Abstract
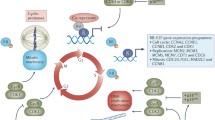
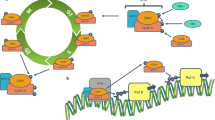
Eukaryotic organisms depend on the cell cycle for their survival though a cyclical biochemical process consisting of tightly controlled, enzymatically driven reactions that result in cell division and the generation of new cells. The cell cycle is regulated by kinases such as cyclin-dependent kinases (CDKs), and non-CDKs, which include the Aurora and Polo-like kinases, as well as checkpoint proteins and mitotic kinesins. The CDK family of serine-theronine kinases is a common target for genetic or epigenetic events, resulting in the amplification or overexpression of these kinases in a myriad of tumor types. Such findings make CDKs rational and attractive targets for cancer therapeutics as their inhibition could potentially result in preferential targeting of malignant cells. However, several cyclin-CDK complexes have been found to be dispensable for cell proliferation owing to functional redundancy, promiscuity, and compensatory mechanisms. Although these issues have hampered their progress into the clinic, several novel compounds are currently in various phases of clinical trial development. This chapter will introduce the role of CDKs in facilitating the cell cycle, their aberrations in malignant progression and pharmacological strategies targeting them.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Nurse P. Genetic control of cell size at cell division in yeast. Nature 1975; 256:547–551.
Masui Y, Markert CL. Cytoplasmic control of nuclear behavior during meiotic maturation of frog oocytes. J Exp Zool 1971; 177:129–145.
Smith LD Ecker RE. The interaction of steroids with Rana pipiens Oocytes in the induction of maturation. Dev Biol 1971;25:232–247.
Lohka MJ, Masui Y. (1983) Formation in vitro of sperm pronuclei and mitotic chromosomes induced by amphibian ooplasmic components. Science 1983;220:719–721.
Sausville EA. Complexities in the development of cyclin-dependent kinase inhibitor drugs. Trends Mol Med 2002;8:S32–37.
Bai C, Richman R, Elledge SJ. Human cyclin F. Embo J 1994; 13:6087–6098.
Kong M, Barnes EA, Ollendorff V, Donoghue DJ. Cyclin F regulates the nuclear localization of cyclin B1 through a cyclin-cyclin interaction. Embo J 2000;19:1378–1388.
Berthet C, Aleem E, Coppola V, Tessarollo L, Kaldis P. Cdk2 knockout mice are viable. Curr Biol 2003;13:1775–1785.
Meyerson M, Enders GH, Wu CL et al. A family of human cdc2-related protein kinases. Embo J, 1992;11:2909–2917.
Lundberg AS, Weinberg RA. Functional inactivation of the retinoblastoma protein requires sequential modification by at least two distinct cyclin-cdk complexes. Mol Cell Biol 1998;18:753–761.
Harbour JW, Luo RX, Dei Santi A, Postigo AA, Dean DC. (1999) Cdk phosphorylation triggers sequential intramolecular interactions that progressively block Rb functions as cells move through G1. Cell 1999;98:859–869.
Sherr CJ. G1 phase progression: cycling on cue. Cell, 1994;79, 551–555.
Pines J. Cyclins: wheels within wheels. Cell Growth Differ, 1991;2, 305–310.
Schwartz GK, Shah MA. Targeting the cell cycle: a new approach to cancer therapy. J Clin Oncol, 2005;23,9408–9421.
Meinhart A, Kamenski T, Hoeppner S, Baumli S, Cramer P. A structural perspective of CTD function. Genes Dev, 2005;19,1401–1415.
Prelich G. RNA polymerase II carboxy-terminal domain kinases: emerging clues to their function. Eukaryot Cell, 2002;1:153–162.
Harper JW, Elledge SJ. The role of Cdk7 in CAK function, a retro-retrospective. Genes Dev, 1998;12:285–289.
Sherr CJ, Roberts JM. Inhibitors of mammalian G1 cyclin-dependent kinases. Genes Dev, 1995;9:1149–1163.
Coleman TR, Dunphy WG. Cdc2 regulatory factors. Curr Opin Cell Biol, 1994;6:877–882.
Collins VP. Gene amplification in human gliomas. Glia, 1995;5:289–296.
Reifenberger G, Reifenberger J, Ichimura K, Meltzer PS, Collins VP. Amplification of multiple genes from chromosomal region 12q13–14 in human malignant gliomas: preliminary mapping of the amplicons shows preferential involvement of CDK4, SAS, and MDM2. Cancer Res, 1994;54:4299–4303.
An HX, Beckmann MW, Reifenberger G, Bender HG, Niederacher D. Gene amplification and overexpression of CDK4 in sporadic breast carcinomas is associated with high tumor cell proliferation. Am J Pathol, 1999;154:113–118.
Wikman H, Nymark P, Vayrynen A et al. CDK4 is a probable target gene in a novel amplicon at 12q13.3-q14.1 in lung cancer. Genes Chromosomes Cancer, 2005;42:193–199.
Forus A, Larramendy ML, Meza-Zepeda LA et al. Dedifferentiation of a well-differentiated liposarcoma to a highly malignant metastatic osteosarcoma: amplification of 12q14 at all stages and gain of 1q22-q24 associated with metastases. Cancer Genet Cytogenet, 2001; 125:100–111.
Khatib ZA, Matsushime H, Valentine M, Shapiro DN, Sherr CJ, Look AT. Coamplification of the CDK4 gene with MDM2 and GLI in human sarcomas. Cancer Res, 1993;53:5535–5541.
Yu Q, Geng Y, Sicinski P. Specific protection against breast cancers by cyclin D1 ablation. Nature, 2001;411:1017–1021.
Yang C, Ionescu-Tiba V, Burns K et al. The role of the cyclin D1-dependent kinases in ErbB2-mediated breast cancer. Am J Pathol, 2004;164:1031–1038.
Landis MW, Pawlyk BS, Li T, Sicinski P, Hinds PW. Cyclin D1-dependent kinase activity in murine development and mammary tumorigenesis. Cancer Cell, 2006;9:13–22.
Yu Q, Sicinska E, Geng Y et al. Requirement for CDK4 kinase function in breast cancer. Cancer Cell, 2006;9:23–32.
Yamamoto H, Monden T, Miyoshi H et al. Cdk2/cdc2 expression in colon carcinogenesis and effects of cdk2/cdc2 inhibitor in colon cancer cells. Int J Oncol, 1998;13:233–239.
Dobashi Y, Shoji M, Jiang SX, Kobayashi M, Kawakubo Y, Kameya T. Active cyclin A-CDK2 complex, a possible critical factor for cell proli-feration in human primary lung carcinomas. Am J Pathol, 1998; 153:963–972.
Ortega S, Prieto I, Odajima J et al. Cyclin-dependent kinase 2 is essential for meiosis but not for mitotic cell division in mice. Nat Genet, 2003;35: 25–31.
Tetsu O, McCormick F. Proliferation of cancer cells despite CDK2 inhibition. Cancer Cell, 2003,3:233–245.
Senderowicz AM. Small-molecule cyclin-dependent kinase modulators. Oncogene, 2003;22:6609–6620.
Senderowicz AM, Sausville EA. Preclinical and clinical development of cyclin-dependent kinase modulators. J Natl Cancer Inst, 2000; 92:376–387.
Carlson B, Lahusen T, Singh S et al. Down-regulation of cyclin D1 by transcriptional repression in MCF-7 human breast carcinoma cells induced by flavopiridol. Cancer Res, 1999;59:4634–4641.
Patel V, Senderowicz AM, Pinto D, Jr. et al. Flavopiridol, a novel cyclin-dependent kinase inhibitor, suppresses the growth of head and neck squamous cell carcinomas by inducing apoptosis. J Clin Invest, 1998;102:1674–1681.
Senderowicz AM, Headlee D, Stinson SF et al. Phase I trial of continuous infusion flavopiridol, a novel cyclin-dependent kinase inhibitor, in patients with refractory neoplasms. J Clin Oncol, 1998;16:2986–2999.
Thomas JP, Tutsch KD, Cleary JF et al. Phase I clinical and pharmacokinetic trial of the cyclin-dependent kinase inhibitor flavopiridol. Cancer Chemother Pharmacol, 2002;50:465–472.
Tan AR, Headlee D, Messmann R et al. Phase I clinical and pharmacokinetic study of flavopiridol administered as a daily 1-hour infusion in patients with advanced neoplasms. J Clin Oncol, 2002; 20:4074–4082.
Arguello F, Alexander M, Sterry JA et al. Flavopiridol induces apoptosis of normal lymphoid cells, causes immunosuppression, and has potent antitumor activity In vivo against human leukemia and lymphoma xenografts. Blood, 1998;91:2482–2490.
Sasaki Y, Sasaki T, Minami H et al. A phase I pharmacokinetic (PK)- pharmacodynamic (PD) study of flavopiridol by 24 hours continuous infusion (CI) repeating every week. Proc Am Soc Clin Oncol 21:2002 (abstr 371).
Stadler WM, Vogelzang NJ, Amato R et al. Flavopiridol, a novel cyclin-dependent kinase inhibitor, in metastatic renal cancer: a University of Chicago Phase II Consortium study. J Clin Oncol, 2000; 18:371–375.
Schwartz GK, Ilson D, Saltz L et al. Phase II study of the cyclin-dependent kinase inhibitor flavopiridol administered to patients with advanced gastric carcinoma. J Clin Oncol, 2001;19:1985–1992.
Shapiro GI, Supko JG, Patterson A et al. A phase II trial of the cyclin-dependent kinase inhibitor flavopiridol in patients with previously untreated stage IV non-small cell lung cancer. Clin Cancer Res, 2001;7:1590–1599.
Lin TS, Howard OM, Neuberg DS, Kim HH, Shipp MA. Seventy-two hour continuous infusion flavopiridol in relapsed and refractory mantle cell lymphoma. Leuk Lymphoma, 2002;43:793–797.
Aklilu M, Kindler HL, Donehower RC, Mani S, Vokes EE. Phase II study of flavopiridol in patients with advanced colorectal cancer. Ann Oncol, 2003;14:1270–1273.
Liu G, Gandara DR, Lara PN, Jr. et al. A Phase II trial of flavopiridol (NSC #649890) in patients with previously untreated metastatic androgen-independent prostate cancer. Clin Cancer Res, 2004;10:924–928.
Flinn IW, Byrd JC, Bartlett N et al. Flavopiridol administered as a 24-hour continuous infusion in chronic lymphocytic leukemia lacks clinical activity. Leuk Res, 2005;29:1253–1257.
Byrd JC, Peterson BL, Gabrilove J et al. Treatment of relapsed chronic lymphocytic leukemia by 72-hour continuous infusion or 1-hour bolus infusion of flavopiridol: results from Cancer and Leukemia Group B study 19805. Clin Cancer Res, 2005;11:4176–4181.
Kouroukis CT, Belch A, Crump M et al. Flavopiridol in untreated or relapsed mantle-cell lymphoma: results of a phase II study of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol, 2003;21:1740–1745.
Burdette-Radoux S, Tozer RG, Lohmann RC et al. Phase II trial of flavopiridol, a cyclin dependent kinase inhibitor, in untreated metastatic malignant melanoma. Invest New Drugs, 2004;22:315–322.
Grendys EC, Jr., Blessing JA, Burger R, Hoffman J. A phase II evaluation of flavopiridol as second-line chemotherapy of endometrial carcinoma: a Gynecologic Oncology Group study. Gynecol Oncol, 2005;98,249–253.
Van Veldhuizen PJ, Faulkner JR, Lara PN, Jr. et al. A phase II study of flavopiridol in patients with advanced renal cell carcinoma: results of Southwest Oncology Group Trial 0109. Cancer Chemother Pharmacol, 2005;56:39–45.
Dispenzieri A, Gertz MA, Lacy MQ et al. Flavopiridol in patients with relapsed or refractory multiple myeloma: a phase 2 trial with clinical and pharmacodynamic end-points. Haematologica, 2006;91:390–393.
Matranga CB, Shapiro GI. Selective sensitization of transformed cells to flavopiridol-induced apoptosis following recruitment to S-phase. Cancer Res, 2002;62:1707–1717.
Motwani M, Delohery TM, Schwartz GK. Sequential dependent enhancement of caspase activation and apoptosis by flavopiridol on paclitaxel-treated human gastric and breast cancer cells. Clin Cancer Res, 1999;5:1876–1883.
Motwani M, Rizzo C, Sirotnak F, She Y, Schwartz GK. Flavopiridol enhances the effect of docetaxel in vitro and in vivo in human gastric cancer cells. Mol Cancer Ther, 2003;2:549–555.
Jung CP, Motwani MV, Schwartz GK. Flavopiridol increases sensitization to gemcitabine in human gastrointestinal cancer cell lines and correlates with down-regulation of ribonucleotide reductase M2 subunit. Clin Cancer Res, 2001;7:2527–2536.
Schwartz GK, O’Reilly E, Ilson D et al. Phase I study of the cyclin-dependent kinase inhibitor flavopiridol in combination with paclitaxel in patients with advanced solid tumors. J Clin Oncol, 2002;20:2157–2170.
Tan AR, Yang X, Berman A et al. Phase I trial of the cyclin-dependent kinase inhibitor flavopiridol in combination with docetaxel in patients with metastatic breast cancer. Clin Cancer Res, 2004;10:5038–5047.
Shah MA, Kortmansky J, Motwani M et al. A phase I clinical trial of the sequential combination of irinotecan followed by flavopiridol. Clin Cancer Res, 2005;11:3836–3845.
Bible KC, Lensing JL, Nelson SA et al. Phase 1 trial of flavopiridol combined with cisplatin or carboplatin in patients with advanced malignancies with the assessment of pharmacokinetic and pharmacodynamic end points. Clin Cancer Res, 2005;11:5935–5941.
Karp JE, Passaniti A, Gojo I et al. Phase I and pharmacokinetic study of flavopiridol followed by 1-beta-D-arabinofuranosylcytosine and mitoxantrone in relapsed and refractory adult acute leukemias. Clin Cancer Res, 2005;11:8403–8412.
Wang Q, Fan S, Eastman A, Worland PJ, Sausville EA, O’Connor PM. UCN-01: a potent abrogator of G2 checkpoint function in cancer cells with disrupted p53. J Natl Cancer Inst, 1996;88: 956–965.
Busby EC, Leistritz DF, Abraham RT, Karnitz LM, Sarkaria JN. The radiosensitizing agent 7-hydroxystaurosporine (UCN-01) inhibits the DNA damage checkpoint kinase hChk1. Cancer Res, 2000;60:2108–2112.
Yu Q, La Rose J, Zhang H, Takemura H, Kohn KW, Pommier Y. UCN-01 inhibits p53 up-regulation and abrogates gamma-radiation-induced G(2)-M checkpoint independently of p53 by targeting both of the checkpoint kinases, Chk2 and Chk1. Cancer Res, 2002;62:5743–5748.
Sato S, Fujita N, Tsuruo T. Interference with PDK1-Akt survival signaling pathway by UCN-01 (7-hydroxystaurosporine). Oncogene, 2002;21:1727–1738.
Hotte SJ, Oza A, Winquist EW et al. Phase I trial of UCN-01 in combination with topotecan in patients with advanced solid cancers: a Princess Margaret Hospital Phase II Consortium study. Ann Oncol, 2006;17:334–340.
Sausville EA, Arbuck SG, Messmann R et al. Phase I trial of 72-hour continuous infusion UCN-01 in patients with refractory neoplasms. J Clin Oncol, 2001;19:2319–2333.
Tamura T, Sasaki Y, Minami H et al. Phase I study of UCN-01 by 3-hour infusion. 1999, ASCO Annual Meeting (Abstract 611).
Dees EC, Baker SD, O’Reilly S et al. A phase I and pharmacokinetic study of short infusions of UCN-01 in patients with refractory solid tumors. Clin Cancer Res, 2005;11:664–671.
Hsueh CT, Kelsen D, Schwartz GK. UCN-01 suppresses thymidylate synthase gene expression and enhances 5-fluorouracil-induced apoptosis in a sequence-dependent manner. Clin Cancer Res, 1998;4:2201–2206.
Kortmansky J, Shah MA, Kaubisch A et al. Phase I trial of the cyclin-dependent kinase inhibitor and protein kinase C inhibitor 7-hydroxy-staurosporine in combination with Fluorouracil in patients with advanced solid tumors. J Clin Oncol, 2005;23:1875–1884.
Pollack IF, Kawecki S, Lazo JS. Blocking of glioma proliferation in vitro and in vivo and potentiating the effects of BCNU and cisplatin: UCN-01, a selective protein kinase C inhibitor. J Neurosurg, 1996;84:1024–1032.
Lara PN, Jr., Mack PC, Synold T et al. The cyclin-dependent kinase inhibitor UCN-01 plus cisplatin in advanced solid tumors: a California cancer consortium phase I pharmacokinetic and molecular correlative trial. Clin Cancer Res, 2005;11:4444–4450.
Rini BI, Weinberg V, Shaw V et al. Time to disease progression to evaluate a novel protein kinase C inhibitor, UCN-01, in renal cell carcinoma. Cancer, 2004;101:90–95.
Ozawa Y, Sugi NH, Nagasu T et al. E7070, a novel sulphonamide agent with potent antitumour activity in vitro and in vivo. Eur J Cancer, 2001;37:2275–2282.
Punt CJ, Fumoleau P, van de Walle B, Faber MN, Ravic M, Campone M. Phase I and pharmacokinetic study of E7070, a novel sulfonamide, given at a daily times five schedule in patients with solid tumors. A study by the EORTC-early clinical studies group (ECSG). Ann Oncol, 2001;12:1289–1293.
Raymond E, ten Bokkel Huinink WW, Taieb J et al. Phase I and pharmacokinetic study of E7070, a novel chloroindolyl sulfonamide cell-cycle inhibitor, administered as a one-hour infusion every three weeks in patients with advanced cancer. J Clin Oncol, 2002;20: 3508–3521.
Terret C, Zanetta S, Roche H et al. Phase I clinical and pharmacokinetic study of E7070, a novel sulfonamide given as a 5-day continuous infusion repeated every 3 weeks in patients with solid tumours. A study by the EORTC Early Clinical Study Group (ECSG). Eur J Cancer, 2003;39:1097–1104.
Dittrich C, Dumez H, Calvert H et al. Phase I and pharmacokinetic study of E7070, a chloroindolyl-sulfonamide anticancer agent, administered on a weekly schedule to patients with solid tumors. Clin Cancer Res, 2003;9:5195–5204.
Droz JP. Phase I trial of five-days continuous infusion E7070 [N-(3-chloro-7-indolyl)-1,4-benzenedisulfonamide] in patients with solid tumors. Proc Am Associ Cancer Res, 2000;41:609.
Haddad RI, Weinstein LJ, Wieczorek TJ et al. A phase II clinical and pharmacodynamic study of E7070 in patients with metastatic, recurrent, or refractory squamous cell carcinoma of the head and neck: modulation of retinoblastoma protein phosphorylation by a novel chloroindolyl sulfonamide cell cycle inhibitor. Clin Cancer Res, 2004;10:4680–4687.
Smyth JF, Aamdal S, Awada A et al. Phase II study of E7070 in patients with metastatic melanoma. Ann Oncol, 2005;16:158–161.
Mainwaring PN, Van Cutsem E, Van Laethem J et al. 2002, ASCO Annual Meeting (Abstract 611).
Talbot D, Norbury C, Slade M et al. 2002, ASCO Annual Meeting (Abstract 1306).
Whittaker SR, Walton MI, Garrett MD and Workman P. The Cyclin-dependent kinase inhibitor CYC202 (R-roscovitine) inhibits retinoblastoma protein phosphorylation, causes loss of Cyclin D1, and activates the mitogen-activated protein kinase pathway. Cancer Res, 2004;64:262–272.
McClue SJ, Blake D, Clarke R et al. In vitro and in vivo antitumor properties of the cyclin dependent kinase inhibitor CYC202 (R-roscovitine). Int J Cancer, 2002;102:463–468.
Goodyear S, Sharma MC. Roscovitine regulates invasive breast cancer cell (MDA-MB231) proliferation and survival through cell cycle regulatory protein cdk5. Exp Mol Pathol 2007 Feb;82(1):25–32.
Raynaud FI, Whittaker SR, Fischer PM et al. In vitro and in vivo pharmacokinetic-pharmacodynamic relationships for the trisubstituted aminopurine cyclin-dependent kinase inhibitors olomoucine, bohemine and CYC202. Clin Cancer Res, 2005;11:4875–4887.
Pierga J, Faivre S, Vera K et al. A phase I and pharmacokinetic (PK) trial of CYC202, a novel oral cyclin-dependent kinase (CDK) inhibitor, in patients (pts) with advanced solid tumors. Proc Am Soc Clin Oncol 22:2003 (abstr 840).
Benson C, White J, Twelves C et al. A phase I trial of the oral cyclin dependent kinase inhibitor CYC202 in patients with advanced malignancy. Proc Am Soc Clin Oncol 2003;22:2003 (abstr 838).
White JD, Cassidy J, Twelves C et al. A phase I trial of the oral cyclin dependent kinase inhibitor CYC202 in patients with advanced malignancy. J Clin Oncol, 2004 ASCO Annual Meeting Proceedings (Post-Meeting Edition). Vol 22, No 14S (July 15 Supplement), 2004: 3042.
Siegel-Lakhai WS, Rodenstein DO, Beijnen JH, Gianella-Borradori A, Schellens JH, Talbot DC. Phase I study of seliciclib (CYC202 or R-roscovitine) in combination with gemcitabine (gem)/cisplatin (cis) in patients with advanced Non-Small Cell Lung Cancer (NSCLC). J Clin Oncol, 2005 ASCO Annual Meeting Proceedings. Vol 23, No. 16S, Part I of II (June 1 Supplement), 2005:2060.
Misra RN, Xiao HY, Kim KS et al. N-(cycloalkylamino)acyl-2- aminothiazole inhibitors of cyclin-dependent kinase 2. N-[5-[[[5-(1,1-dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4-piperidinecarbo-xamide (BMS-387032), a highly efficacious and selective antitumor agent. J Med Chem, 2004;47:1719–1728.
Blagden S, de Bono J. Drugging cell cycle kinases in cancer therapy. Curr Drug Targets, 2005;6:325–335.
Jones SF, Burris HA, Kies M et al. A phase I study to determine the safety and pharmacokinetics (PK) of BMS-387032 given intravenously every three weeks in patients with metastatic refractory solid tumors. Proc Am Soc Clin Oncol 22:2003 (abstr 798).
McCormick J, Gadgeel SM, Helmke W et al. Phase I study of BMS-387032, a cyclin dependent kinase (CDK) 2 inhibitor Proc Am Soc Clin Oncol 22:2003 (abstr 835)
Shapiro G, Lewis N, Bai S et al. A phase I study to determine the safety and pharmacokinetics (PK) of BMS-387032 with a 24-hr infusion given every three weeks in patients with metastatic refractory solid tumors Proc Am Soc Clin Oncol 22:2003 (abstr 799).
Siemeister G, Luecking U, Wagner C, Detjen K, Mc Coy C and Bosslet K. Molecular and pharmacodynamic characteristics of the novel multi-target tumor growth inhibitor ZK 304709. Biomed Pharmacother, 2006;60:269–272.
Graham J, Wagner K, Plummer R et al. Phase I dose-escalation study of novel oral multi-target tumor growth inhibitor (MTGI) ZK 304709 administered daily for 7 days of a 21-day cycle to patients with advanced solid tumors. J Clin Oncol, 2006 ASCO Annual Meeting Proceedings Part I. Vol 24, No. 18S (June 20 Supplement), 2006: 2073.
Ahmed S, Molife R, Shaw H et al. Phase I dose-escalation study of ZK 304709, an oral multi-target tumor growth inhibitor (MTGI), administered for 14 days of a 28-day cycle. J Clin Oncol, 2006 ASCO Annual Meeting Proceedings Part I. Vol 24, No. 18S (June 20 Supplement), 2006:076.
Toogood PL, Harvey PJ, Repine JT et al. Discovery of a potent and selective inhibitor of cyclin-dependent kinase 4/6. J Med Chem, 2005;48:2388–2406.
Shapiro GI. Cyclin-dependent kinase pathways as targets for cancer treatment. J Clin Oncol, 2006;24: 1770–1783.
Fry DW, Harvey PJ, Keller PR et al. Specific inhibition of cyclin-dependent kinase 4/6 by PD 0332991 and associated antitumor activity in human tumor xenografts. Mol Cancer Ther, 2004;3: 1427–1438.
Lin TS, Dalton JT, Wu D et al. Flavopiridol given as a 30-min intravenous (IV) bolus followed by 4-hr continuous IV infusion (CIVI) results in clinical activity and tumor lysis in refractory chronic lymphocytic leukemia (CLL). J Clin Oncol, 2004 ASCO Annual Meeting Proceedings (Post-Meeting Edition). Vol 22, No 14S (July 15 Supplement), 2004:6564.
Martin A, Odajima J, Hunt SL et al. Cdk2 is dispensable for cell cycle inhibition and tumor suppression mediated by p27(Kip1) and p21(Cip1). Cancer Cell, 2005;7:591–598.
Fritz B, Schubert F, Wrobel G et al. Microarray-based copy number and expression profiling in dedifferentiated and pleomorphic liposarcoma. Cancer Res, 2002;62:2993–2998.
Arlander SJ, Eapen AK, Vroman BT, McDonald RJ, Toft DO, Karnitz LM. Hsp90 inhibition depletes Chk1 and sensitizes tumor cells to replication stress. J Biol Chem, 2003;278:52572–52577.
Richon VM, Sandhoff TW, Rifkind RA, Marks PA. Histone deacetylase inhibitor selectively induces p21WAF1 expression and gene-associated histone acetylation. Proc Natl Acad Sci U S A, 2000;97: 10014–10019.
Vassilev LT, Vu BT, Graves B et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science, 2004;303: 844–848.
Kim DM, Koo SY, Jeon K et al. Rapid induction of apoptosis by combination of flavopiridol and tumor necrosis factor (TNF)-alpha or TNF-related apoptosis-inducing ligand in human cancer cell lines. Cancer Res,2003;63:621–626.
Dai Y, Rahmani M, Grant S. Proteasome inhibitors potentiate leukemic cell apoptosis induced by the cyclin-dependent kinase inhibitor flavopiridol through a SAPK/JNK- and NF-kappaB-dependent process. Oncogene, 2003;22:7108–7122.
Senderowicz AM. Targeting cell cycle and apoptosis for the treatment of human malignancies. Curr Opin Cell Biol, 2004;16:670–678.
McArthur GA, Raleigh J, Blasina A et al. Imaging with FLT-PET demonstrates that PF-477736, an inhibitor of CHK1 kinase, overcomes a cell cycle checkpoint induced by gemcitabine in PC-3 xenografts. Journal of Clinical Oncology, 2006 ASCO Annual Meeting Proceedings Part I. Vol 24, No. 18S (June 20 Supplement), 2006:3045.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Humana Press, a part of Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Yap, T.A., Molife, L.R., Bono, J.S.d. (2008). CDK Inhibitors as Anticancer Agents. In: Dai, W. (eds) Checkpoint Responses in Cancer Therapy. Cancer Drug Discovery and Development•. Humana Press. https://doi.org/10.1007/978-1-59745-274-8_6
Download citation
DOI: https://doi.org/10.1007/978-1-59745-274-8_6
Publisher Name: Humana Press
Print ISBN: 978-1-58829-930-7
Online ISBN: 978-1-59745-274-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)