(7):419–22. https://doi.org/10.1055/s-0035-1545344. Epub 2015 Apr 8
Riddell MC, Gallen IW, Smart CE, Taplin CE, Adolfsson P, Lumb AN, et al. Exercise management in type 1 diabetes: a consensus statement. Lancet Diabetes Endocrinol. 2017;5(5):377–90. https://doi.org/10.1016/S2213-8587(17)30014-1. Epub 2017 Jan 24
Look AHEAD Research Group. Eight-year weight losses with an intensive lifestyle intervention: the look AHEAD study. Obesity (Silver Spring). 2014;22(1):5–13.
Franz MJ, Boucher JL, Rutten-Ramos S, VanWormer JJ. Lifestyle weight-loss intervention outcomes in overweight and obese adults with type 2 diabetes: a systematic review and meta-analysis of randomized clinical trials. J Acad Nutr Diet. 2015;115(9):1447–63. doi: 10.016/j.jand.2015.02.031. Epub Apr 29.
Moholdt T, Lavie CJ, Nauman J. Sustained Physical Activity, Not Weight Loss, Associated With Improved Survival in Coronary Heart Disease. J Am Coll Cardiol. 2018;71(10):1094–101. doi: 10.16/j.jacc.2018.01.011.
Nilsson A, Bergens O, Kadi F. Physical activity alters inflammation in older adults by different intensity levels. Med Sci Sports Exerc. 2018;16(10):0000000000001582.
Fuller KNZ, Summers CM, Valentine RJ. Effect of a single bout of aerobic exercise on high-fat meal-induced inflammation. Metabolism. 2017;71:144–152.(doi):https://doi.org/10.1016/j.metabol.2017.03.001. Epub Mar 8.
Rohling M, Herder C, Stemper T, Mussig K. Influence of acute and chronic exercise on glucose uptake. J Diabetes Res. 2016;2016:2868652.
Dube JJ, Allison KF, Rousson V, Goodpaster BH, Amati F. Exercise dose and insulin sensitivity: relevance for diabetes prevention. Med Sci Sports Exerc. 2012;44(5):793–9.
Lieb DC, Parson HK, Mamikunian G, Vinik AI. Cardiac autonomic imbalance in newly diagnosed and established diabetes is associated with markers of adipose tissue inflammation. Exp Diabetes Res. 2012;2012:878760.
Maahs DM, Nadeau K, Snell-Bergeon JK, Schauer I, Bergman B, West NA, et al. Association of insulin sensitivity to lipids across the lifespan in people with type 1 diabetes. Diabet Med. 2011;28(2):148–55.
D'Hooge R, Hellinckx T, Van Laethem C, Stegen S, De Schepper J, Van Aken S, et al. Influence of combined aerobic and resistance training on metabolic control, cardiovascular fitness and quality of life in adolescents with type 1 diabetes: a randomized controlled trial. Clin Rehabil. 2011;25(4):349–59.
Tikkanen-Dolenc H, Waden J, Forsblom C, Harjutsalo V, Thorn LM, Saraheimo M, et al. Physical Activity Reduces Risk of Premature Mortality in Patients With Type 1 Diabetes With and Without Kidney Disease. Diabetes Care. 2017;16(10):dc17–0615.
Tikkanen-Dolenc H, Waden J, Forsblom C, Harjutsalo V, Thorn LM, Saraheimo M, et al. Frequent and intensive physical activity reduces risk of cardiovascular events in type 1 diabetes. Diabetologia. 2017;60(3):574–80. https://doi.org/10.1007/s00125-016-4189-8. Epub 2016 Dec 24
Lee JH, Lee R, Hwang MH, Hamilton MT, Park Y. The effects of exercise on vascular endothelial function in type 2 diabetes: a systematic review and meta-analysis. Diabetol Metab Syndr. 2018;10:15.(doi):https://doi.org/10.1186/s13098-018-0316-7. eCollection 2018.
Riebe D, Franklin BA, Thompson PD, Garber CE, Whitfield GP, Magal M, et al. Updating ACSM's recommendations for exercise Preparticipation health screening. Med Sci Sports Exerc. 2015;47(11):2473–9.
Young LH, Wackers FJ, Chyun DA, Davey JA, Barrett EJ, Taillefer R, et al. Cardiac outcomes after screening for asymptomatic coronary artery disease in patients with type 2 diabetes: the DIAD study: a randomized controlled trial. JAMA. 2009;301(15):1547–55.
Physical Activity Guidelines Advisory Committee. 2018 Physical Activity Guidelines Advisory Committee Scientific Report. In: US Department of Health and Human Services, ed. Washington, D.C. 2018.
Kemps H, Kränkel N, Dörr M, Moholdt T, Wilhelm M, Paneni F, et al. Exercise training for patients with type 2 diabetes and cardiovascular disease: What to pursue and how to do it. A Position Paper of the European Association of Preventive Cardiology (EAPC). European Journal of Preventive Cardiology.0(0):2047487318820420.
Snowling NJ, Hopkins WG. Effects of different modes of exercise training on glucose control and risk factors for complications in type 2 diabetic patients: a meta-analysis. Diabetes Care. 2006;29(11):2518–27.
Umpierre D, Ribeiro PA, Kramer CK, Leitao CB, Zucatti AT, Azevedo MJ, et al. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2011;305(17):1790–9.
Umpierre D, Ribeiro PA, Schaan BD, Ribeiro JP. Volume of supervised exercise training impacts glycaemic control in patients with type 2 diabetes: a systematic review with meta-regression analysis. Diabetologia. 2013;56(2):242–51.
Francois ME, Little JP. Effectiveness and safety of high-intensity interval training in patients with type 2 diabetes. Diabetes Spectr. 2015;28(1):39–44.
Gillen JB, Little JP, Punthakee Z, Tarnopolsky MA, Riddell MC, Gibala MJ. Acute high-intensity interval exercise reduces the postprandial glucose response and prevalence of hyperglycaemia in patients with type 2 diabetes. Diabetes Obes Metab. 2012;14(6):575–7.
Moser O, Tschakert G, Mueller A, Groeschl W, Pieber TR, Obermayer-Pietsch B, et al. Effects of high-intensity interval exercise versus moderate continuous exercise on glucose homeostasis and hormone response in patients with type 1 diabetes mellitus using novel ultra-long-acting insulin. PLoS One. 2015;10(8):e0136489.
Garcia-Garcia F, Kumareswaran K, Hovorka R, Hernando ME. Quantifying the acute changes in glucose with exercise in type 1 diabetes: a systematic review and meta-analysis. Sports Med. 2015;45(4):587–99.
Yang Z, Scott CA, Mao C, Tang J, Farmer AJ. Resistance exercise versus aerobic exercise for type 2 diabetes: a systematic review and meta-analysis. Sports Med. 2014;44(4):487–99.
Dunstan DW, Daly RM, Owen N, Jolley D, Vulikh E, Shaw J, et al. Home-based resistance training is not sufficient to maintain improved glycemic control following supervised training in older individuals with type 2 diabetes. Diabetes Care. 2005;28(1):3–9.
Dunstan DW, Daly RM, Owen N, Jolley D, De Courten M, Shaw J, et al. High-intensity resistance training improves glycemic control in older patients with type 2 diabetes. Diabetes Care. 2002;25(10):1729–36.
Egger A, Niederseer D, Diem G, Finkenzeller T, Ledl-Kurkowski E, Forstner R, et al. Different types of resistance training in type 2 diabetes mellitus: effects on glycaemic control, muscle mass and strength. Eur J Prev Cardiol. 2013;20(6):1051–60.
Zeppetzauer M, Drexel H, Vonbank A, Rein P, Aczel S, Saely CH. Eccentric endurance exercise economically improves metabolic and inflammatory risk factors. Eur J Prev Cardiol. 2013;20(4):577–84.
Church TS, Blair SN, Cocreham S, Johannsen N, Johnson W, Kramer K, et al. Effects of aerobic and resistance training on hemoglobin A1c levels in patients with type 2 diabetes: a randomized controlled trial. JAMA. 2010;304(20):2253–62.
Sigal RJ, Kenny GP, Boule NG, Wells GA, Prud'homme D, Fortier M, et al. Effects of aerobic training, resistance training, or both on glycemic control in type 2 diabetes: a randomized trial. Ann Intern Med. 2007;147(6):357–69.
Yang P, Swardfager W, Fernandes D, Laredo S, Tomlinson G, Oh PI, et al. Finding the Optimal volume and intensity of Resistance Training Exercise for Type 2 Diabetes: The FORTE Study, a Randomized Trial. Diabetes Res Clin Pract. 2017;130:98–107.(doi):https://doi.org/10.1016/j.diabres.2017.05.019.
Vinik AI, Vinik EJ, Colberg SR, Morrison S. Falls risk in older adults with type 2 diabetes. Clin Geriatr Med. 2015;31(1):89–99. viii
Morrison S, Simmons R, Colberg SR, Parson HK, Vinik AI. Supervised balance training and Wii fit-based exercises lower falls risk in older adults with type 2 diabetes. J Am Med Dir Assoc. 2017;27(17):004.
Morrison S, Colberg SR, Mariano M, Parson HK, Vinik AI. Balance training reduces falls risk in older individuals with type 2 diabetes. Diabetes Care. 2010;33(4):748–50.
Herriott MT, Colberg SR, Parson HK, Nunnold T, Vinik AI. Effects of 8 weeks of flexibility and resistance training in older adults with type 2 diabetes. Diabetes Care. 2004;27(12):2988–9.
Biswas A, Oh PI, Faulkner GE, Bajaj RR, Silver MA, Mitchell MS, et al. Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults: a systematic review and meta-analysis. Ann Intern Med. 2015;162(2):123–32.
Larsen RN, Dempsey PC, Dillon F, Grace M, Kingwell BA, Owen N, et al. Does the type of activity "break" from prolonged sitting differentially impact on postprandial blood glucose reductions? An exploratory analysis. Appl Physiol Nutr Metab. 2017;42(8):897–900. https://doi.org/10.1139/apnm-2016-0642. Epub 2017 Mar 24
Dempsey PC, Larsen RN, Sethi P, Sacre JW, Straznicky NE, Cohen ND, et al. Benefits for type 2 diabetes of interrupting prolonged sitting with brief bouts of light walking or simple resistance activities. Diabetes Care. 2016;39(6):964–72.
Garber CE, Blissmer B, Deschenes MR, Franklin BA, Lamonte MJ, Lee IM, et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43(7):1334–59.
Yardley JE, Colberg SR. Update on Management of Type 1 diabetes and type 2 diabetes in athletes. Curr Sports Med Rep. 2017;16(1):38–44.
Colberg SR, Sigal RJ. Prescribing exercise for individuals with type 2 diabetes: recommendations and precautions. Phys Sportsmed. 2011;39(2):13–26.
Karstoft K, Christensen CS, Pedersen BK, Solomon TP. The acute effects of interval- vs continuous-walking exercise on glycemic control in subjects with type 2 diabetes: a crossover, controlled study. J Clin Endocrinol Metab. 2014;99(9):3334–42.
Balducci S, Zanuso S, Cardelli P, Salvi L, Bazuro A, Pugliese L, et al. Effect of high- versus low-intensity supervised aerobic and resistance training on modifiable cardiovascular risk factors in type 2 diabetes; the Italian diabetes and exercise study (IDES). PLoS One. 2012;7(11):e49297.
Abate M, Schiavone C, Salini V, Andia I. Management of limited joint mobility in diabetic patients. Diabetes Metab Syndr Obes. 2013;6:197–207.
Yardley JE, Kenny GP, Perkins BA, Riddell MC, Balaa N, Malcolm J, et al. Resistance versus aerobic exercise: acute effects on glycemia in type 1 diabetes. Diabetes Care. 2013;36(3):537–42.
Yardley JE, Kenny GP, Perkins BA, Riddell MC, Malcolm J, Boulay P, et al. Effects of performing resistance exercise before versus after aerobic exercise on glycemia in type 1 diabetes. Diabetes Care. 2012;35(4):669–75.
Guelfi KJ, Ratnam N, Smythe GA, Jones TW, Fournier PA. Effect of intermittent high-intensity compared with continuous moderate exercise on glucose production and utilization in individuals with type 1 diabetes. Am J Physiol Endocrinol Metab. 2007;292(3):E865–70.
Sigal RJ, Fisher SJ, Halter JB, Vranic M, Marliss EB. Glucoregulation during and after intense exercise: effects of beta-adrenergic blockade in subjects with type 1 diabetes mellitus. J Clin Endocrinol Metab. 1999;84(11):3961–71.
Plockinger U, Topuz M, Riese B, Reuter T. Risk of exercise-induced hypoglycaemia in patients with type 2 diabetes on intensive insulin therapy: comparison of insulin glargine with NPH insulin as basal insulin supplement. Diabetes Res Clin Pract. 2008;81(3):290–5.
McMahon SK, Ferreira LD, Ratnam N, Davey RJ, Youngs LM, Davis EA, et al. Glucose requirements to maintain euglycemia after moderate-intensity afternoon exercise in adolescents with type 1 diabetes are increased in a biphasic manner. J Clin Endocrinol Metab. 2007;92(3):963–8.
Cryer PE. Hypoglycemia-associated autonomic failure in diabetes. Handb Clin Neurol. 2013;117:295–307.
Bremer JP, Jauch-Chara K, Hallschmid M, Schmid S, Schultes B. Hypoglycemia unawareness in older compared with middle-aged patients with type 2 diabetes. Diabetes Care. 2009;32(8):1513–7.
Chu L, Hamilton J, Riddell MC. Clinical management of the physically active patient with type 1 diabetes. Phys Sportsmed. 2011;39(2):64–77.
Poirier P, Tremblay A, Catellier C, Tancrede G, Garneau C, Nadeau A. Impact of time interval from the last meal on glucose response to exercise in subjects with type 2 diabetes. J Clin Endocrinol Metab. 2000;85(8):2860–4.
Wackers FJ, Young LH, Inzucchi SE, Chyun DA, Davey JA, Barrett EJ, et al. Detection of silent myocardial ischemia in asymptomatic diabetic subjects: the DIAD study. Diabetes Care. 2004;27(8):1954–61.
Colberg SR, Swain DP, Vinik AI. Use of heart rate reserve and rating of perceived exertion to prescribe exercise intensity in diabetic autonomic neuropathy. Diabetes Care. 2003;26(4):986–90.
Riddell MC, Zaharieva DP, Yavelberg L, Cinar A, Jamnik VK. Exercise and the Development of the Artificial Pancreas: One of the More Difficult Series of Hurdles. J Diabetes Sci Technol. 2015;9(6):1217–26. doi: 10.177/1932296815609370.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Case Study Wrap-Up
Case Study Wrap-Up
Case Vignette: A 54-year-old man presents for an assessment of his physical fitness. Two years ago, he experienced a non-ST-elevation myocardial infarction of the anterior wall, which necessitated placement of a drug-eluting stent in his left descending artery. He also reports that his GP told him that he has “elevated blood sugar levels” for which he received lifestyle advice. His family history is unremarkable for cardiovascular diseases and he reports no angina or other cardiovascular disorders. The patient smokes one pack of cigarettes per day (and has done so for 30 years). As for physical activity, he reports engaging in 30–45 min of jogging once a week. His present medications include 100 mg of acetylsalicylic acid, a beta-blocker (bisoprolol 5 mg 1–0-0), an angiotensin converting enzyme (ACE)-inhibitor (ramipril 2.5 mg 1–0-0), and a statin (simvastatin 20 mg 0–0-1).
On physical examination, his body mass index (BMI, 31.1 kg/m2), waist circumference (105 cm), and blood pressure (145/95 mmHg) are elevated. Auscultation of heart and lungs reveal no pathologic findings and no signs of heart failure are noted. The ECG reveals a sinus rhythm with 65 beats per minute, normal indices, and no Q-waves. A maximal exercise stress test reveals a reduced exercise capacity (75% of age predicted, or 151 Watts), but it is negative for ischemia and other abnormalities.
Blood tests show a fasting plasma glucose of 6.6 mmol/l (119 mg/dl), HbA1c of 6.9%, and dyslipidemia despite use of a statin: total cholesterol, 7.0 mmol/l (270 mg/dl); low density lipoprotein (LDL)-cholesterol, 4.0 mmol/L (155 mg/dl); high density lipoprotein (HDL)-cholesterol, 2.2 mmol/l (85 mg/dl); and triglycerides, 1.7 mmol/l (148 mg/dl). Urinary analysis shows some microalbuminuria and glucosuria.
1.1 Questions
-
1.
In addition to optimization of his medications (change of statin, upped titration of ramipril, and addition of antidiabetic medications) and smoking cessation, what exercise advice would you give this patient?
-
2.
Would you recommend blood glucose monitoring before, during and after exercise? What precisely do you recommend?
-
3.
Would you perform a graded exercise stress test on this patient prior to his starting a more formal exercise program?
-
4.
During routine exercise stress testing conducted on this patient, he becomes symptomatic with fatigue, dizziness, and a dry mouth. What is the most likely cause of his symptoms?
1.2 Answers
-
1.
All adults with diabetes are recommended to undertake at least 150 min per week of moderate to vigorous aerobic exercise training, although up to 300 min likely bestows additional health benefits. Given his underlying CVD (including a prior myocardial infarction and stent placement), his recommended intensity should be light to moderate, at least to start. He may choose to continue jogging (at a moderate pace) and include other types of aerobic training spread throughout the week, allowing no more than 2 days to lapse between bouts of activity. In addition, he should add in moderate resistance training exercises at least twice weekly (on nonconsecutive days) to enhance retention of his muscle mass. Engaging in flexibility exercises and balance training at least 2–3 days per week should also be recommended to someone his age (see Table 38.1), along with staying more active overall on a daily basis and breaking up sedentary time with frequent bouts of activity.
At least initially, this patient’s blood glucose should be monitored closely before, during and after exercise. Depending on which diabetes medications this patient is prescribed, monitoring blood glucose levels may remain important around exercise given that beta-blocker therapy can mask the onset of hypoglycemia. Done before and after exercise, such monitoring may also provide positive feedback regarding the glycemic benefits of regular exercise training, which may result in greater subsequent long-term adherence to his exercise training prescription. This is particularly important since exercise is a cornerstone for the management of all types of diabetes and its potential health complications.
-
2.
Although routine monitoring around exercise is not recommended for all adults with type 2 diabetes (given that most have a low risk of developing exercise-related hypoglycemia), when such patients begin an exercise program, their blood glucose responses to exercise should be checked. Monitoring and recording blood glucose levels before and after exercise, at least initially, is important because it may:
-
Allow for early detection and prevention of hypoglycemia or hyperglycemia
-
Help determine appropriate pre-exercise blood glucose levels to lower risk of glycemic imbalances resulting from activities
-
Identify patients who may benefit from continued monitoring around exercise
-
Provide information for modifying prescribed exercise based on glycemic responses
-
Allow for better adjustment of diabetes regimens to manage all activities
-
Motivate patients to remain more active to better manage their diabetes
-
-
3.
Likely yes, but it depends. Since this patient is already somewhat active, a stress test is only advisable if he plans to start new activities more vigorous than his current daily ones, which include weekly jogging (which he has been undertaking without any problems or symptoms arising). As stated, previously sedentary older individuals (≥ 40 years) with diabetes and anyone ≥30 years of age with a high cardiovascular risk should likely undergo medical screening and obtain medical clearance prior to performing vigorous intensity exercise. In addition, this patient does meet several criteria for a possible pre-participation exercise stress test, including his age, cigarette smoking, dyslipidemia, and known coronary artery disease (see section on “General Indications for Exercise Stress Testing in Patients with Diabetes”).
-
4.
Routine pre-session blood glucose measurement is 11.2 mmol/l (200 mg/dl), which compares well to his previous pre-session measurements. He starts to train on a cycle ergometer. After 10 min of ergometer training his heart rate starts to rise, although his workload is constantly decreasing from 75 to 45 W. The patient also reports feeling dizziness and a dry mouth. No ECG changes are evident, however.
His immediate blood glucose is 19.4 mmol/l (350 mg/dl), indicating an increase in hyperglycemia in this patient. Some of his symptoms correspond to possible symptoms of hyperglycemia, which can include fatigue, hyperventilation, and dry mouth, along with polyphagia, polydipsia, polyuria, blurred vision, weight loss, poor wound healing, dry or itchy skin, impotence (male), recurrent infections such as vaginal yeast infections, groin rash, external ear infections (swimmer’s ear), cardiac arrhythmias, stupor, and coma. Moreover, physical exhaustion causes glucose production in the liver (glycogenesis and glycogenolysis) plus enhanced free fatty acid release by adipose tissue and reduced muscle uptake of glucose and, consequently, may contribute to hyperglycemia in exercising patients with diabetes.
After a few minutes of rest, the patient’s symptoms resolve, but his blood glucose remains elevated at 19.4 mmol/l (342 mg/dl) 15 min later. A urinary analysis reveals moderate ketone bodies. When asked further, the patient reports having had a demanding week at his job and furthermore some domestic problems, resulting in significant insomnia during the prior week. To reduce his mental stress, he had exercised on a cycle ergometer for 2 h before this scheduled testing. He is instructed not to perform any exercise for the rest of the day and to continue to monitor his blood glucose levels frequently. The next day the patient’s urine is rechecked and found to be free of ketones. His blood glucose is 9.6 mmol/l (174 mg/dl) and he reports feeling completely recovered after a day of rest. He reports that his GP has recommended that he start on diabetes medications immediately to lower his blood glucose levels and that he plans to do so.
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Colberg, S.R., Niederseer, D. (2020). Impact of Exercise on Cardiovascular Risk Factors: Diabetes Mellitus. In: Pressler, A., Niebauer, J. (eds) Textbook of Sports and Exercise Cardiology. Springer, Cham. https://doi.org/10.1007/978-3-030-35374-2_38
Download citation
DOI: https://doi.org/10.1007/978-3-030-35374-2_38
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-35373-5
Online ISBN: 978-3-030-35374-2
eBook Packages: MedicineMedicine (R0)