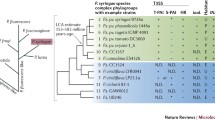
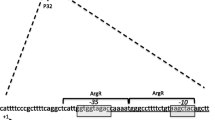
Abstract
Pseudomonas syringae is a member of the gamma subgroup of the Proteobacteria and a plant pathogen that infects many different plants30, Certain P syringae strains can infect only one or a few plant species, and on this basis, P syringae has been separated into greater than 50 pathovars106. While P syringae will eventually kill plant cells (i.e., it is necrogenic), it is typically considered a biotrophic pathogen and it can live on plant leaves as an epiphyte3, 49. The host specificity that P syringae displays is at least partly due to pathogen avirulence genes that encode proteins (i.e., Avr proteins) that trigger the disease resistance (R)-gene-based plant innate immune system in resistant plants53, One of the plant defense responses triggered by Avr proteins is the hypersensitive response (HR), which is a programmed cell death of plant tissue that is associated with successful defense against pathogens. P syringae mutants unable to elicit an HR led to the discovery of hrp genes—the genes that encode the P syringae type III proteins secretion system (TT88) 63. P syringae pathogenesis relies on the TT88 and the effector proteins it translocates into host cells similar to most of the animal and plant pathogens that possess TT88s27.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Abramovitch R.B., Kim Y.I, Chen S., Dickman M.B., and Martin G.B., 2003, Pseudomonas type III effector AvrPtoB induces plant disease susceptibility by inhibition of host programmed cell death. EMBO J., 22:60–69.
Alfano J.R., Charkowski A.O., Deng W, Badel J.L., Petnicki-Ocwieja T., van Dijk K., and Collmer A., 2000, The Pseudomonas syringae Hrp pathogenicity island has a tripartite mosaic structure composed of a cluster of type III secretions genes bounded by exchangeable effector and conserved effector loci that contribute to parasitic fitness and pathogenicity in plants. Proc. Natl. Acad. Sci. USA, 97:4856–4861.
Alfano J.R. and Collmer A., 1996, Bacterial pathogens in plants: Life up against the wall. Plant Cell, 8:1683–1698.
Alfano J.R. and Collmer A., 1997, The type III (Hrp) secretion pathway of plant pathogenic bacteria: Trafficking harpins, Avr proteins, and death. J. Bacteriol., 179:5655–5662.
Alfano J.R. and Guo M., 2002, The Pseudomonas syringae Hrp (type III) protein secretion system: Advances in the new millenium, In G. Stacey and N.T. Keen (ed.), Plant-Microbe Interactions, Vol. 6, pp. 227–258. APS Press St. Paul, MN.
Arrieta J., Hernandez L., Coego A., Suarez V., Balmori E., Menendez C., Petit-Glatron M.E, Chambert R., and Selman-Housein G., 1996, Molecular characterization of the levansucrase gene from the endophytic sugarcane bacterium Acetobacter diazotrophicus SRT4. Microbiology, 142:1077–1085.
Asai T., Tena G., Plotnikova J., Willmann M.R., Chiu W-L., Gomez-Gomez L., Boller T., Ausubel EM., and Sheen J., 2002, MAP kinase signalling cascade in Arabidopsis innate immunity. Nature, 415:977–983.
Axtell MJ., Chisholm S.T., Dahlbeck D., and Staskawicz BJ., 2003, Genetic and molecular evidence that the Pseudomonas syringae type III effector protein AvrRpt2 is a cysteine protease. Mol. Microbiol., 49:1537–1546.
Axtell M.J. and Staskawicz B.J., 2003, Initiation of RPS2-specified disease resistance in Arabidopsis is coupled to the AvrRpt2-directed elimination of RIN4. Cell, 112:369–377.
Badel J.L., Charkowski A.O., Deng W.-L., and Collmer A., 2002, A gene in the Pseudomonas syringae pv. tomato Hrp pathogenicity island conserved effector locus, hopPtoA1, contributes to efficient formation of bacterial colonies in planta and is duplicated elsewhere in the genome. Mol. Plant-Microbe Interact., 15:1014–1024.
Badel IL., Nomura K., Bandyopadhyay S., Shimizu R., Collmer A., and He S.Y., 2003, Pseudomonas syringae pv. tomato DC3000 HopPtoM (CEL ORF3) is important for lesion formation but not growth in tomato and is secreted and translocated by the Hrp type III secretion system in a chaperone-dependent manner. Mol. Microbiol., 49:1239–1251.
Bauer D.W. and Collmer A., 1997, Molecular cloning, characterization, and mutagenesis of a pel gene from Pseudomonas syringae pv. lachrymans encoding a member of the Erwinia chrysanthemi PelADE family ofpectate lyases. Mol. Plant-Microbe Interact., 10:369–379.
Bell K.S., Avrova A.O., Holeva M.C., Cardle L., Morris W, De Jong W, Toth I.K., Waugh R., Bryan G.J., and Birch P.R., 2002, Sample sequencing of a selected region of the genome of Erwinia carotovora subsp. atroseptica reveals candidate phytopathogenicity genes and allows comparison with itEscherichia coli. Microbiology, 148:1367–1378.
Bender C.L., Alarcon-Chaidez F., and Gross D.C., 1999, Pseudomonas syringae phytotoxins: Mode of action, regulation, and biosynthesis by peptide and polyketide synthetases. Microbiol. Mol. Biol. Rev., 63:266–292.
Bent A.F., Kunkel B.N., Dahlbeck D., Brown K.L., Schmidt R., Giraudat J., Leung J., and Staskawicz B.J., 1994, RPS2 of Arabidopsis thaliana: A leucine-rich repeat class of plant disease resistance genes. Science, 265:1856–1860.
Binet R., Letoffe S., Ghigo J.M., Delepelaire P., and Wandersman C., 1997, Protein secretion by gram-negative bacterial ABC exporters. Gene, 192:7–11.
Boch J., Joardar V., Gao L., Robertson T.L., Lim M., and Kunkel B.N., 2002, Identification of Pseudomonas syringae pv. tomato genes induced during infection of Arabidopsis thaliana. Mol. Microbiol., 44:73–88.
Boller T., 1995, Chemoperception of microbial signals in plant cells. Annu. Rev. Plant Physiol. Plant Mol. Biol., 46:189–214.
Boyd A.P., Lambermont I., and Cornelis G., 2000, Competition between the Yops of Yersinia enterocolitica for delivery into eukaryotic cells: Role of the SycE chaperone binding domain of YopE. J. Bacteriol., 182:4811–4821.
Braun V. and Braun M., 2002, Iron transport and signaling in Escherichia coli. FEBS Lett., 529:78–85.
Bretz J.R., Mock N.M., Charity J.C., Zeyad S., Baker C.J., and Hutcheson S.W., 2003, A translocated protein tyrosine phosphatase of Pseudomonas syringae pv. tomato DC3000 modulates plant defence response to infection. Mol. Microbiol., 49:389–400.
Brown I., Mansfield J., and Bonas D., 1995, hrp genes in Xanthomonas campestris pv. vesicatoria determine ability to suppress papilla deposition in pepper mesophyll cells. Mol. Plant-Microbe Interact., 8:825–836.
Buell C.R., Joardar V., Lindeberg M., Selengut J., Paulsen I.T., Gwinn M.L., Dodson R.J., Deboy R.T., Durkin A.S., Kolonay J.F. et al., 2003, The complete sequence of the Arabidopsis and tomato pathogen Pseudomonas syringae pv. tomato DC3000. Proc. Natl. Acad. Sci. USA, 100:10181–10186.
Bultreys A. and Gheysen I., 2000, Production and comparison of peptide siderophores from strains of distantly related pathovars of Pseudomonas syringae and Pseudomonas viridiflava LMG 2352, Appl. Environ. Microbiol., 66:325–331.
Casper-Lindley C., Dahlbeck D., Clark E.T., and Staskawicz B.J., 2002, Direct biochemical evidence for type III secretion-dependent translocation of the AvrBs2 effector protein into plant cells. Proc. Natl. Acad. Sci. USA, 99:8336–8341.
Collmer A., Lindeberg M., Petnicki-Ocwieja T., Schneider D., and Alfano J.R., 2002, Genomic mining type III secretion system effectors in Pseudomonas syringae yields new picks for all TTSS prospectors. Trends Microbiol., 10:462–469.
Cornelis G. and van Gijsegem F., 2000, Assembly and function of type III secretion systems. Annu. Rev. Microbiol., 54:734–774.
Comelis P. and Matthijs S., 2002, Diversity of siderophore-mediated iron uptake systems in fluorescent pseudomonads: not only pyoverdines. Environ. Microbiol., 4:787–798.
Coyne M.J., Jr., Russell K.S., Coyle C.L., and Goldberg J.B., 1994, The Pseudomonas aeruginosa algC gene encodes phosphoglucomutase, required for the synthesis of a complete lipopolysaccharide core. J. Bacteriol., 176:3500–3507.
Cuppels D.A., 1986, Generation and characterization of Tn5 insertion mutations in Pseudomonas syringae pv. tomato. Appl. Environ. Microbiol., 51:323–327.
da Silva A.C., Ferro J.A., Reinach F.C., Farah C.S., Furlan L.R., Quaggio R.B., Monteiro-Vitorello C.B., Van Sluys M.A., Almeida N.F., Alves L.M. et al., 2002, Comparison of the genomes of two Xanthomonas pathogens with differing host specificities. Nature, 417:459–463.
Dangl J.L., 1994, The enigmatic avirulence genes of phytopathogenic bacteria. In J.L. Dangl (ed.), Current topics in Microbiology and Immunology: Bacterial Pathogenesis of Plants and Animals—Molecular and Cellular Mechanisms, Vol. 192, pp. 99–118. Springer-Verlag, Berlin.
Deng W.L., Rehm A.H., Charkowski A.O., Rojas C.M., and Collmer A., 2003, Pseudomonas syringae exchangeable effector loci: Sequence diversity in representative pathovars and virulence function in P. syringae pv. syringae B728a. J. Bacteriol., 185:2592–2602.
Espinosa A., Guo M., Tam V.C., Fu Z.Q., and Alfano J.R., 2003, The Pseudomonas syringae type III-secreted protein HopPtoD2 possesses protein tyrosine phosphatase activity and suppresses programmed cell death in plants. Mol. Microbiol., 49:377–387.
Feldman M.F. and Cornelis G.R., 2003, The multitalented type III chaperones: All you can do with 15 kDa. FEMS Microbiol. Lett., 219:151–158.
Fett W.E and Dunn M.F., 1989, Exopolysaccharides produced by phytopathogenic Pseudomonas syringae pathovars in infected leaves of susceptible hosts. Plant Physiol., 89:5–9.
Feys BJ.E, Benedetti C.E., Penfold C.N., and Turner IG., 1994, Arabidopsis mutants selected for resistance to the phytotoxin coronatine are male sterile, insensitive to methyl jasmonate, and resistant to a bacterial pathogen. Plant Cell, 6:751–759.
Fouts D.E., Abramovitch R.B., Alfano J.R., Baldo A.M., Buell C.R., Cartinhour S., Chatterjee A.K., D’Ascenzo M., Gwinn M., Lazarowitz S.G. et al., 2002, Genomewide identification of Pseudomonas syringae pv. tomato DC3000 promoters controlled by the HrpL alternative sigma factor. Proc. Natl.Acad. Sci. USA, 99:2275–2280.
Gacesa P, 1998, Bacterial alginate biosynthesis—recent progress and future prospects. Microbiology, 144:1133–1143.
Gaudriault S., Paulin J P., and Barny M. A., 2002, The DspB/F protein of Erwinia amylovora is a type III secretion chaperone ensuring efficient intrabacterial production of the Hrp-secreted DspA/E pathogenicity factor. Mol. Plant Pathol., 3:313–320.
Glickmann E., Gardan L., Jacquet S., Hussain S., Elasri M., Petit A., and Dessaux Y., 1998, Auxin production is a common feature of most pathovars of Pseudomonas syringae. Mol. Plant-Microbe Interact., 11:156–162.
Gomez-Gomez L. and Boller T., 2000, FLS2: An LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol. Cell, 5:1003–1011.
Goodner B., Hinkle G., Gattung S., Miller N., Blanchard M., Qurollo B., Goldman B.S., Cao Y., Askenazi M., Halling C. et al., 2001, Genome sequence of the plant pathogen and biotechnology agent Agrobacterium tumefaciens C58. Science, 294:2323–2328.
Greenberg J.T. and Vinatzer B.A., 2003: Identifying type III effectors of plant pathogens and analyzing their interaction with plant cells. Curr. Opin. Microbiol., 6:20–28.
Guttman D.S., Vinatzer B.A., Sarkar S.E, Ranall M.V, Kettler G., and Greenberg J.T., 2002, A functional screen for the type III (Hrp) secretome of the plant pathogen Pseudomonas syringae. Science, 295:1722–1726.
Hauck P., Thilmony R., and He S.Y., 2003, A Pseudomonas syringae type III effector suppresses cell wall-based extracellular defense in susceptible Arabidopsis plants. Proc. Natl. Acad. Sci. USA, 100:8577–8582.
Heath M.C., 2000, Hypersensitive response-related death. Plant Mol. Biol., 44:321–334.
Heithoff D.M., Conner C.P., and Mahan M.J., 1997, Dissecting the biology of a pathogen during infection. Trends Microbiol., 5:509–513.
Hirano S.S. and Upper C.D., 2000, Bacterial in the leaf ecosystem with emphasis on Pseudomonas syringae—a pathogen, ice nucleus, and epiphyte. Microbiol. Mol. Biol. Rev., 64:624–653.
Innes R.W., 2001, Targeting the targets of type III effector proteins secreted by phytopathogenic bacteria. Mol. Plant Pathol., 2:109–115.
Innes R.W., Bent A.F., Kunkel B.N., Bisgrove S.R., and Staskawicz B.J., 1993, Molecular analysis of avirulence gene avrRpt2 and identification of a putative regulatory sequence common to all known Pseudomonas syringae avirulence genes. J. Bacteriol., 175:4859–4869.
Jack D.L., Paulsen I.T., and Saier M.H., 2000, The amino acid/polyamine/organocation (APC) superfamily of transporters specific for amino acids, polyamines and organocations. Microbiology, 146:1797–1814.
Keen N.T., 1990, Gene-for-gene complementarity in plant-pathogen interactions. Annu. Rev. Genet., 24:447–463.
Kim J.E, Charkowski A.O., Alfano J.R., Collmer A., and Beer S.V., 1998,Transposable elements and bacteriophage sequences flanking Pseudomonas syringae avirulence genes. Mol. Plant-Microbe Interact., 11:1247–1252.
Kim J.E, Ham J.H., Bauer D.W, Collmer A., and Beer S.V:, 1998, The hrpC and hrpN operons of Erwinia chrysanthemi EC16 are flanked by plcA and homologs of hemolysin/adhesin genes and accompanying activator/transporter genes. Mol. Plant-Microbe Interact., 11:563–567.
Kim, Y.C., Miller C.D., and Anderson A.J., 1999, Transcriptional regulation by iron and role during plant pathogenesis of genes encoding iron-and manganese-superoxide dismutase of Pseudomonassyringae pv.syringae B728a. Physiol. Mol. Plant Pathol., 55:327–339.
Kim Y.J., Lin N.-C., and Martin G.B., 2002, Two distinct Pseudomonas effector proteins interact with the Pto kinase and activate plant immunity. Cell, 109:589–598.
Kiraly Z., El-Zahaby H., Galal A., Abdou S., and Adam A., 1993, Effect of oxygen free radicals on plant pathogenic bacteria and fungi and on some plant diseases, In G. Mozsik, J. Emerit, J. Feher, B. Matkovics, and A. Vincze (ed.), Oxygen Free Radicals and Scavengers in the Natural Sciences, pp. 9–19. Akademiai Kiado Budapest.
Kloek A.P., Verbsky M.L., Sharma S.B., Schoelz JE., Vogel J., Klessig D.F., and Kunkel B.N., 2001, Resistance to Pseudomonas syringae conferred by an Arabidopsis thaliana coronatine-insensitive (coil) mutation occurs through two distinct mechanisms. Plant J., 26:509–522.
Klotz M.G. and Hutcheson S.W., 1992, Multiple periplasmic catalases in phytopathogenic strains of Pseudomonassyringae. Appl. Environ. Microbiol., 58:2468–2473.
Leach J.E. and White F.F., 1996, Bacterial avirulence genes. Annu. Rev. Phytopathol., 34:153–179.
Li H. and Ullrich M.S., 2001, Characterization and mutational analysis of three allelic lsc genes encoding levansucrase in Pseudomonas syringae. J. Bacteriol., 183:3282–3292.
Lindgren P.B., Peet R.C., and Panopoulos N.J.;1986, Genecluster of Pseudomonas syringae pv.phaseolicola controls pathogenicity of bean plants and hypersensitivity on nonhost plants. J. Bacteriol., 168:512–522.
Lloyd S.A., Norman M., Rosqvist R., and Wolf-Watz H., 2001, Yersinia YopE is targeted for type III secretion by N-terminal, not mRNA, signals. Mol. Microbiol., 39:520–531.
Lloyd S.A., Sjostrom M., Andersson S., and Wolf-Watz H., 2002, Molecular characterization of type III secretion signals via analysis of synthetic N-terminal amino acid sequences. Mol. Microbiol., 43:51–59.
Locht C., Antoine R., and Jacob-Dubuisson F., 2001, Bordetella pertussis, molecular pathogenesis under multiple aspects. Curr. Opin. Microbiol., 4:82–89.
Ma Q., Zhai Y., Schneider J.C., Ramseier T.M., and Saier M.H., Jr., 2003, Protein secretion systems of Pseudomonas aeruginosa and P. fluorescens. Biochim. Biophys. Acta, 1611:223–233.
Mackey D., Belkhadir Y., Alonso J.M., Ecker J.R., and Dangl J.L., 2003, Arabidopsis RIN4 is a target of the type III virulence effect or AvrRpt2 and modulates RPS2-mediated resistance. Cell, 112:379–389.
Mahan M.J., Slauch J.M., and Mekalanos J.J., 1993, Selection of bacterial virulence genes that are specifically induced in host tissues. Science, 259:686–688.
Mazzola M. and White F.F., 1994, A mutation in the indole-3-acetic acid biosynthesis pathway of Pseudomonas syringae pv. syringae affects growth in Phaseolus vulgaris and syringomycin production. J. Bacteriol., 176:1374–1382.
Medzhitov R. and Janeway C.A., Jr., 1997, Innate immunity: The virtues of a nonclonal system of recognition. Cell, 91:295–298.
Miller D.A., Luo L., Hillson N., Keating T.A., and Walsh C.T., 2002, Yersiniabactin synthetase: a four-protein assembly line producing the non ribosomal peptide/polyketide hybrid siderophore of Yersinia pestis. Chem. Biol., 9:333–344.
Mindrinos M., Katagiri E, Yu G.-L., and Ausubel F.M., 1994, The A. thaliana disease resistance gene RPS2 encodes a protein containing a nucleotide-binding site and leucine-rich repeats. Cell, 78:1089–1099.
Mittler R., 2002, Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci., 7:405–410.
Moore R.A., Starratt A.N., Ma S.-W, Morris V.L., and Cuppels D.A., 1989, Identification of a chromosomal region required for biosynthesis of the phytoxin coronatine by Pseudomonas syringae pv. tomato. Can. J. Microbiol., 35:910–917.
Nelson K.E., Weinel C., Paulsen I.T., Dodson RJ., Hilbert H., Martins dos Santos V.A., Fouts D.E., Gill S.R., Pop M., Holmes M. et al., 2002, Complete genome sequence and comparative analysis of the metabolically versatile Pseudomonas putida KT2440. Environ. Microbiol., 4:799–808.
Nurnberger T. and Scheel D., 2001, Signal transmission in the plant immune response. Trends Plant Sci., 6:372–379.
Osbourn A.E., Barber C.E., and Daniels MJ., 1987, Identification of plant-induced genes of the bacterial pathogen Xanthomonas campestris pathovar campestris using a promoter-probe plasmid. EMBO, 6:23–28.
Overmyer K., Brosche M., and Kangasjarvi J., 2003, Reactive oxygen species and hormonal control of cell death. Trends Plant Sci., 8:335–342.
Parsot C., Hamiaux C., and Page A.L., 2003, The various and varying roles of specific chaperones in type III secretion systems. Curr. Opin. Microbiol., 6:7–14.
Petnicki-Ocwieja T., Schneider DJ., Tam V.C., Chancey S.T., Shan L., Jamir Y., Schechter L.M., Janes M.D., Buell C.R., Tang X. et al., 2002, Genomewide identification of proteins secreted by the Hrp type III protein secretion system of Pseudomonas syringae pv. tomato DC3000. Proc. Natl. Acad. Sci. USA, 99:7652–7657.
Rainey P.B. and Preston G.M., 2000, In vivo expression technology stratagies: valuable tools for biotechnology. Curr. Opin. Microbiol., 11:440–444.
Reymond P and Farmer E.E., 1998, Jasmonate and salicylate as global signals for defense gene expression. Curr. Opin. Plant Biol., 1:404–411.
Roine E., Raineri D.M., Romantschuk M., Wilson M., and Nunn D.N., 1998, Characterization of type IV pilus genes in Pseudomonas syringae pv. tomato DC3000. Mol. Plant-Microbe Interact., 11:1048–1056.
Rojas C.M., Ham J.H., Deng WL., Doyle J.J., and Collmer A., 2002, HecA, a member of a class of adhesins produced by diverse pathogenic bacteria, contributes to the attachment, aggregation, epidermal cell killing, and virulence phenotypes of Erwinia chrysanthemi EC16 on Nicotiana clevelandii seedlings. Proc. Natl. Acad. Sci. USA, 99: 13142–13147.
Romantschuk M., 1992, Attachment of plant pathogenic bacteria to plant surfaces. Annu. Rev. Phytopathol., 30:225–243.
Salanoubat M., Genin S., Artiguenave F., Gouzy J., Mangenot S., Arlat M., Billault A., Brottier P., Camus J.C., Cattolico L. et al. 2002, Genome sequence of the plant pathogen Ralstonia solanacearum. Nature, 415:497–502.
Salmond G.P.C., 1994, Secretion of extracellular virulence factors by plant pathogenic bacteria. Annu. Rev. Phytopathol., 32:181–200.
Sandkvist M., 2001, Biology of type II secretion. Mol. Microbiol., 40:271–283.
Santos R., Franza T., Laporte M.L., Sauvage C., Touati D., and Expert D., 2001, Essential role of superoxide dismutase on the pathogenicity of Erwinia chrysanthemi strain 3937. Mol. Plant-Microbe Interact., 14:758–767.
Shao E, Golstein C., Ade J., Stoutemyer M., Dixon J.E., and Innes R.W, 2003, Cleavage of Arabidopsis PBS1 by a bacterial type III effector. Science, 301:1230–1233.
Shao F., Merritt P.M., Bao Z., Innes R.W, and Dixon J.E., 2002, A Yersinia effector and a Pseudomonas avirulence protein define a family of cysteine proteases functioning in bacterial pathogenesis. Cell, 109:575–588.
Shen H. and Keen N.T., 1993, Characterization of the promoter of avirulence gene D from Pseudomonas syringae pv. tomato. J. Bacteriol., 175:5916–5924.
Simpson A.J.G., Reinach F.C., Arruda P., Abreu F.A., Acencio M., Alvarenga R., Alves L.M.C., Araya J.E., Baia G.S., Baptista C.S. et al., 2000, The genome sequence of the plant pathogen Xylella fastidiosa. Nature, 406: 151–157.
Solomon P.S. and Oliver R.P., 2001, The nitrogen content of the tomato leaf apoplast increases during infection by Cladosporium fulvum. Planta, 213:241–249.
Sory M.P. and Cornelis G.R., 1994, Translocation of a hybrid YopE-adenylate cyclase from Yersinia enterocolitica into HeLa cells. Mol. Microbiol., 14:583–594.
Stover C.K., Pham X.Q., Erwin A.L., Mizoguchi S.D., Warrener P., Hickey M.J., Brinkman F.S., Hufnagle WO., Kowalik D.J., Lagrou M. et al., 2000, Complete genome sequence of Pseudomonas aeruginosa PA01, an opportunistic pathogen. Nature, 406:959–964.
Thomas N.A. and Finlay B.B., 2003, Establishing order for type III secretion substrates—a hierarchical process. Trends Microbiol., 11:398–403.
van Dijk K., Tam V.C., Records A.R., Petnicki-Ocwieja T., and Alfano J.R., 2002, The ShcA protein is a molecular chaperone that assists in the secretion of the HopPsyA effector from the type III (Hrp) protein secretion system of Pseudomonas syringae. Mol. Microbiol., 44: 1469–1481.
Wall D. and Kaiser D., 1999, Type IV pili and cell motility. Mol. Microbiol., 32:1–10.
Whalen M.C., Innes R.W., Bent A.F., and Staskawicz B.J., 1991, Identification of Pseudomonas syringae pathogens of Arabidopsis and a bacterial locus determining avirulence on both Arabidopsis and soybean. Plant Cell, 3:49–59.
Wood D.W, Setubal J.C., Kaul R., Monks D.E., Kitajima J.P., Okura V.K., Zhou Y., Chen L., Wood G.E., Almeida N.F. et al., 2001, The genome of the natural genetic engineer Agrobacterium tumefaciens C58. Science, 294:2317–2323.
Xiao Y., Heu S., Yi J., Lu Y., and Hutcheson S.W, 1994, Identification of a putative alternate sigma factor and characterization of a multicomponent regulatory cascade controlling the expression of Pseudomonas syringae pv. syringae Pss61 hrp and hrmA genes. J Bacteriol., 176:1025–1036.
Xiao Y. and Hutcheson S.W., 1994, A single promoter sequence recognized by a newly identified alternate sigma factor directs expression of pathogenicity and host range determinants in Pseudomonas syringae. J. Bacteriol., 176:3089–3091.
Xu X.Q. and Pan S.Q., 2000, An Agrobacterium catalase is a virulence factor involved in tumorigenesis. Mol. Microbiol., 35:407–414.
Young J.M., Saddler G.S., Takikawa Y., De Boer S.H., Vauterin L., Gardan L., Gvozdyak R.I., and Stead D.E., 1996, Names of plant pathogenic bacteria 1864–1995. Rev. Plant Pathol., 75:721–763.
Yu J., Penaloza-Vazquez A., Chakrabarty A.M., and Bender C.L., 1999, Involvement of the exopolysaccharide alginate in the virulence and epiphytic fitness of Pseudomonas syringae pv. syringae. Mol. Microbiol., 33:712–720.
Zwiesler-Vollick J., Plovanich-Jones A.E., Nomura K., Bandyopadhyay S., Joardar V., Kunkel B.N., and He S.Y., 2002, Identification of novel hrp-regulated genes through functional genomic analysis of the Pseudomonas syringae pv. tomato DC3000 genome. Mol. Microbiol., 45:1207–1218.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2004 Springer Science+Business Media New York
About this chapter
Cite this chapter
Jamir, Y., Tang, X., Alfano, J.R. (2004). The Genome of Pseudomonas syringae Tomato DC3000 and Functional Genomic Studies to Better Understand Plant Pathogenesis. In: Ramos, JL. (eds) Pseudomonas. Springer, Boston, MA. https://doi.org/10.1007/978-1-4419-9086-0_4
Download citation
DOI: https://doi.org/10.1007/978-1-4419-9086-0_4
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-4788-0
Online ISBN: 978-1-4419-9086-0
eBook Packages: Springer Book Archive