Abstract
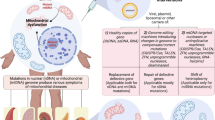
The major function of mitochondria is to generate energy. Mitochondria fulfill this main task by synthesizing ATP from the energy released during the oxidation of substrates and cofactors, a process known as oxidative phosphorylation. Essential for this process is the respiratory chain, a four-enzyme system composed of multiprotein complexes located at the inner mitochondrial membrane. The complexes are responsible for the redox processes that are accompanied by electron transfer and proton pumping activities leading to a membrane potential across the inner mitochondrial membrane. The entire oxidative phosphorylation system is completed by a fifth complex, the ATP synthase, which converts the membrane potential into ATP by condensing ADP and phosphate. The genes required for this energy-generating system are distributed throughout two genomes—the nuclear and the mitochondrial genome. While mitochondria contain the only source of endogenous extrachromosomal DNA, its genes contain the information for only 13 polypeptides of the oxidative phosphorylation system (OXPHOS) system as well as a set of 2 rRNAs and 22 tRNAs essential for their expression. The human mtDNA has evolved to show remarkable economy of organization, containing only a short section of noncoding DNA that is known as the D-loop. However, this region does contain sequences important for replication and transcription of the genome.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Anderson, S., Bankier, A. T., Barrell, B. G., de Bruijn, M. H., Coulson, A. R., Drouin, J., Eperon, I. C., Nierlich, D. P., Roe, B. A., Sanger, F., Schreier, P. H., Smith, A. J., Staden, R., and Young, I. G., 1981, Sequence and organization of the human mitochondrial genome, Nature 290: 457–465.
Bordonne, R., Bandlow, W., Dirheimer, G., and Martin, R. P., 1987, A single base change in the extra-arm of yeast mitochondrial tyrosine tRNA affects its conformational stability and impairs aminoacylation, Mol. Gen. Genet. 206:498–504.
Chang, D. D., and Clayton, D. A., 1987, A mammalian mitochondrial RNA processing activity contains nucleus-encoded RNA, Science 235:1178–1184.
Chang, D. D., and Clayton, D. A., 1989, Mouse RNAase MRP RNA is encoded by a nuclear gene and contains a decamer sequence complementary to a conserved region of mitochondrial RNA substrate, Cell 56:131–139.
Chrzanowska-Lightowlers, Z. M., Lightowlers, R. N., and Turnbull, D. M., 1995, Gene therapy for mitochondrial DNA defects: Is it possible?, Gene Then 2:311–316.
Clayton, D. A., 1991, Nuclear gadgets in mitochondrial DNA replication and transcription, Trends Biochem. Sci. 16:107–111.
Enriquez, J. A., Chomyn, A., and Attardi, G., 1995, MtDNA mutation in MERRF syndrome causes defective aminoacylation of tRNA(Lys) and premature translation termination, Nat. Genet. 10:47–55.
Glick, B., and Schatz, G., 1991, Import of proteins into mitochondria, Annu. Rev. Genet. 25:21–44.
Glick, B. S., Beasley, E. M., and Schatz, G., 1992, Protein sorting in mitochondria, Trends Biochem. Sci. 17:453–459.
Hsieh, C. L., Donlon, T. A., Darras, B. T., Chang, D. D., Topper, J. N., Clayton, D. A., and Francke, U., 1990, The gene for the RNA component of the mitochondrial RNA-processing endo-ribonuclease is located on human chromosome 9p and on mouse chromosome 4, Genomics 6:540–544.
Jacobs, H. T., 1995, Allotropic expression of mitochondrial genes in the nucleus: Towards a gene therapy strategy for mitochondrial diseases, EUROMIT III, Abstractbook, p. 9.
Li, K., Smagula, C. S., Parsons, W. J., Richardson, J. A., Gonzalez, M., Hagler, H. K., and Williams, R. S., 1994, Subcellular partitioning of MRP RNA assessed by ultrastructural and biochemical analysis, J. Cell Biol. 124:871–882.
Mahapatra, S., Ghosh, T., and Adhya, S., 1994, Import of small RNAs into leishmania mitochondria in vitro, Nucleic Acids Res. 22:3381–3386.
Moraes, C. T., Ricci, E., Petruzzella, V., Shanske, S., DiMauro, S., Schon, E. A., and Bonilla, E., 1992, Molecular analysis of the muscle pathology associated with mitochondrial DNA deletions, Nat. Genet. 1:359–367.
Munnich, A., Rustin, P., Rotig, A., Chretien, D., Bonnefont, J. P., Nuttin, C., Cormier, V., Vassault, A., Parvy, P., and Bardet, J., 1992, Clinical aspects of mitochondrial disorders, J. Inherit. Metab. Dis. 15:448–455.
Nagley, P., Farrell, L. B., Gearing, D. P., Nero, D., Meltzer, S., and Devenish, R. J., 1988, Assembly of functional proton-translocating ATPase complex in yeast mitochondria with cyto-plasmically synthesized subunit 8, a polypeptide normally encoded within the organelle, Proc. Natl. Acad. Sci. USA 85:2091–2095.
Neupert, W., Haiti, F. U., Craig, E. A., and Pfanner, N., 1990, How do polypeptides cross the mitochondrial membranes? Cell 63:447–450.
Pak, Y. K., and Weiner, H., 1990, Import of chemically synthesized signal peptides into rat liver mitochondria, J. Biol. Chem. 265:14298–14307.
Pfanner, N., and Neupert, W., 1990, The mitochondrial protein import apparatus, Annu. Rev. Biochem. 59:331–353.
Pfanner, N., Söllner, T., and Neupert, W., 1991, Mitochondrial import receptors for precursor proteins, Trends Biochem. Sci. 16:63–67.
Schmitt, M. E., and Clayton, D. A., 1993, Nuclear RNase MRP is required for correct processing of pre-5.8S rRNA in Saccharomyces cerevisiae, Mol. Cell Biol. 13:7935–7941.
Schon, E. A., and King, M. P., 1995, Physical communication between mammalian mitochondria: A genetic approach, EUROMIT III, Abstractbook, p. 7.
Seibel, P., and Seibel, A., 1994, Chimäres Peptid-Nukleinsäure-Fragment, Verfahren zu seiner Herstellung und Verfahren zur zielgerichteten Nukleinsäureeinbringung in Zellorganellen und Zellen, German Patent P 44 21 079.
Seibel, P., and Seibel, A., 1995a, Replikatives und transkriptionsaktives Peptid-Nukleinsäureplasmid, sowie sein Verwendung zur Einbringung in Zellen und Zellorganellen, German Patent P 195 208 15.
Seibel, P., and Seibel, A., 1995b, Chimäres Peptid-Nukleinsäure-Fragment, Verfahren zu seiner Herstellung, sowie seine Verwendung zur zielgerichteten Nukleinsäureeinbringung in Zellorganellen und Zellen, International Patent PCT DE 95/00775.
Seibel, P., Degoul, F., Bonne, G., Romero, N., Francois, D., Paturneau Jouas, M., Ziegler, F., Eymard, B., Fardeau, M., Marsac, C., and Kadenbach, B., 1991, Genetic biochemical and pathophysiological characterization of a familial mitochondrial encephalomyopathy (MERRF), J. Neurol. Sci. 105:217–224.
Seibel, P., Trappe, J., Villani, G., Klopstock, T., Papa, S., and Reichmann, H., 1995, Transfection of mitochondria: Strategy towards a gene therapy of mitochondrial DNA diseases, Nucleic Acids Res. 23:10–17.
Small, I., Marechal Drouard, L., Masson, J., Pelletier, G., Cosset, A., Weil, J.H., and Dietrich, A., 1992, In vivo import of a normal or mutagenized heterologous transfer RNA into the mitochondria of transgenic plants: Towards novel ways of influencing mitochondrial gene expression? EMBO J. 11:1291–1296.
Takiguchi, M., Murakami, T., Miura, S., and Mori, M., 1987, Structure of the rat ornithine carbamoyltransferase gene, a large, X chromosome-linked gene with an atypical promoter, Proc. Natl. Acad. Sci. USA 84:6136–6140.
Tarassov, I., Entelis, N., and Martin, R. P., 1995a, Mitochondrial import of a cytoplasmic lysine-tRNA in yeast is mediated by cooperation of cytoplasmic and mitochondrial lysyl-tRNA synthetases, EMBO J. 14:3461–3471.
Tarassov, I., Entelis, N., and Martin, R. P., 1995b, An intact protein translocating machinery is required for mitochondrial import of a yeast cytoplasmic tRNA, J. Mol. Biol. 245:315–323.
Vestweber, D., and Schatz, G., 1989, DNA-protein conjugates can enter mitochondria via the protein import pathway, Nature 338:170–172.
Wallace, D. C., 1986, Mitotic segregation of mitochondrial DNAs in human cell hybrids and expression of chloramphenicol resistance, Somat. Cell Mol. Genet. 12:41–49.
Wallace, D. C., 1992a, Diseases of the mitochondrial DNA, Annu. Rev. Biochem. 61:1175–1212.
Wallace, D. C., 1992b, Mitochondrial genetics: A paradigm for aging and degenerative diseases? Science 256:628–632.
Wallace, D. C., 1995, 1994 William Allan Award Address. Mitochondrial DNA variation in human evolution, degenerative disease, and aging, Am. J. Hum. Genet. 57:201–223.
Wallace, D. C., Singh, G., Lott, M. T., Hodge, J. A., Schurr, T. G., Lezza, A. M., Elsas, L. J., and Nikoskelainen, E. K., 1988, Mitochondrial DNA mutation associated with Leber’s hereditary optic neuropathy, Science 242:1427–1430.
Yoneda, M., Chomyn, A., Martinuzzi, A., Hurko, O., and Attardi, G., 1992, Marked replicative advantage of human mtDNA carrying a point mutation that causes the MELAS encepha-lomyopathy, Proc. Natl. Acad. Sci. USA 89:11164–11168.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1999 Springer Science+Business Media New York
About this chapter
Cite this chapter
Seibel, P., Schliebs, M., Flierl, A. (1999). Strategy toward Gene Therapy of Mitochondrial DNA Disorders. In: Papa, S., Guerrieri, F., Tager, J.M. (eds) Frontiers of Cellular Bioenergetics. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-4843-0_26
Download citation
DOI: https://doi.org/10.1007/978-1-4615-4843-0_26
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-7196-0
Online ISBN: 978-1-4615-4843-0
eBook Packages: Springer Book Archive