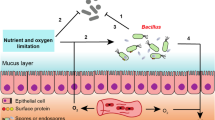
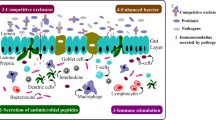
Abstract
Although crude mixtures of microorganisms (e.g., fermented milk products or poultices of moldy bread) have been used since antiquity to treat infections, the first scientific description of a biotherapeutic effect was made at the beginning of this century by the Russian Metchnikoff for which he received the Nobel Prize in 1908. Metchnikoff demonstrated that certain species of bacteria were able to enhance the proliferation of Vibrio cholerae, whereas other species inhibited its growth (1). A preliminary, but fundamental distinction needs to be made between the concept of “prebiotic” and “probiotic.” A “prebiotic” can be defined as a nonmetabolized, nonabsorbed substrate that is useful for the host by selectively enhancing the growth and/or the metabolic activity of a bacterium or of a group of bacteria (e.g., lactulose effect on the colonic flora) (2). A “probiotic” or a “biotherapeutic agent” (BTA) is a living microorganism administered to promote the health of the host by treating or preventing infections owing to strains of pathogens (3,4). Both terms of “probiotic” and “biotherapeutic agent” have been used in the literature to describe microorganisms that exert antagonistic activity against pathogens in vivo (4,5). BTA seems to be the more appropriate term, because it denotes a microorganism having therapeutic properties (3). Ideally, BTA should be innocuous, act against pathogens by multiple mechanisms to minimize the development of resistance (Table 1), and marshal host defenses to destroy the invading pathogen. An additional desirable property would be an immediate onset of action (in contrast to a vaccine that takes several weeks to stimulate antibody production).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Altwegg, M. (1992) La biothérapie dans la diarrhée. Der Informiete Arz 13, 1–16.
Gibson, G. R., and Roberfroid, M. B. (1995) Dietary modulation of the human colonic microflora: Introducing the concept of prebiotics. J. Nutr. 125, 1405–1412.
Elmer, G. W., Surawicz, C. M., and McFarland, L. V. (1996) Biotherapeutic agents. A neglected modality for the treatment and prevention of selected intestinal and vaginal infections. DAMA 275, 870–876.
Fuller, R. (1991) Probiotics in human medicine. Gut 32, 489–492.
McFarland, L. V. and Elmer, G. W. (1995) Biotherapeutic agents: past, present and future. Microecology Ther 23, 46–73.
Roffe, C. (1996) Biotherapy for antibiotic-associated and other diarrhoeas. J. Infect. 32, 1–10.
Tvede, M. and Rask-Madsen, J. (1989) Bacteriotherapy for chronic relapsing Clostridium difficile diarrhoea in six patients. Lancet 27, 1156–1160.
Marteau, P. and Rambaud, J. C. (1993) Potential of using lactic acid bacteria for therapy and immunomodulation in man. FEMS Microbiol. Rev. 12, 207–220.
Alm, L. and Petterson, L. (1980) Survival rate of Lactobacilli during digestion. An in vitro study. Am. J. Clin. Nutr. 33, 2543–2550.
Bergogne-Bérézin, E. (1995) Impact écologique de l’antibiothérapie, place des microorganismes de substitution dans le contrôle des diarrhées et colites associées aux antibiotiques. Presse Med. 24, 145–156.
Goldin, B. R., Gorbach, S. L., Saxelin, M., Barakat, S., Gualtieri, L., and Salminen, S. (1992) Survival of Lactobacillus species (strain GG) in human gastrointestinal tract. Dig. Dis. Sci. 37, 121–128.
Lidbeck, A., Gustafsson, J. A., and Nord, C. E. (1987) Impact of Lactobacillus acidophilus supplements on the human oropharyngeal and intestinal microflora. Scand. J. Infect. Dis. 19 , 531-537.
Bouhnik, Y., Pochart, P., Marteau, P., Arlet, G., Goderel, I., and Rambaud, J. C. (1992) Fecal recovery in humans of viable Bifidobacterium sp. ingested in fermented milk. Gastroenterology 102, 875–878.
Silva, M., Jacobus, N. V., Deneke, C., and Gorbach, S. L. (1987) Antimicrobial substance from a human Lactobacillus strain. Antimicrob. Agents Chemother. 31, 1231–1233.
Vandenbergh, P. A. (1993) Lactic acid bacteria, their metabolic products and interference with microbial growth. FEMS Microbiol. Rev. 12, 221–238.
Ramare, F., Nicoli, J., Dabard, J. et al. (1993) Trypsin-dependent production of an antimicrobial substance by a human Peptostreptococcus strain in gnotobiotic rats and in vitro. Appl. Environ. Microbiol. 59, 2876–2883.
Kotz, C. M., Peterson, L. R., Moody, J. A., Savaiano, D. A., and Levitt, M. D. (1992) Effect of yoghurt on Clindamycin-induced Clostridium difficile colitis in hamsters. Dig. Dis. Sci. 37, 129–132.
Blomberg, L., Henmiksson, A., and Conway, P. L. (1993) Inhibition of adhesion of Escherichia coli K88 to piglet ileal mucus by Lactobacillus spp. Appl. Environ. Microb. 59, 34–39.
Bernet, M. F, Brassart, D., Neeser, J. R., and Servin, A. L. (1994) Lactobacillus acidophilus LAI binds to cultured human intestinal cell lines and inhibits cell attachment and cell invasion by enterovirulent bacteria. Gut 35, 483–489.
Coconnier, M. H., Francoise, B., Chauvière, G., and Servin, A. L. (1993) Adhering heat-killed human Lactobacillus acidophilus. Strain LB inhibits the process of pathogenicity of diarrhoeagenic bacteria in cultured human intestinal cells. J. Diarrhoea! Dis. Res. 11, 235–242.
Chauvière, G, Coconnier, M. H., Kernies, S., Darfeville-Michaud, A., Joly, B., and Servin, A. L. (1992) Competitive exclusion of diarrhoeagenic Escherichia coli ETEC from human enterocyte-like CACO-2 cells by “heat-killed Lactobacillus.” FEMS Microbiol. Lett. 91, 213–218.
Clements, M. L., Levine, M. M., Black, R. E., Robins-Browne Luis, A., Cisneros, R. M., et al. (1981) Lactobacillus prophylaxis for diarrhoea due to enterotoxigenic Escherichia coli. Antimicrob. Agents Chemother. 20, 104–108.
Pozo-Olano, J. D., Warram, J. H., Gomez, R. G., and Cavazos, M. G. (1978) Effect of a Lactobacilli preparation on traveller’s diarrhoea. Gastroenterology 74, 829, 830.
Peetermans, W. (1991) Reizigerdiarree. Tijschr voor Geneeskunde 47, 895–903.
Perdigon, G., De Macias, M. E. N., Alvarez, S., Olivier, G., and De Ruiz Holgada, A. A. P. (1986) Effect of perorally administered Lactobacilli on macrophage activation in mice. Infect. Immun. 3, 404–410.
Kaila, M., Isolauri, E., Soppi, E., Virtanen, E., Laine, S., and Arvilommi, H. (1992) Enhancement of the circulating antibody secreting cell response in human diarrhoea by a human Lactobacillus strain. Pediatr Res. 32, 141–144.
Kaila, M., Isolauri, E., Saxelin, M., Arvilommi, H., and Vesikari, T. (1995) Viable versus inactivated Lactobacillus strain GG in acute Rotavirus diarrhea. Arch. Dis. Child. 72, 51–53.
Majamaa. (1995) Lactic acid bacteria in the treatment of acute Rotavirus gastroenteritis. J. Med. Gastroenterol. Nutr. 20, 333–338.
McFarland, L. V. and Bernasconi, P. (1993) Saccharomyces boulardii: A review of an innovative biotherapeutic agent. Microbial. Ecology Health Dis. 6, 157–171.
McFarland, L. V. (1996) Saccharomyces boulardii is not Saccharomyces cerevisiae. Clin. Infect. Dis. 22, 200, 201.
Berg, R., Bernasconi, P., Fowler, D., and Gautreaux, M. (1993) Inhibition of Candida albicans translocation from the gastrointestinal tract of immunosuppressed mice by oral treatment with Saccharomyces boulardii. J. Infect. Dis. 168, 1314–1318.
Blehaut, H., Massot, J., Elmer, G. W., and Levy, R. H. (1989) Disposition kinetics of Saccharomyces boulardii in man and rat. Biopharm. Drug Dispos. 10, 353–364.
Boddy, A. V., Elmer, G. W., McFarland, L. V., and Levy, R. H. (1991) Influence of antibiotics on the recovery and kinetics of Saccharomyces boulardii in rats. Pharm. Res. 8, 796–800.
Ducluzeau, R. and Bensaada, M. (1982) Effet comparé de l’administration unique ou en continu du Saccharomyces boulardii sur l’établissement de diverses souches de Candida dans le tractus digestif de souris gnotoxéniques. Annales de Microbiologie (Paris) 133, 491–501.
Klein, S. M., Elmer, G. W., McFarland, L. V., Surawicz, C. M., and Levy, R. H. (1993) Recovery and elimination of the biotherapeutic agent, Saccharomyces boulardii, in healthy human volunteers. Pharm. Res. 10, 1615–1619.
Elmer, G. W., Moyer, K. A., Vega, R., Surawicz, C. M., Collier, A. C., Hooton, T. M., et al. (1995) Evaluation of Saccharomyces boulardii for patients with HIV-related diarrhea and healthy volunteers receiving antifungals. Microecology Ther. 25, 23–31.
Rigothier, M. C., Maccario, J., and Gayral, P. (1994) Inhibitory activity of Saccharomyces yeasts on the adhesion of Entamoeba histolytica trophozoites to human erythrocytes in vitro. Parasitol. Res. 80, 10–15.
Rigothier, M. C., Maccario, J., Vuong, P. N., and Gayral, P. (1990) Effets des levures Saccharomyces boulardii sur les trophozoites d’Entamoeba histolytica in vitro et dans l’amibiase caecale du jeune rat. Ann. de Parasitologie Humaine et Comparée 65, 51–60.
Knause, W., Matheis, H., and Wulf, K. (1969) Fungaemia and funguria after oral administration of Candida albicans. Lancet i, 598–600.
Vincent, P., Colombel, J. F., Lescut, D., Fournier, L., Savage, C, Cortot, A., et al. (1988) Bacterial translocation in patients with colorectal cancer. J. Infect. Dis. 158, 1395, 1396.
Vidon, N., Huchet, B., and Rambaud, J. C. (1986) Influence de Saccharomyces boulardii sur la sécrétion jéjunale induite chez le rat par la toxine cholérique. Gastroentérol. Clin. Biol. 10, 13–16.
Czerucka, D., Roux, I., and Rampal, P. (1994) Saccharomyces boulardii inhibits secretagogue-mediated adenosine 3’,5’-cyclic monophosphate induction in intestinal cells. Gastroenterology 106, 65–72.
Delmée, M. and Buts, J. P. (1993) Clostridium difficile-associated diarrhoea in children, in Management of Digestive and Liver Disorders in Infants and Children. Textbook (Buts, J. P. and Sokal, E., eds.), Elsevier Sciences, Amsterdam, pp. 371–379.
Buts, J. P., Corthier, G., and Delmée, M. (1993) Saccharomyces boulardii for Clostridium difficile-associated enteropathies in infants. J. Pediatr. Gastroenterol. Nutr. 16, 419–425.
Buts, J. P., Weber, A. M., Roy, C. C., and Morin, C. L. (1977) Pseudomembranous enterocolitis in childhood. Gastroenterology 73, 823–827.
Bartlett, J. G., Chang, T. W., Gurwith, M., Gorbach, S. L., and Onderdonck, A. B. (1978) Antibiotic-associated pseudomembranous colitis due to toxin-producing Clostridia. N. Engl. J. Med. 298, 531–534.
McFarland, L. V., Mulligan, M. E., Kwok, R. Y. Y, and Stamm, W. E. (1989) Nosocomial acquisition of Clostridium difficile infection. N. Engl. J. Med. 320, 204–210.
McFarland, L. V., Surawicz, C. M., and Stamm, W. E. (1991) Risk factors for Clostridium difficile carriage and C. difficile-associated diarrhea in a cohort of hospitalized patients. J. Infect. Dis. 162, 678–684.
Johnson, S., Gerding, D. N., Olson, M. M., Weiler, M. D., Hughes R. A., Clabots, C. R., et al. (1990) Prospective controlled study of vinyl glove use to interrupt Clostridium difficile nosocomial transmission. Am. J. Med. 88, 137–140.
Delmée, M., Verellen, G., Avesani, V., and François, G. (1988) Clostridium difficile in neonates: serogrouping and epidemiology. Eur. J. Pediatr. 147, 36–40.
Corthier, G., Dubos, F., and Ducluzeau, R. (1986) Prevention of Clostridium difficile induced mortality in gnotobiotic mice by Saccharomyces boulardii. Can. J. Microbiol. 32, 894–896.
Elmer, G. W., and Corthier, G. (1991) Modulation of Clostridium difficile induced mortality as a function of the dose and the viability of the Saccharomyces boulardii used as a preventative agent in gnotobiotic mice. Can. J. Microbiol. 37, 315–317.
Elmer, G. W. and McFarland, L. V. (1987) Suppression by Saccharomyces boulardii of toxinogenic Clostridium difficile overgrowth after vancomycin treatment in hamsters. Antimicrob. Agents Chemother. 31, 129–131.
Massot, J., Sanchez, O., Couchy, R., Astoin, J., and Parodi, A. L. (1984) Bakteriopharmakologische aktivität von Saccharomyces boulardii bei der Clindamycininduzierten kolitis in hamster. Arzneimittel Forshung 794–797.
Corthier, G., Dubos-Ramare, F., Muller, M. C., Mahé, S., Vernet, A., Elmer, G. W., et al. (1991) Etude expérimentale chez les souris à flore contrôlée, des moyens écologiques de lutte contre la pathologie à Clostridium difficile, in Clostridium difficile-Associated Intestinal Diseases ( Rambaud, J. C., and Ducluzeau, R., eds.), Springer-Verlag, Paris, pp. 105–113.
Castex, F., Corthier, G., Jouvert, S., Elmer, G. W., Guibal, J., Lucas, F., et al. (1989) Prevention of experimental pseudomembranous cecitis by Saccharomyces boulardii: topographical histology of the mucosa, bacterial counts and analysis of toxin production, in Microecology and Therapy, vol. 19 (Dougherty, S. J., Hentges, D. L., Lyerly, D. M., et al., eds.), Institute for Microbiology Herborn-Hill, p. 241.
Corthier, G., Lucas, R., Jouvert, S., and Castex, F. (1992) Effect of oral Saccharomyces boulardii treatment on the activity of Clostridium difficile toxins in mouse digestive tract. Toxicon 30, 1583–1589.
Czerucka, D., Nano, J. L., Bernasconi, P., and Rampal, P. (1991) Réponse aux toxines A et B de Clostridium difficile d’une lignée de cellules épithéliales intestinales de rat: IRD 98. Effet de Saccharomyces boulardii. Gastroentérol. Clin. Biol. 15, 22–27.
Pothoulakis, C., Kelly, C. P., Joshi, M. A., Gao, N., O’Keane, C. J., Castagliuolo, I., et al. (1993) Saccharomyces boulardii inhibits Clostridium difficile toxin A binding and enterotoxicity in rat ileum. Gastroenterology 104, 1108–1115.
Pothoulakis, C., LaMont, J. T., Eglow, R., Gao, N., Rubbins, J. B., Theoharides, T. C., et al. (1991) Characterization of rabbit ileal receptors for Clostridium difficile toxin A. Evidence for a receptor-coupled G protein. J. Clin. Invest. 88, 119–125.
Castagliuolo, I., LaMont, J. T., Nikulasson, S. T., and Pothoulakis, C. (1996) Saccharomyces boulardii protease inhibits Clostridium difficile toxin A effects in the rat ileum. Infect. Immun. 64, 5225–5232.
Machado, P., Caetano, J. A., Parames, M. T., Babo, M. J., Santos, A., Bandeira Ferreira, A., et al. (1986) Immunopharmacological effects of Saccharomyces boulardii in healthy volunteers. Int. J. Immunopharmacol. 8, 245–249.
Buts, J. P., Bernasconi, P., Vaerman, J. P., and Dive, C. (1990) Stimulation of secretory IgA and secretory component of immunoglobulins in small intestine of rats treated with Saccharomyces boulardii. Dig. Dis. Sci. 35, 251–256.
Caetano, J. A., Parames, M. T., Babo, M. J., et al. (1986) Immunopharmacological effects of Saccharomyces boulardii in healthy human volunteers. Int. J. Immunopharmacol. 8, 249–259.
Buts, J. P., Bernasconi, P., Van Craynest, M. P., Maldague, P., and De Meyer, R. (1986) Response of human and rat small intestinal mucosa to oral administration of Saccharomyces boulardii. Pediatr. Res. 20, 192–196.
Jahn, H. U., Ullrich, R., Schneider, T., Liehr, R. M., Schieferdecker, H. M., Holst, H., et al. (1996) Immunological and trophical effects of Saccharomyces boulardii on the small intestine of healthy human volunteers. Digestion 57, 95–104.
Buts, J. P., De Keyser, N., Marandi, S., Hermans, D., Chae Yhe, Lambotte, L., et al. (1997) Saccharomyces boulardii upgrades cellular adaptation following proximal enterectomy in rats. Am. J. Physiol,submitted.
Miret, W., Solari, A. J., Barderi, P. A., and Goldenberg, S. H. (1992) Polyamines and cell wall organization in Saccharomyces boulardii. Yeast 8, 1033–1041.
Buts, J. P., De Keyser, N., and De Raedemaeker, L. (1994) Saccharomyces boulardii enhances rat intestinal enzyme expression by endoluminal release of polyamines. Pediatr. Res. 36, 522–527.
Buts, J. P., De Keyser, N., Kolanowski, J. Sokal, E. M., and Van Hoof, F. (1993) Maturation of villus and crypt cell functions in rat small intestine: role of dietary polyamines. Dig. Dis. Sci. 38, 1091–1098.
Kobayashi, M., Iseki, K., Saitoh, H., and Miyazaki, K. (1992) Uptake characteristics of polyamines into rat intestinal brush border membrane. Biochem. Biophys. Acta. 1105, 177–183.
Iseki, K., Kobayashi, M., and Miyazaki, K. (1991) Spermine uptake by rat brush border membrane vesicles. Biochem. Biophys. Acta. 1068, 105–110.
Kumagai, J. and Johnson, L. R. (1988) Characteristics of putrescine uptake in isolated rat enterocytes. Am. J. Physiol. 254, G81–G86.
Buts, J. P., De Keyser, N., Romain, N., Dandrifosse, G., Sokal, E. M., and Nsengiyumva, T. (1994) Response of rat immature enterocytes to insulin: regulation by receptor binding and endoluminal polyamine uptake. Gastroenterology 106, 58–68.
Kelly, D., King, T. P., Brown, D. S., and McFadyen, M. (1991) Polyamine profiles of porcine milk and of intestinal tissue of pigs during suckling. Reprod. Nutr. Rev. 31, 73–80.
Buts, J. P., De Keyser, N., and De Raedemaeker, L. (1995) Polyamine profile in human milk, infant artificial formulae and semi-elemental diets. J. Pediatr. Gastroenterol. Nutr. 21, 44–49.
Buts, J. P. (1996) Polyamines in milk, in Bioactive Factors in Milk. Ann. Nestlé 54, 98–104.
Osbourne, D. L. and Seidel, E. R. (1989) Microflora derived polyamines modulate obstruction-induced mucosal hypertrophy. Am. J. Physiol. 256, G1049 — G1057.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1999 Springer Science+Business Media New York
About this chapter
Cite this chapter
Buts, JP. (1999). Mechanisms of Action of Biotherapeutic Agents. In: Elmer, G.W., McFarland, L.V., Surawicz, C.M. (eds) Biotherapeutic Agents and Infectious Diseases. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-59259-711-6_2
Download citation
DOI: https://doi.org/10.1007/978-1-59259-711-6_2
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-4757-4652-5
Online ISBN: 978-1-59259-711-6
eBook Packages: Springer Book Archive