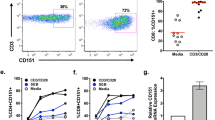
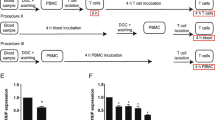
Abstract
Membrane alanyl aminopeptidase (APN, EC 3.4.1 1.2) is a 150-kDa metalloprotease which has been identified as the leukocyte surface differentiation antigen CD131. In humans the APN gene is located on the long arm of chromosome 15 (ql 1-gter)2 with the coding part of the gene encoded by 20 exons3. Within the haematopoetic system APN is dominantely expressed in cells of the myelo-monocytic lineage and is used, therefore, as a standard marker in the diagnosis of leukaemia. Aminopeptidase N of leukocytes is supposed to be involved in the degradation of neuropeptides4–7 and cytokines8,9, but its function remains to be fully elucidated. APN may function as a corona virus receptor10–13 and seems to contribute in tumor invasion and matrix degradation14–15. APN is also implicated in antigen processing16. Furthermore, anti-CD13 monoclonal antibodies have been shown to neutralize CMV17. In recent years evidence accumulated showing that malignant B and T cells18–23 as well as activated T cells24–26 are capable of expressing APN on the cell surface. A similar mechanism of induction may underlie the CD13 surface expression of tumor infiltrating T cells27, or T cells28 and NK cells29 derived from local sites of inflammation.
Chapter PDF
Similar content being viewed by others
Keywords
- Acute Lymphoblastic Leukemia
- Aminopeptidase Activity
- CD13 Surface Expression
- Corona Virus
- Aminopeptidase Inhibitor
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Look, A.T., Ashmun, R.A., Shapiro, L.H., and Peiper, S.C. (1989). Human myeloid plasma membrane glycoprotein CD13 (gp150) is identical to aminopeptidase N. J. Clin. Invest. 83, 1299–1307.
Watt, V.M. and Willard, H.F. (1990). The human aminopeptidase N gene: isolation, chromosome localization, and DNA polymorphism analysis. Hum. Genet. 85, 651–654.
Lerche, C., Vogel, L.K., Shapiro, L.H., Noren, O., and Sjöström, H (1996). Human aminopeptidase N is encoded by 20 exons. Mammalian Genome 7, 712–713.
Furuhashi, M., Mizutani, S., Kurauchi, O., Kasugai, M., Narita, O., and Tomoda, Y. (1988). In vitro degradation of opioid peptides by human placental aminopeptidase M. Exp. Clin. Endocrinol. 92, 235–237.
Giros, B., Gros, C., Solhonne, B., and Schwartz, J.C. (1986). Characterization of amino-peptidases responsible for inactivating endogenous (MetS)enkephalin in brain slices using peptidase inhibitors and anti-aminopeptidase M antibodies. Mol. Pharmacol. 29, 281–287.
Ahmad, S., Wang, L., and Ward, P.E. (1992). Dipeptidyl(amino)peptidase IV and aminopeptidase M metabolize circulating substance P in vivo. J. Pharmacol. Exp. Ther. 260, 1257–1261.
Shimamura, M., Hazato, T., and Iwaguchi, T. (1991). Enkephalin-degrading aminopeptidase in the longitudinal muscle layer of guinea pig small intestine: its properties and action on neuropeptides. J. Biochem. Tokyo./09, 492–497.
Kanayama, N., Kajiwara, Y.. Goto, J.. el Maradny, E., Maehara, K., Andou, K., and Terao, T. (1995). Inactivation of interleukin-8 by aminopeptidase N (CD13). J. Leukoc. Biol. 57, 129–134.
Hoffmann, T., Faust, J., Neubert, K., and Ansorge, S. (1993). Dipeptidyl peptidase IV (CD 26) and aminopeptidase N (CD 13) catalyzed hydrolysis of cytokines and peptides with N-terminal cytokine sequences. FEBS Lett. 336, 61–64.
Delmas, B., Gelfi, J., L’Haridon, R., Vogel, L.K., Sjöström, H., Noren, O., and Laude, H. (1992). Aminopeptidase N is a major receptor for the entero-pathogenic coronavirus TGEV. Nature 357, 417–420.
Delmas, B., Gelfi, J., Sjöström, H., Noren, O., and Laude, H. (1993). Further characterization of aminopeptidase-N as a receptor for coronaviruses. Adv. Exp. Med. Biol. 342, 293–298.
Delmas, B., Gelfi, J., Kut, E., Sjöström, H., Noren, O., and Laude, H. (1994). Determinants essential for the transmissible gastroenteritis virus-receptor interaction reside within a domain of aminopeptidase-N that is distinct from the enzymatic site. J. Virol. 68, 5216–5224.
Yeager, C.L., Ashmun, R.A., Williams, R.K., Cardellichio, C.B., Shapiro, L.H., Look, A.T., and Holmes, K.V. (1992). Human aminopeptidase N is a receptor for human coronavirus 229E. Nature 357, 420–422.
Saiki, I., Fujii, H., Yoneda, J., Abe, E, Nakajima, M., Tsuruo, T., and Azuma, 1. (1993). Role ofaminopeptidase N (CD13) in tumor-cell invasion and extracellular matrix degradation. Int. J. Cancer 54, 137–143.
Fujii, H., Nakajima, M., Saiki, I., Yoneda, J., Azuma, 1., and Tsuruo, T. (1995). Human melanoma invasion and metastasis enhancement by high expression of aminopeptidase N/CDI3. Clin. Exp. Metastasis 13, 337–344.
Hansen, A.S., Noren, O., Sjöström, H., and Werdelin, O. (1993). A mouse aminopeptidase N is a marker for antigen-presenting cells and appears to be co-expressed with major histocompatibility complex class II molecules. Eur. J. Immunol. 23, 2358–2364.
Giugni, T.D., Söderberg, C., Ham, D.J., Bautista, R.M., Hedlund, K.-O., Möller, E., and Zaia, J.A. (1996). Neutralization of human cytomegalovirus by human CD13-specific antibodies. The Journal of Infectious Diseases 173, 1062–1071.
Hsu, P.N., Tien, H.F., Wang, C.H., Chen, Y.C., Shen, M.C., Lin, D.T., Lin, K.H., Liang, D.C., and Lin, K.S. (1991). A subset of acute lymphoblastic leukemia with co-expression of myeloid antigens: prevalence and clinical significance. J. Formos. Med. Assoc. 90, 225–231.
Hara, J., Kawa Ha, K., Yumura Yagi, K., Kurahashi, H., Tawa, A., Ishihara, S., Inoue, M., Murayama, N., and Okada, S. (1991). In vivo and in vitro expression of myeloid antigens on B-lineage acute lymphoblastic leukemia cells. Leukemia 5, 19–25.
Ferrara, F., De Rosa, C., Fasanaro, A., Mele, G., Finizio, O.. Schiavone, E.M., Spada, O.A., Rametta, V., and Del Vecchio, L. (1990), Myeloid antigen expression in adult acute lymphoblastic leukemia: clinicohematological correlations and prognostic relevance. Hematol. Pathol. 4, 93–98.
Drexler, H.G., Thiel, E., and Ludwig, W.-D. (1991), Review of the incidence and clinical relevance of myeloid antigen-positive acute lymphoblastic leukemia. Leukemia 5, 637–645.
Dreno, B., Bureau, B., Stalder, J.F., and Litoux, P. (1990). MY7 monoclonal antibody for diagnosis of cutaneous T-cell lymphoma. Arch. Dermatol. 126, 1454–1456.
Dixon, J., Kaklamanis, L., Turley, H., Hickson, I.D., Leek, R.D., Harris, A.L., and Latter, K.C. (1994). Expression of aminopeptidase-n (CD 13) in normal tissues and malignant neoplasms of epithelial arid lymphoid origin. J. Clin. Pathol. 47, 43–47.
Kunz, D., Bühling, F.. Hütter, H.J., Aoyagi, T., and Ansorge, S. (1993). Aminopeptidase N (CD13, EC 3.3.4.11.2) occurs on the surface of resting and concanavalin A-stimulated lymphocytes. Biol. Chem Hoppe Seyler 374, 291–296.
Lendeckel, U., Wex, T., Reinhold, D., Kähne, T.. Frank, K., Faust, J., Neubert. K., and Ansorge, S… (1996) Biochem. J. 319, 817–821.
Ansorge, S., Schön, E., and Kunz, D. (1991). Membrane-bound peptidases of lymphocytes: functional implications. Biomed. Biochtm, Acta 50, 799–807.
Riemann, D., Göhring, B., and Langner, J. (1994). Expression of aminopeptidase N/CD13 in tumour-infiltrating lymphocytes from human renal cell carcinoma. Immunol. Lett. 42, 19–23.
Riemann, D., Schwachula, A., Hentschel, M., and Langner, J. (1993). Demonstration of CD13/aminopeptidase N on synovial fluid T cells from patients with different forms of joint effusions. tmmunobiology /57, 24–35.
Bühling, F., Kunz, D., Lendeckel, U.. Reinhold, D., and Ansorge. S. (1992). Aminopeptidase N (APN; CDI3) on natural killer cells from peripheral blood and synovial fluid. Immunobiology 186, 157–158.
Lendeckel, U., Wex, T., Kähne, T., Frank, K., Reinhold, D., and Ansorge, S. (1994). Expression of the aminopeptidase N (CDI3) gene in the human T cell lines HuT78 and H9. Cell. Immunol. 153. 214–226.
Wex, T., Lendeckel, U., Wex, H., Frank, K., and Ansorge, S. (1995). Quantification of aminopcptidase N mRNA in T cells by competitive PCR. FEBS Lett. 374, 341–344.
Ino K., Goto S., Kosaki A., Nomura S., Asada E., Misawa T., Furuhashi Y.. Mizutani S.. and Tomoda Y. (1991). Groth inhibitory effect of bestatin on choriocarcinoma cell lines in vitro. Biotherapy 3. 351–357.
no, K., Isobe, K., Goto, S., Nakashima, I., and Tomoda, Y. (1992). Inhibitory effect of bestatin on the growth of human lymphocytes. lmmunophannacology 23, 163–171
Ino, K., Goto, S., Okamoto, T., Nomura, S., Nawa, A., Isobe, K.. Mizutani, S., and Tomoda, Y. (1994). Expression of aminopcptidase N on human choriocarcinoma cells and cell growth suppression by the inhibition of aminopeptidase N activity. Jpn. J. Cancer Res. 85. 927–933.
Sakurada K., Imamura M., Kobayashi M., Tachibana N., Abe K., Tanaka M., Okabe M., Morioka M.. Kasai M., and Sugiura T. (1990). Inhibitory effect of bestatin on the growth of human leukemic cells. Acta Oncologica 29, 799–802.
Tieku, S. and Hooper, N.M. (1992). Inhibition of aminopeptidases N, A, and W. Biochemical Pharmacology 44, 1725–1730.
Aoyagi T., Yoshida S., Nakamura Y., Shigihara Y., Hamada M., and Takeuchi T. (1990). Probestin, a new inhibitor of aminopeptidase M, produced by streptomyces azureus MH663–2F6. The Journal of Antibiotics XL111, 143–148.
Yoshida S., Nakamura Y., Naganawa H., Aoyagi T., and Takeuchi T. (1990). Probestin. a new inhibitor of aminopeptidase M, produced by streptomyces azureus MH663–2F6. The Journal of Antibiotics XL111, 149–156.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1997 Springer Science+Business Media New York
About this chapter
Cite this chapter
Lendeckel, U. et al. (1997). Activation-Dependent Induction of T Cell Alanyl Aminopeptidase and Its Possible Involvement in T Cell Growth. In: Ansorge, S., Langner, J. (eds) Cellular Peptidases in Immune Functions and Diseases. Advances in Experimental Medicine and Biology, vol 421. Springer, Boston, MA. https://doi.org/10.1007/978-1-4757-9613-1_8
Download citation
DOI: https://doi.org/10.1007/978-1-4757-9613-1_8
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4757-9615-5
Online ISBN: 978-1-4757-9613-1
eBook Packages: Springer Book Archive