Abstract
This paper focuses on statutory regulation of learning machines that qualify as medical devices. After a brief case study, the article takes a procedural perspective and presents the main features of the European regulatory framework that applies to medical devices in order to identify the regulatory peculiarities in the use of machine learning. In this context, the Chapter will analyse the immanent risks of machine learning applications as medical devices as well as the role of machine learning in their regulation. The overall finding is that due to its lack of expertise and material equipment the state activates private companies for market access control, which are commissioned with the preventive inspection of medical devices. As a result, security measures adopted by the authority are in principle limited to the period after market-entry. This leads to a structural information deficit for the authority, which has no systematic information about the products on the market. The authority is limited to a challenging overall market observation. This raises the question addressed in the fifth part of the paper: does the law guarantee sufficient instruments for the systematic transfer of knowledge from the risk actors to the authority about the potential risk of medical devices and does this in fact remedy the information deficit of the authority and ensure an effective post market-entry control of learning machines as medical devices?
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Notes
- 1.
De Fauw et al. (2018), p. 1342 et seq.
- 2.
See Wischmeyer, paras 2 et seq.
- 3.
On the reliability of an output of a learning algorithm in the context of search algorithms, see European Court of Justice C-131/12 ‘Google Spain SL/AEPD’ (13 May 2014) margin no. 61 et seq.
- 4.
See in detail Molnár-Gábor, paras 38 et seq.
- 5.
On the term of ‘machine learning’ see Hoffmann-Riem, para 3 and Hermstüwer, para 6.
- 6.
- 7.
Cf. Hadank (2017), p. 811.
- 8.
- 9.
Trute (2018), p. 1.
- 10.
- 11.
- 12.
- 13.
Cf. Council Directive 93/42/EEC, former Article 100a EEC.
- 14.
- 15.
Part of the European Medical Devices Law is also the Council Directive 98/79/EC concerning in vitro diagnostic medical devices, which shouldn’t be considered in this contribution.
- 16.
Cf. Recital 8 of the Council Directive 93/42/EEC.
- 17.
Cf. Recital 15 of the Council Directive 93/42/EEC.
- 18.
- 19.
For further details see below, paras 29 et seq., which describes the control-related process modules.
- 20.
Bieback (2008), p. 214.
- 21.
Cf. Article 16(3) of the Council Directive 93/42/EEC; implemented in detail in national law by Section 15 MPG.
- 22.
Gesetz über Medizinprodukte (Medizinproduktegesetz – MPG) in the version published on 07th August 2002 (BGBl. I p. 3146), last amended by Article 7 of the Act of the 18th July 2017 (BGBl. I p. 2757).
- 23.
For an overview of the historical development of national Medical Devices Law, in particular the implementation of the European directives by the MPG amendments, see Tomasini (2015), pp. 19 et seq.
- 24.
An overview is provided by Webel (2018), p. 852, margin no. 3.
- 25.
Regulation (EU) 2017/745 of the European Parliament and of the Council of 5th April 2017 on medical devices, amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009 and repealing Council Directives 90/385/EEC and 93/42/EEC, OJ EU 2017, No. L 117/1.
- 26.
Cf. Recitals 1, 13 and Annex IX section 4.5. of the MDR.
- 27.
Cf. Article 123(2) of the MDR.
- 28.
Tomasini (2015), p. 18.
- 29.
Irmer (2013), p. 145.
- 30.
The legal qualification of software in the medical scope was simplified by the 4th amendment of the MPG in 2010, cf. Gesetz zur Änderung medizinprodukterechtlicher Vorschriften vom 29. Juli 2009 (BGBl. I p. 2326); Irmer (2013), p. 145.
- 31.
Cf. Section 3 No. 1 MPG, adopted literally from Article 2 of the Council Directive 2007/47/EC; Tomasini (2015), p. 37.
- 32.
Cf. Bill of the Rederal Government, Parliamentary Printing Matter 16/12258, p. 26.
- 33.
See paras 22 et seq., in which the specific risk construction of learning machines is elaborated as a basis for their classification.
- 34.
- 35.
- 36.
Gesetz über den Verkehr mit Arzneimitteln (Arzneimittelgesetz—AMG) in the version published on 12th December 2005 (BGBl. I p. 3394), last amended by Article 1 of the Act of the 18th July 2017 (BGBl. I p. 2757).
- 37.
Lücker (2014), pp. 1272 et seq., margin no. 7.
- 38.
- 39.
MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 4.
- 40.
Cf. Section 3 No. 1 MPG.
- 41.
Irmer (2013), p. 145.
- 42.
Ibid.; Prinz (2017), p. 19.
- 43.
Merten (2004), p. 1212.
- 44.
MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 18.
- 45.
Sachs (2013), p. 31.
- 46.
- 47.
- 48.
Oen (2009), p. 55.
- 49.
- 50.
- 51.
Klümper and Vollebregt (2009), p. 100.
- 52.
Tomasini (2015), p. 44.
- 53.
MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 7; Tomasini (2015), p. 39.
- 54.
Cf. Gesetz zur Änderung medizinprodukterechtlicher Vorschriften vom 29. Juli 2009 (BGBl. I p. 2326).
- 55.
MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 7; Pramann and Albrecht (2015), p. 132.
- 56.
Tomasini (2015), p. 40; MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 8.
- 57.
MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 8.
- 58.
Irmer (2013), p. 145.
- 59.
Cf. Recital 20 of the Council Directive 2007/47/EC.
- 60.
Cf. Bill of the Federal Government, Parliamentary Printing Matter 16/12258, p. 26.
- 61.
Other voices in the literature argue that software cannot be an accessory since the law on the amendment of medical device regulations (4th MPG amendment) came into force. This difference of views has been processed by Tomasini (2015), pp. 46 et seq.; also illustrated by Oen (2009), p. 56; Gassner (2016), p. 111.
- 62.
Cf. Article 1(2)(b) of the Council Directive 93/42/EEC; misleadingly implemented in Section 3 No. 9 MPG.
- 63.
Anhalt et al. (2017), p. 58.
- 64.
- 65.
To this see Tomasini (2015), p. 45.
- 66.
MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 12; thereafter Tomasini (2015), p. 48.
- 67.
Cf. Article 1(1) of the Council Directive 93/42/EEC.
- 68.
Anhalt et al. (2017), p. 57.
- 69.
Cf. Article 1(1) of the Council Directive 93/42/EEC and the implementation in Section 2(1) MPG.
- 70.
Tomasini (2015), p. 49.
- 71.
Cf. Recital 19 of the Regulation (EU) 2017/745.
- 72.
European Court of Justice C-329/16 ‘Philips France/ Ministre des Affaires sociales et de la Santé’ (7 December 2017), margin no. 36.
- 73.
Ibid.
- 74.
MEDDEV 2.1/6 (2016), Guidance document concerning the qualification and classification of stand-alone software, p. 17 et seq.
- 75.
Anhalt et al. (2017), p. 61.
- 76.
MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 4; Braun and Püschel (2014), p. 136.
- 77.
- 78.
Cf. Article 3 of the Council Directive 93/42/EEC; Braun and Püschel (2014), p. 136.
- 79.
Tomasini (2015), p. 86; for further details on the basic requirements see para 30.
- 80.
Cf. Annex IX of the Council Directive 93/42/EEC.
- 81.
Section 13(1) MPG.
- 82.
Each medical device is classified separately, this also applies to accessories on medical devices, cf. Annex IX, section 2.2 of the Council Directive 93/42/EEC.
- 83.
Anhalt et al. (2017), p. 61.
- 84.
Cf. Annex IX of the Council Directive 93/42/EEC; MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 7.
- 85.
MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 4.
- 86.
If the manufacturer is unsure how to classify his product, he can consult a notified body. In case of a dispute between the manufacturer and the notified body resulting from the application of the classification rules, the matter shall be referred for decision to the relevant Competent Authority to which the notified body is subject, cf. Article 9(2) of the Council Directive 93/42/EEC.
- 87.
Cf. Annex IX, section 2.5 of the Council Directive 93/42/EEC; MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 13.
- 88.
Anhalt et al. (2017), p. 61.
- 89.
MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 37.
- 90.
Anhalt et al. (2017), p. 61; MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 39.
- 91.
Anhalt et al. (2017), p. 61.
- 92.
Ibid.; MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 40.
- 93.
Anhalt et al. (2017), p. 61; MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 47.
- 94.
Anhalt et al. (2017), p. 61.
- 95.
MEDDEV 2.4/1 Rev 9 (2010), Guidance document concerning the classification of medical devices, p. 37.
- 96.
MEDDEV 2.4/1 Rev. 9 (2010), Guidance document concerning the classification of medical devices, p. 38.
- 97.
The BfArM has published a statistical evaluation of the risk reports on software errors in medical devices that were conclusively evaluated between 01st January 2005 and 31th December 2016. Available at: https://www.bfarm.de/SharedDocs/Downloads/DE/Service/Statistik/MP-Statistik/statist-Auswert_Fehlerart_Software.jpg?__blob=poster&v=6.
- 98.
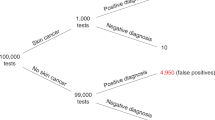
For a description of instances for false negative and false positive errors see Hermstüwer, para 45.
- 99.
Trute (2018), p. 1.
- 100.
- 101.
See in detail Molnár-Gábor, paras 25–28.
- 102.
To the right not to know see Taupitz (1998), p. 591.
- 103.
To risk information in context of informed consent of patients see Molnár-Gábor, paras 34 et seq.
- 104.
Van Cauter (1988), p. E786.
- 105.
Herberger (2018), p. 2827.
- 106.
Ibid.
- 107.
Herberger (2018), pp. 2827 et seq.
- 108.
Everything else would be unsatisfactory in the medical field, so Herberger (2018), p. 2828. On Explainable AI see Rademacher, para 33, Wischmeyer paras 27 et seq.
- 109.
For illustrative examples of qualification and classification of software used in the healthcare environment, see Annex 1 of MEDDEV 2.1/6 (2016), Guidance document concerning the Qualification and Classification of stand alone software, pp. 19 et seq.
- 110.
Scherzberg and Heym (2013), p. 176.
- 111.
Annex IX of the MDD is also referred to by national law in Section 13(1) sentence 2 MPG.
- 112.
Cf. Annex IX, section 2.3 of the Council Directive 93/42/EEC; also Annex VIII section 3.3 of the Regulation (EU) 2017/745; illustratively Tomasini (2015), p. 98.
- 113.
Frankenberger (2017), p. 127.
- 114.
Cf. Annex VIII section 3.3 of the Regulation (EU) 2017/745; Graf (2017a), p. 59.
- 115.
Cf. Annex IX, section 1.4 of the Council Directive 93/42/EEC; also Article 2 of the Regulation (EU) 2017/745.
- 116.
Namely rules 9 to 12 of Annex IX, section 3 of the Council Directive 93/42/EEC.
- 117.
See paras 22 et seq.
- 118.
In accordance with rule 12.
- 119.
For further details on the Essential Requirements see para 30.
- 120.
Cf. Annex XIII of the Council Directive 93/42/EEC; Gassner points out the danger of overregulation, cf. Gassner (2016), p. 112.
- 121.
Cf. Rule 11 in Annex VIII section 6.3 of the Council Regulation (EU) 2017/745.
- 122.
The rule covers for instance hospital beds and patient lifts, cf. Frankenberger (2017), p. 139.
- 123.
See paras 1 et seq.
- 124.
Section 7 MPG refers to Annex I of the MDD.
- 125.
- 126.
Kage (2005), p. 248.
- 127.
- 128.
Further details on the basic requirements in this para.
- 129.
- 130.
Pilniok (2017), p. 12.
- 131.
Ibid.
- 132.
Cf. Annex I, section 17 of the Council Regulation (EU) 2017/745; categorized by Prinz (2017), p. 26.
- 133.
For an overview of the selectable modules depending on the risk class of the device, see Article 11 of the Council Directive 93/42/EEC with reference to the relevant Annexes to the Directive.
- 134.
Scherzberg and Heym (2013), p. 184.
- 135.
- 136.
Cf. Article 11(10) of the Council Directive 93/42/EEC; also Article 53(4) of the Council Regulation (EU) 2017/745.
- 137.
- 138.
Kage (2005), p. 246.
- 139.
Voßkuhle (2002), p. 331.
- 140.
See Scherzberg and Heym (2013), p. 185.
- 141.
- 142.
Scherzberg and Heym (2013), p. 185.
- 143.
Merten (2005), p. 90.
- 144.
See Martini (2017b), p. 453. In Germany, ‘TÜV’ is the abbreviation for ‘Technischer Überwachungsverein’ and refers to associations that carry out safety checks as a technical testing organization. These are often safety inspections prescribed by law and carried out on a private basis as indirect state administration. The best-known of these is the main inspection for motor vehicles, which is also colloquially called ‘TÜV’.
- 145.
Merten (2005), p. 109; see also Hoffmann-Riem, para 39, on the idea of a public body specifically tasked with monitoring AI applications.
- 146.
Merten (2005), p. 90.
- 147.
- 148.
Kage (2005), p. 248.
- 149.
Cf. exemplarily Section 15(2) sentence 4 and Section 18(3) No. 2 MPG.
- 150.
Cf. Section 25(1), Section 26(2) sentence 2 and 4 and Section 28(1) MPG.
- 151.
The European legislator maintains this instrument even when the new Regulation on Medical Devices is enacted, cf. Article 45 of the Council Regulation (EU) 2017/745.
- 152.
Merten (2005), p. 110.
- 153.
Cf. Section 26(2) sentence 2 MPG.
- 154.
Merten (2005), p. 111.
- 155.
Ibid.
- 156.
See also below, paras 37 et seq. for further remarks.
- 157.
Merten (2004), p. 1211.
- 158.
Kage (2005), p. 248.
- 159.
Ibid.
- 160.
- 161.
- 162.
For further details see Wischmeyer, paras 12 et seq.
- 163.
See also above para 1.
- 164.
Cf. Section 15(2) sentence 4 MPG, which can be systematically assigned to the official monitoring of the activities of the notified bodies, but which, with regard to the range of tasks of the notified bodies, includes conformity assessment of medical devices in compliance with the law.
- 165.
Cf. Section 26(2) sentence 4, (3) MPG.
- 166.
Cf. Section 26(4) sentence 1 MPG.
- 167.
If such a network of system premises exists, an efficient post market-entry control could be conceivable by a continuous, automatic data transfer of the changed premises to the supervising authority. A comparable situation is already known in the field of remote emission monitoring, cf. German (Federal) Constitutional Court 7 C 47/95 (13 February 1997). Solved from questions of the administrative burden and data protection concerns in the health-relevant field, however, considerable constitutional concerns would have to be included in the consideration of a transferability of the model, which would not only affect the manufacturer’s right to exercise an occupation, but also the user’s right to informational self-determination.
- 168.
- 169.
Explanation of the rapporteur’s draft of 14th May 2013, 2012/0266(COD), PE510.741v03-00, p. 120; see also Hoffmann-Riem, para 14.
- 170.
Braun and Püschel (2014), p. 139.
- 171.
Cf. Recital 41 of the Council Regulation (EU) 2017/745; Graf (2017b), p. 318.
- 172.
Cf. Article 10(7), Article 27(1) of the Council Regulation (EU) 2017/745.
- 173.
Cf. Article 2 No. 15 and Annex VI Part C section 1 of the Council Regulation (EU) 2017/745.
- 174.
Cf. Article 28(1) of the Council Regulation (EU) 2017/745.
- 175.
Cf. Article 14(2), Article 29(1) of the Council Regulation (EU) 2017/745.
- 176.
Cf. Article 27(7) of the Council Regulation (EU) 2017/745.
- 177.
Cf. Article 92(1) of the Council Regulation (EU) 2017/745.
- 178.
Graf (2017b), p. 318.
- 179.
Cf. Annex VI Part C section 6.5.2 of the Council Regulation (EU) 2017/745.
- 180.
Cf. Annex VI Part C section 6.5.3 of the Council Regulation (EU) 2017/745.
- 181.
Cf. Annex VI Part. C section 5.2 and 5.8 of the Council Regulation (EU) 2017/745.
- 182.
For further details on the inspection competences see above para 35.
- 183.
Cf. Article 93(3)(b) of the Council Regulation (EU) 2017/745; Petersen (2018), p. 105.
References
Anhalt E, Dieners P, Westphal V (2017) Einführung in die Grundlagen und die Systematik des deutschen Medizinprodukterechts. In: Anhalt E, Dieners P (eds) Medizinprodukterecht. Praxishandbuch, 2nd edn. C.H. Beck, München, pp 45–69
Bieback K (2008) Zertifizierung und Akkreditierung. Das Zusammenwirken staatlicher und nichtstaatlicher Akteure in gestuften Prüfsystemen. Nomos Verlagsgesellschaft, Baden-Baden
Braun J, Püschel C (2014) Medizinprodukterecht und Produktzulassung – Was bringt die Zukunft? In: Schwegel P, Da-Cruz P, Hemel U et al (eds) Medizinprodukte Management. P.C.O.-Verlag, Bayreuth, pp 136–147
Cauter E (1988) Estimating false-positive and false-negative errors in analyses of hormonal pulsatility. Am J Physiol 254:E786–E794
Colquhoun D (2017) The reproducibility of research and the misinterpretation of p-values. http://rsos.royalsocietypublishing.org/content/4/12/171085.full.pdf
Courtin E (1997) Software in Medizinprodukten. Medizinprodukte-Journal:62–68
Czettritz P, Strelow T (2017) ‘Beam me up, Scotty’ – die Klassifizierung von Medical Apps als Medizinprodukte. Pharmarecht:433–435
Di Fabio U (1994) Das Arzneimittelrecht als Repräsentant der Risikoverwaltung. Die Verwaltung 27:345–360
Di Fabio U (1996) Produktharmonisierung durch Normung und Selbstüberwachung. Carl Heymanns Verlag KG, Köln
Dieners P, Vivekens S (2017) Europarechtliche Rahmenbedingungen. In: Anhalt E, Dieners P (eds) Medizinprodukterecht. Praxishandbuch, 2nd edn. C.H. Beck, München, pp 1–43
Düwert H, Klümper M (2013) Gesundheits-Apps: Ein neuer Trend und seine rechtlichen Herausforderungen. Medizinprodukte-Journal:23–30
Ernst C (2017) Algorithmische Entscheidungsfindung und personenbezogene Daten. JuristenZeitung 2017:1026–1036
Fauw J, Ledsam J, Romera-Paredes B et al (2018) Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat Med 24:1342–1350. https://doi.org/10.1038/s41591-018-0107-6
Frankenberger H (2017) Klassifizierung von Medizinprodukten. In: Anhalt E, Dieners P (eds) Medizinprodukterecht. Praxishandbuch, 2nd edn. C.H. Beck, München, pp 111–160
Gassner U (2016) Software als Medizinprodukt – zwischen Regulierung und Selbstregulierung. Medizin Produkte Recht 2016:109–115
Gassner U (2017) Die neue Medizinprodukteverordnung. Aktueller Text mit Einführung. Bundesanzeiger Verlag GmbH, Köln
Graf A (2017a) Revision des europäischen Rechtsrahmens für Medizinprodukte. Einfluss auf die Klassifizierung von Medizinprodukten. Pharmarecht:57–61
Graf A (2017b) Revision des europarechtlichen Rechtsrahmens für Medizinprodukte. Neue Vorgaben zu Vigilanz und Marktbeobachtung und die Eudamed-Datenbank als zentrales Regulierungsinstrument. Pharmarecht:317–322
Hadank B (2017) App statt Arzt? Entscheidungsfindung durch Algorithmen im Gesundheitswesen – Bericht zur Tagung ‘Gesundheitsentscheidung durch Algorithmen - rechtliche Rahmenbedingungen der Digitalisierung des Gesundheitswesens’, 6.9.2017 in Berlin. GesundheitsRecht:811–813
Heil M (2014) Zulassung für Medizinprodukte? Die ENVI-Änderungsvorschläge. In: Gassner U (ed) Reform des EU-Medizinprodukterechts – Stand und Perspektiven. Shaker Verlag, Aachen, pp 15–23
Herberger M (2018) ‘Künstliche Intelligenz’ und Recht. Ein Orientierungsversuch. Neue Juristische Wochenschrift 71:2825–2829
Hötzel G (2018) ‘There is an app for that’ – Aspekte des Rechts der Medizinprodukte und des ärztlichen Berufs- und Vergütungsrechts. Zeitschrift für das gesamte Medizin- und Gesundheitsrecht:16–20
Igl G (2018) § 27 Medizinprodukte (Überblick). In: Igl G, Welti F (eds) Gesundheitsrecht. Eine systematische Einführung, 3rd edn. Verlag Franz Vahlen, München, pp 155–156
Irmer F (2013) Medizinprodukterechtliche Einordnung von Software. Updates & Upgrades. Medizin Produkte Recht:145–148
Jäkel C (2016) Europarechtliche Aspekte der Zulassung von Medizinprodukten. Medizinrecht:601–602
Jaumann A (1980) Die Regelung technischer Sachverhalte. In: Lukes R (ed) Rechtliche Ordnung der Technik als Aufgabe der Industriegesellschaft. Heymann Verlag, Köln, pp 5–22
Kage U (2005) Das Medizinproduktegesetz. Staatliche Risikosteuerung unter dem Einfluss europäischer Harmonisierung. Springer-Verlag, Berlin
Klümper M, Vollebregt E (2009) Die Regulierung von Software bei Medizinprodukten. Die geänderten Anforderungen für die CE-Kennzeichnung und Konformitätsbewertung auf Grund der Richtlinie 2007/47/EG. Medizinprodukte-Journal 16:99–105
Langkafel P (2015) Auf dem Weg zum Dr. Algorithmus? Potenziale von Big Data in der Medizin. Aus Politik und Zeitgeschichte 65:27–32
Laufs A (2001) Arzneimittelprüfung. Neue Juristische Wochenschrift 54:3381–3382
Lücker V (2014) Kommentierung zum Gesetz über Medizinprodukte (Medizinproduktegesetz - MPG). In: Spickhoff A (ed) Beck’scher Kurzkommentar Medizinrecht, 2nd edn. C.H. Beck, München, pp 1260–1354
Martini M (2017a) Algorithmen als Herausforderung für die Rechtsordnung. JuristenZeitung 72:1017–1025
Martini M (2017b) Transformation der Verwaltung durch Digitalisierung. Die Öffentliche Verwaltung:443–455
Merten J (2004) Benannte Stellen: Private Vollzugsinstanzen eines Europäischen Verwaltungsrechts. Deutsches Verwaltungsblatt:1211–1216
Merten J (2005) Private Entscheidungsträger und Europäisierung der Verwaltungsrechtsdogmatik. Zur Einbindung benannter Stellen in den Vollzug des Medizinprodukterechts. Dunker & Humblot GmbH, Berlin
Meyer S (2011) Risikovorsorge als Eingriff in das Recht auf körperliche Unversehrtheit. Gesetzliche Erschwerung medizinischer Forschung aus Sicht des Patienten als Grundrechtsträger. Archiv des öffentlichen Rechts 136:428–478
Neubauer G, Uilaky R (2004) Bedeutung von Innovationen für die Medizinprodukteindustrie. In: Oberender P, Schommer R, Da-Cruz P (eds) Zukunftsorientiertes Management in der Medizinprodukteindustrie. P.C.O.-Verlag, Bayreuth, pp 149–161
Oen R (2009) Software als Medizinprodukt. Medizin Produkte Recht 2009:55–57
Ostermayer K, Kexel W (2009) Software – Ein Medizinprodukt mit hohem Risikopotenzial. Medizinprodukte-Journal:106–107
Petersen N (2018) Marktüberwachung bei Medizinprodukten. Was ändert sich für die nationalen Behörden nach der MDR? Medizinprodukte-Journal:102–107
Pilniok A (2017) Zertifizierung und Akkreditierung als Regulierungsstrategie im Wirtschaftsverwaltungsrecht. In: Krüper J (ed) Zertifizierung und Akkreditierung als Instrumente qualitativer Glücksspielregulierung. Mohr Siebeck, Tübingen, pp 1–33
Pramann O, Albrecht U (2015) Medizinische Software. Im regulatorischen Umfeld des Medizinprodukterechts am Beispiel von Medical Apps. Zeitschrift zum Innovations- und Technikrecht:132–137
Prinz T (2017) Entwicklung und Herstellung medizinischer Software. Normen in der Medizintechnik. VDE Verlag, Berlin
Röhl HC (2000) Akkreditierung und Zertifizierung im Produktsicherheitsrecht. Zur Entwicklung einer neuen Europäischen Verwaltungsstruktur. Springer, Berlin
Sachs G (2013) Software in Systemen und Problembehandlungseinheiten. In: Gassner U (ed) Software als Medizinprodukt – IT vs. Medizintechnik? Shaker Verlag, Aachen, pp 31–36
Scherzberg A (2002) Wissen, Nichtwissen und Ungewissheit im Recht. In: Engel C, Halfmann J, Schulte M (eds) Wissen – Nichtwissen – Unsicheres Wissen. Nomos, Baden-Baden, pp 113–144
Scherzberg A, Heym S (2013) Neue medizintechnische Möglichkeiten als Herausforderung an das Recht. In: Peter C, Funcke D (eds) Wissen an der Grenze. Zum Umgang mit Ungewissheit und Unsicherheit in der modernen Medizin. Campus, Frankfurt a. M., pp 161–207
Schmitt J (2014) Aktuelle und zukünftige Marktentwicklung im Bereich der Medizinprodukteindustrie. In: Schwegel P, Da-Cruz P, Hemel U et al (eds) Medizinprodukte Management. P.C.O.-Verlag, Bayreuth, pp 26–35
Singh M (2014) Produktemanagement in der Medizintechnik. In: Schwegel P, Da-Cruz P, Hemel U et al (eds) Medizinprodukte Management. P.C.O.-Verlag, Bayreuth, pp 148–161
Stiemerling O (2015) ‘Künstliche Intelligenz’. Automatisierung geistiger Arbeit, Big Data und das Internet der Dinge. Computer und Recht:762–765
Stoll P (2008) Wissensarbeit als staatliche Aufgabe. Wissen als Leitbegriff für Reformüberlegungen. In: Döhmann I, Collin P (eds) Generierung und Transfer staatlichen Wissens im System des Verwaltungsrechts. Mohr Siebeck, Tübingen, pp 34–49
Taupitz J (1998) Das Recht auf Nichtwissen. In: Hanau P, Lorenz E, Matthes H (eds) Festschrift für Günther Wiese zum 70. Geburtstag. Luchterhand, Neuwied, pp 583–602
Thuemmler C (2017) The Case for Health 4.0. In: Thuemmler C, Bai C (eds) Health 4.0. How virtualization and big data are revolutionizing healthcare. Springer, Cham, pp 1–22
Tomasini R (2015) Standalone-Software als Medizinprodukt. Shaker Verlag, Aachen
Trute H (2018) Der Stellenwert von Real-World-Evidenz im deutschen Sozial- und Gesundheitsrecht. https://doi.org/10.1007/s00772-018-0471-z
Voßkuhle A (2002) Strukturen und Bauformen ausgewählter neuer Verfahren. In: Hoffmann-Riem W, Schmidt-Aßmann E (eds) Verwaltungsverfahren und Verwaltungsverfahrensgesetz. Nomos, Baden-Baden, pp 277–347
Webel D (2018) Kommentierung zum Gesetz über Medizinprodukte (Medizinproduktegesetz – MPG). In: Bergmann K, Pauge B, Steinmeyer H (eds) Nomoskommentar Gesamtes Medizinrecht, 3rd edn. Nomos, Baden-Baden
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Jabri, S. (2020). Artificial Intelligence and Healthcare: Products and Procedures. In: Wischmeyer, T., Rademacher, T. (eds) Regulating Artificial Intelligence. Springer, Cham. https://doi.org/10.1007/978-3-030-32361-5_14
Download citation
DOI: https://doi.org/10.1007/978-3-030-32361-5_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-32360-8
Online ISBN: 978-3-030-32361-5
eBook Packages: Law and CriminologyLaw and Criminology (R0)