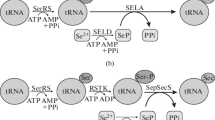
Abstract
In 1954, J. Pinsent discovered that Escherichia coli produces gas from formic acid only when the medium is supplemented with the trace elements molybdenum and selenium (1). Thus, E. coli was the first organism for which a biological role of selenium was demonstrated. It took almost 40 years to identify the enzyme responsible for formate oxidation under the aerobic conditions used by Pinsent:It is formate dehydrogenase 0 (FDH0), which presumably is a respiratory chain-linked enzyme coupling formate oxidation to 02 reduction (2). In addition to FDH0, E. coli possesses two other formate dehydrogenases:FDHN couples formate oxidation to nitrate reduction (3). FDHH is a component of the formate hydrogenlyase complex and delivers the electrons withdrawn from formate to hydrogenase 3, thereby reducing protons to elemental hydrogen (4,5).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Pinsent, J. The need for selenite and molybdate in the formation of formic dehydrogenase by members of the Coliaerogenes group of bacteria. Biochem J 57:10 – 16; 1954.
Sawers, G, Heider, J, Zehelein, E, Bock, A. Expression and operon structure of the sel genes of Escherichia coli and identification of a third selenium-containing formate dehydrogenase isoenzyme J Bacteriol 173:4983 – 4993; 1991.
Enoch, HG, Lester, RL. The purification and properties of formate dehydrogenase and nitrate reductase from Escherichia coli. J Biol Chem 250:6693 – 6705; 1975.
Peck, HD, Gest, H. Formic dehydrogenase and the hydrogenlyase enzyme complex in coli-aerogenes bacteria. J Bacteriol 73:706 – 721; 1957.
Cox, JC, Edwards, ES, DeMoss, JA. Resolution of distinct selenium- containing formate dehydrogenases from Escherichia coli. J Bacteriol 145:1317 – 1324; 1981.
Schlindwein, C, Giordano, G, Santini, C-L, Mandrand, M-A. Identification and expression of the Escherichia coli fdhD and fdhE genes, which are involved in the formation of respiratory formate dehydrogenase. J Bacteriol 172:6112 – 6121; 1990.
Pecher, A, Zinoni, F, Böck, A. The selenopolypeptide of formic dehydrogenase (formate-hydrogen-lyase-linked) from Escherichia coli:genetic analysis. Arch Microbiol 141:359 – 363; 1985.
Andreesen, JR, Ljungdahl, LG. Formate dehydrogenase of Clostridium thermoaceticum, incorporation of selenium-75, and the effect of selenite, molybdate, and tungstate on the enzyme. J Bacteriol 116:867 – 873; 1973.
Jones, JB, Stadtman, TC. Selenium-dependent and selenium- independent formate dehydrogenases of Methanococcus vannielii. J Biol Chem 256:656 – 663; 1981.
Stadtman, TC, Davis, JN, Ching, W-M, Zinoni, F, Böck, A. Amino acid sequence analysis of Escherichia coli formate dehydrogenase (FDHH) confirms that TGA in the gene encodes selenocysteine in the gene product. BioFactors 3:21 – 27; 1991.
Arkowitz, RA, Abeles, RH. Mechanism of action of clostridial glycine reductase:isolation and characterization of a covalent acetyl enzyme intermediate. Biochemistry 30:4090 – 4097; 1991.
Cone, JE, Martin del Rio, R, Davis, JN, Stadtman, TC. Chemical characterization of the organoselenium moiety. Proc Natl Acad Sci USA 73:2659 – 2663; 1976.
Przybyla, A, Robbins, J, Menon, N, Peck Jr, HD. Structure-function relationships among the nickel-containing hydrogenases. FEMS Microbiol Rev 88:109 – 136; 1992.
Eidsness, MK, Scott, RA, Prickril, C, DerVartanian, DV, LeGall, J, Moura, I, Moura, JJG, Peck Jr, HD. Evidence for selenocysteine coordination to the active site nickel in the NiFeSe hydrogenases from Desulfovibrio baculatus. Proc Natl Acad Sci USA 86:147 – 151; 1989.
Voordouw, G, Menon, NK, LeGall, J, Choi, E-S, Peck Jr, HD, Przybyla, AE. Analysis and comparison of nucleotide sequences encoding the genes for [NiFe] and [NiFeSe] hydrogenases from Desulfovibrio gigas and Desulfovibrio baculatus. J Bacteriol 171:2894–2899; 1989.
Stadtman, TC. Selenium-dependent enzymes. Ann Rev Biochem 49:93–110; 1980.
Chambers, I, Frampton, J, Goldfarb, P, Affara, N, McBain, W, Harrison, PR. The structure of the mouse glutathione peroxidase gene:the selenocysteine in the active site is encoded by the termination codon, TGA. EMBO J 5:1221 – 1227; 1986.
Zinoni, F, Birkmann, A, Stadtman, TC, Böck, A. Nucleotide sequence and expression of the selenocysteine containing polypeptide of formate dehydrogenase (formate-hydrogen-lyase-linked) from Escherichia coli. Proc Natl Acad Sci USA 83:4650 – 4654; 1986.
Leinfelder, W, Forchhammer, K, Zinoni, F, Sawers, G, Mandrand-Berthelot, M-A, Böck, A. Escherichia coli genes whose products are involved in selenium metabolism. J Bacteriol 170:540 - 546; 1988.
Zinoni, F, Birkmann, A, Leinfelder, W, Böck, A. Co-translational insertion of selenocysteine into formate dehydrogenase from Escherichia coli directed by a UGA codon. Proc Natl Acad Sci USA 84:3156 - 3161; 1987.
Leinfelder, W, Stadtman, TC, Böck, A. Occurrence in vivo of selenocysteyl-tRNAucA in Escherichia coli. J Biol Chem 264:9720–9723; 1989.
Haddock, BA, Mandrand-Berthelot, M-A. Escherichia coli formate- to-nitrate respiratory chain:genetic analysis. Biochem Soc Trans 10:478 - 480; 1982.
Leinfelder, W, Zehelein, E, Mandrand-Berthelot, M-A, Böck, A. Gene for a novel tRNA species that accepts L-serine and cotranslationally inserts selenocysteine. Nature 331:723 - 725; 1988.
Schon, A, Böck, A, Ott, G, Sprinzl, M, Söll, D. The selenocysteine- inserting opal suppressor serine tRNA from E. coli is highly unusual in structure and modification. Nucleic Acids Res 17:7159 - 7165; 1989.
Baron, Ch, Böck, A. The length of the aminoacyl-acceptor stem of the selenocysteine-specific tRNASec of Escherichia coli is the determinant for binding to elongation factors SELB or Tu. J Biol Chem 266:20375–20379; 1991.
Heider, J, Leinfelder, W, Böck, A. Occurrence and functional compatibility within Enterobacteriaceae of a tRNA species which inserts selenocysteine into protein. Nucleic Acids Res 17:2529—2540; 1989.
Mizutani, T, Maruyama, N, Hitaka, T, Sukenaga, Y. The detection of natural opal suppressor seryl-tRNA in Escherichia coli by the dot blot hybridization and its phosphorylation by a tRNA kinase. FEBS Lett 247:345–348; 1989.
Hatfield, D. Suppression of termination codons in higher eukaryotes. Trends Biochem Sci 10:245–250; 1985.
Forchhammer, K, Leinfelder, W, Boesmiller, K, Veprek, B, Bock, A. Selenocysteine synthase from Escherichia coli:nucleotide sequence of the gene (selA) and purification of the protein. J Biol Chem 266:6318 - 6323; 1991.
Forchhammer, K, Böck, A. Selenocysteine synthase from Escherichia coli:analysis of the reaction sequence. J Biol Chem 266:6324 - 6328; 1991.
Reed, LJ, Cox, DJ. Macromolecular organization of enzyme systems. Ann Rev Biochem 35:57–84; 1966.
Brzovic, P, Litzenberger Holbrook, E, Greene, RC, Dunn, MF. Reaction mechanism of Escherichia coli cystathionine γ-synthase:Direct evidence for a pyridoxamine derivative of vinylglyoxylate as a key intermediate in pyridoxal phosphate dependent γ-elimination and γ-replacement reac-tions. Biochemistry 29:442–451; 1990.
Ehrenreich, A, Forchhammer, K, Tormay, P, Veprek, B, Böck, A. Selenoprotein synthesis in E. coli. Purification and characterisation of the enzyme catalysing selenium activation. Eur J Biochem 206:767 - 773; 1992.
Veres, Z, Tsai, L, Scholz, TD, Politino, M, Balaban RS, Stadtman, TC. Synthesis of 5-methylaminomethyl-2-selenouridine in tRNAs:31P NMR studies show the labile selenium donor synthesized by the selD gene product contains selenium bonded to phosphorus. Proc Natl Acad Sci USA 89:2975–2979; 1992.
Engelhardt, H, Forchhammer, K, Müller, S, Engel, A, Böck, A. Structure of selenocysteine synthase from Escherichia coli and location of tRNA in the seryl-tRNASec -enzyme complex. Molec Microbiol 6:3461 — 3467, 1992.
Forchhammer, K, Boesmiller, K, Böck, A. The function of selenocysteine synthase and SELB in the synthesis and incorporation of seleno¬cysteine. Biochimie 73:1481 – 1486; 1991.
Forchhammer, K, Leinfelder, W, Böck, A. Identification of a novel translation factor necessary for the incorporation of a selenocysteine into protein. Nature 342:453 – 456; 1989.
Förster, Ch, Ott, G, Forchhammer, K, Sprinzl, M. Interaction of a selenocysteine-incorporating tRNA with elongation factor Tu from E. coli. Nucleic Acids Res 18:487 – 491; 1990.
Zinoni, F, Heider, J, Böck, A. Features of the formate dehydrogenase mRNA necessary for decoding of the UGA codon as selenocysteine. Proc Natl Acad Sci USA 87:4660 – 4664; 1990.
Heider, J, Baron, C, Böck, A. Coding from a distance:Dissection of the mRNA determinants required for the incorporation of selenocysteine into protein. EMBO J 11:3759 – 3766; 1992.
Berg, BL, Baron, C, Stewart, V. Nitrate-inducible formate dehydrogenase in Escherichia coli K-12. J Biol Chem 266:22386 – 22391; 1991.
Baron, Ch, Heider, J, Böck, A. Mutagenesis of selC, the gene for the selenocysteine-inserting tRNA-species in E. coli:effects on in vivo function. Nucleic Acids Res 18:6761 – 6766; 1990.
Garcia, GE, Stadtman, TC. Selenoprotein A component of the glycine reductase complex from Clostridium purinolyticum:nucleotide sequence of the gene shows that selenocysteine is encoded by UGA. J Bacteriol 173:2093 – 2098; 1991.
Forchhammer, K, Rücknagel, K-P, Böck, A. Purification and biochemical characterisation of SELB, a translation factor involved in selenoprotein synthesis. J Biol Chem 265:9346 – 9350; 1990.
Li, W-Q, Yarns, M. Bar to normal UGA translation by the selenocysteine tRNA. J Mol Biol 223:9 – 15; 1992.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1994 Springer-Verlag New York
About this chapter
Cite this chapter
Böck, A. (1994). Incorporation of Selenium into Bacterial Selenoproteins. In: Burk, R.F. (eds) Selenium in Biology and Human Health. Springer, New York, NY. https://doi.org/10.1007/978-1-4612-2592-8_2
Download citation
DOI: https://doi.org/10.1007/978-1-4612-2592-8_2
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4612-7597-8
Online ISBN: 978-1-4612-2592-8
eBook Packages: Springer Book Archive